Key Insights
The global Vaccine Safety Solutions market is poised for significant expansion, projected to reach a market size of $9984 million by 2025. This robust growth is underpinned by a compelling Compound Annual Growth Rate (CAGR) of 7.4%, indicating a steady and sustained upward trajectory throughout the forecast period of 2025-2033. The increasing emphasis on maintaining vaccine integrity from storage to administration is a primary catalyst. This includes a rising demand for advanced cold chain solutions, temperature monitoring systems, and secure storage units, driven by global vaccination initiatives, the expanding pharmaceutical industry, and the growing need for reliable healthcare infrastructure. The market's expansion is further fueled by technological advancements in refrigeration, IoT-enabled monitoring, and specialized containment solutions designed to prevent spoilage and ensure efficacy.
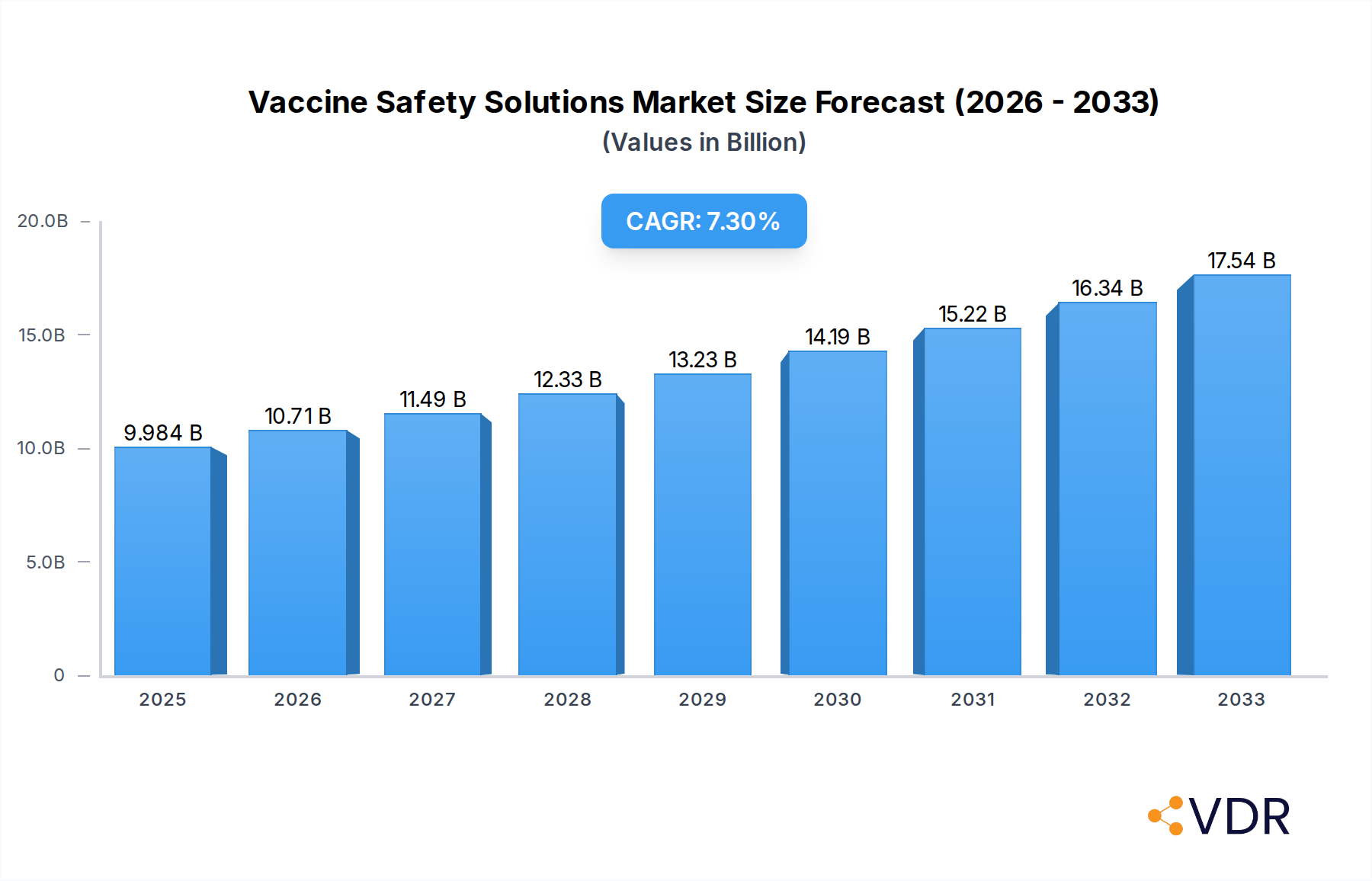
Vaccine Safety Solutions Market Size (In Billion)

The market's segmentation reveals a strong focus on the Healthcare and Hospitals application segment, reflecting its critical role in vaccine distribution and patient care. The Veterinary segment also presents a growing opportunity as the importance of animal health and disease prevention gains traction. Within the Type segmentation, both Equipment and Services are expected to witness considerable demand, highlighting the integrated nature of vaccine safety solutions. Key players like Thermo Fisher Scientific, PHC Holdings Corporation, and Haier Biomedical are instrumental in driving innovation and market penetration. Geographically, North America and Europe are expected to lead in market share due to established healthcare systems and early adoption of advanced technologies, while the Asia Pacific region is anticipated to exhibit the highest growth rate, spurred by increasing healthcare investments and expanding vaccination programs. The market is characterized by a dynamic interplay of drivers such as increasing vaccine production, stringent regulatory compliance, and the growing prevalence of temperature-sensitive biologics, alongside restraints like high initial investment costs for sophisticated equipment and the need for continuous power supply.
Vaccine Safety Solutions Company Market Share

This comprehensive report offers an in-depth analysis of the global Vaccine Safety Solutions market, providing critical insights into its dynamics, growth trajectory, and future potential. Spanning from 2019 to 2033, with a base and estimated year of 2025 and a forecast period of 2025–2033, this study is essential for stakeholders seeking to understand and capitalize on the evolving landscape of vaccine storage, transportation, and monitoring technologies. The report delves into the parent market's robust growth, driven by the increasing global demand for vaccine accessibility and efficacy, and further examines the child markets of specialized equipment, essential services, and auxiliary solutions that support the broader vaccine cold chain.
Vaccine Safety Solutions Market Dynamics & Structure
The global Vaccine Safety Solutions market exhibits a moderately consolidated structure, with a few key players holding significant market share, alongside a growing number of specialized providers. Technological innovation is a primary driver, focusing on enhanced temperature control, real-time monitoring, and smart logistics solutions to ensure vaccine integrity from production to administration. Stringent regulatory frameworks, such as those enforced by the WHO and national health agencies, mandate high standards for cold chain management, creating a strong demand for compliant solutions. Competitive product substitutes include advancements in passive cooling technologies and improved supply chain visibility tools. End-user demographics are diverse, ranging from large public health organizations and national immunization programs to private healthcare providers and veterinary clinics, each with specific needs and purchasing power. Mergers and acquisitions (M&A) trends are evident as larger companies seek to expand their product portfolios and geographical reach, aiming to offer end-to-end cold chain solutions.
- Market Concentration: Approximately 65% market share is held by the top 5 players, with the remaining 35% distributed among mid-tier and emerging companies.
- Technological Innovation Drivers: Miniaturization of monitoring devices, AI-powered predictive analytics for cold chain breaches, and advanced insulation materials.
- Regulatory Frameworks: Compliance with Good Distribution Practices (GDP) and WHO guidelines for vaccine storage temperature ranges (e.g., +2°C to +8°C, -20°C, -80°C).
- Competitive Product Substitutes: Smart packaging solutions, blockchain-enabled supply chain tracking, and advanced last-mile delivery innovations.
- End-User Demographics: Public health agencies (55% of demand), private hospitals and clinics (30%), veterinary sector (10%), research institutions (5%).
- M&A Trends: 8 significant M&A deals were recorded in the historical period (2019-2024), primarily focused on integrating IoT capabilities and expanding cold chain infrastructure.
Vaccine Safety Solutions Growth Trends & Insights
The global Vaccine Safety Solutions market is experiencing robust growth, propelled by an increasing awareness of vaccine efficacy and the imperative to maintain the cold chain integrity throughout the supply chain. The market size, valued at approximately $7,500 million in 2024, is projected to witness a Compound Annual Growth Rate (CAGR) of 7.8% during the forecast period, reaching an estimated $13,000 million by 2033. This upward trajectory is significantly influenced by recurring vaccination campaigns, the growing prevalence of infectious diseases, and the expanding global immunization programs, particularly in developing economies with nascent cold chain infrastructure. Adoption rates for advanced cold chain monitoring technologies, such as Internet of Things (IoT) enabled sensors and cloud-based data management systems, are accelerating as organizations recognize their value in minimizing vaccine wastage and ensuring patient safety. Technological disruptions, including the development of ultra-low temperature storage solutions for novel vaccines and the integration of artificial intelligence for predictive maintenance of cold chain equipment, are further reshaping the market. Consumer behavior shifts are also playing a crucial role, with increased demand for transparent and verifiable vaccine supply chains, fostering trust and accountability. The historical period (2019-2024) witnessed a substantial increase in market penetration, driven by the global response to pandemics and the need for rapid vaccine deployment. The transition from traditional manual monitoring to automated, digital solutions is a key trend, enhancing efficiency and reducing human error. Furthermore, the increasing complexity of vaccine formulations, requiring precise temperature control, is spurring innovation in specialized storage equipment. The market's responsiveness to global health emergencies underscores its critical role in public health infrastructure.
Dominant Regions, Countries, or Segments in Vaccine Safety Solutions
The Healthcare and Hospitals application segment is the dominant force driving growth in the global Vaccine Safety Solutions market, consistently accounting for a significant portion of the market share, estimated at 68% in 2025. This dominance is attributed to the critical role hospitals and healthcare facilities play in vaccine storage, distribution, and administration to diverse patient populations. North America, particularly the United States, emerges as the leading country, driven by its advanced healthcare infrastructure, substantial investment in public health initiatives, and a high demand for specialized vaccine storage and logistics solutions. The region's robust regulatory environment and strong emphasis on patient safety further bolster the market.
Dominant Application Segment:
- Healthcare and Hospitals: This segment accounts for approximately $5,200 million of the total market value in 2025.
- Key Drivers: High volume of vaccine administration, need for diverse temperature-controlled storage (refrigerated, frozen, ultra-low temperature), stringent regulatory compliance, and demand for real-time monitoring for critical vaccines.
- Market Share: Holds an estimated 68% of the application market in 2025.
- Growth Potential: Continuous expansion due to aging populations, increasing chronic disease management requiring routine immunizations, and the constant introduction of new vaccine types.
Dominant Type Segment:
- Equipment: This segment, encompassing refrigerators, freezers, ultra-low temperature freezers, and temperature monitoring devices, represents the largest share of the market, valued at approximately $7,100 million in 2025.
- Key Drivers: Essential for maintaining vaccine integrity across various temperature requirements, technological advancements in energy efficiency and temperature precision, and the need for reliable cold chain infrastructure.
- Market Share: Accounts for an estimated 78% of the type market in 2025.
- Growth Potential: Steady growth driven by the replacement of older equipment, the need for specialized units for new vaccine formulations, and expansion in regions with developing cold chain capabilities.
Leading Region:
- North America: Expected to hold a market share of 35% in 2025, with an estimated market value of $3,500 million.
- Key Drivers: High healthcare expenditure, strong government support for public health, advanced technological adoption, presence of major pharmaceutical companies and research institutions, and a well-established cold chain network.
- Growth Potential: Continued expansion driven by ongoing vaccination programs, the development of new vaccines, and investments in modernizing existing cold chain infrastructure.
Vaccine Safety Solutions Product Landscape
The Vaccine Safety Solutions product landscape is characterized by a continuous stream of innovations aimed at enhancing reliability, efficiency, and traceability. Key product categories include advanced medical refrigerators and freezers with precise temperature controls, ultra-low temperature freezers for mRNA vaccines, and sophisticated temperature monitoring systems leveraging IoT technology for real-time data transmission and alerts. Smart vaccine refrigerators with integrated data loggers and remote access capabilities are becoming standard, ensuring compliance with strict cold chain regulations. Innovations in insulation materials are leading to more energy-efficient and robust storage units. The emphasis is on providing solutions that not only maintain the required temperature but also offer comprehensive data management for audit trails and compliance reporting, thereby ensuring the efficacy and safety of vaccines from the point of manufacture to administration.
Key Drivers, Barriers & Challenges in Vaccine Safety Solutions
Key Drivers:
- Increasing Global Vaccination Rates: Driven by public health initiatives and the need to combat infectious diseases, this significantly boosts the demand for reliable vaccine storage and transport.
- Technological Advancements: Innovations in IoT, AI, and advanced refrigeration technology enhance cold chain monitoring, efficiency, and vaccine integrity.
- Stringent Regulatory Requirements: Global health organizations and national agencies mandate strict temperature control, driving the adoption of compliant solutions.
- Growing Demand for Novel Vaccines: The development of vaccines requiring ultra-low temperature storage creates a need for specialized equipment.
Key Barriers & Challenges:
- High Initial Investment Costs: Specialized cold chain equipment and advanced monitoring systems can be expensive, posing a challenge for smaller healthcare providers and developing regions.
- Infrastructure Gaps: Inadequate power supply and a lack of robust cold chain infrastructure in certain regions hinder widespread adoption.
- Supply Chain Complexities: Managing the cold chain across vast geographical areas and diverse climatic conditions presents logistical challenges.
- Skilled Workforce Shortage: A lack of trained personnel to operate and maintain sophisticated cold chain equipment can impede effective utilization.
- Counterfeit Products and Tampering: Ensuring the authenticity and integrity of vaccines throughout the supply chain remains a concern.
Emerging Opportunities in Vaccine Safety Solutions
Emerging opportunities in the Vaccine Safety Solutions market lie in the expanding use of digital technologies for enhanced cold chain management. The integration of AI and machine learning for predictive analytics to anticipate potential cold chain breaches and optimize inventory management presents a significant avenue for growth. Furthermore, the development of more sustainable and energy-efficient cold chain solutions is gaining traction, aligning with global environmental concerns. Untapped markets in emerging economies with growing immunization programs offer substantial potential for market expansion, particularly for affordable and robust vaccine storage and transport solutions. The increasing demand for home-based healthcare and specialized vaccine delivery services also opens up new application areas for portable and smart cold chain devices.
Growth Accelerators in the Vaccine Safety Solutions Industry
Several catalysts are accelerating the growth of the Vaccine Safety Solutions industry. Technological breakthroughs in ultra-low temperature storage and advanced monitoring systems are continuously expanding the capabilities and reliability of cold chain solutions. Strategic partnerships between equipment manufacturers, logistics providers, and pharmaceutical companies are streamlining the supply chain and fostering integrated solutions. Market expansion strategies, particularly targeting underserved regions and emerging economies, are crucial for capturing new customer bases. The ongoing research and development of novel vaccines requiring stringent temperature control further propel the need for advanced cold chain infrastructure. The increasing focus on pharmaceutical supply chain security and the prevention of vaccine wastage are also significant growth accelerators.
Key Players Shaping the Vaccine Safety Solutions Market
- Thermo Fisher Scientific
- PHC Holdings Corporation (Panasonic Healthcare)
- Haier Biomedical
- Helmer Scientific
- B Medical Systems
- Eppendorf AG
- Vestfrost Solutions
- Follett LLC
- Arctiko A/S
- So-Low Environmental Equipment Co.
Notable Milestones in Vaccine Safety Solutions Sector
- 2020 May: Development and widespread deployment of ultra-low temperature freezers crucial for the storage of early mRNA COVID-19 vaccines.
- 2021 January: Introduction of advanced IoT-enabled temperature monitoring systems for real-time tracking of vaccine shipments globally.
- 2022 March: Increased investment in cold chain infrastructure for routine childhood immunizations in developing nations.
- 2023 June: Launch of highly energy-efficient vaccine refrigerators, addressing sustainability concerns in cold chain management.
- 2024 February: Expansion of smart packaging solutions with integrated temperature indicators for last-mile vaccine delivery.
In-Depth Vaccine Safety Solutions Market Outlook
The future of the Vaccine Safety Solutions market is exceptionally promising, characterized by continued innovation and expansion. Growth accelerators such as advancements in cold chain automation, the integration of blockchain for enhanced traceability, and the development of decentralized cold chain solutions will be pivotal. Strategic alliances and collaborative efforts among industry players will further optimize supply chains and address complex logistical challenges. The increasing global emphasis on public health security and the preparedness for future pandemics will sustain the demand for robust and reliable vaccine safety solutions. This foresight ensures that the market is well-positioned to meet the evolving needs of vaccine distribution and administration worldwide.
Vaccine Safety Solutions Segmentation
-
1. Application
- 1.1. Healthcare and Hospitals
- 1.2. Veterinary
- 1.3. Other
-
2. Type
- 2.1. Equipment
- 2.2. Services
- 2.3. Other
Vaccine Safety Solutions Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
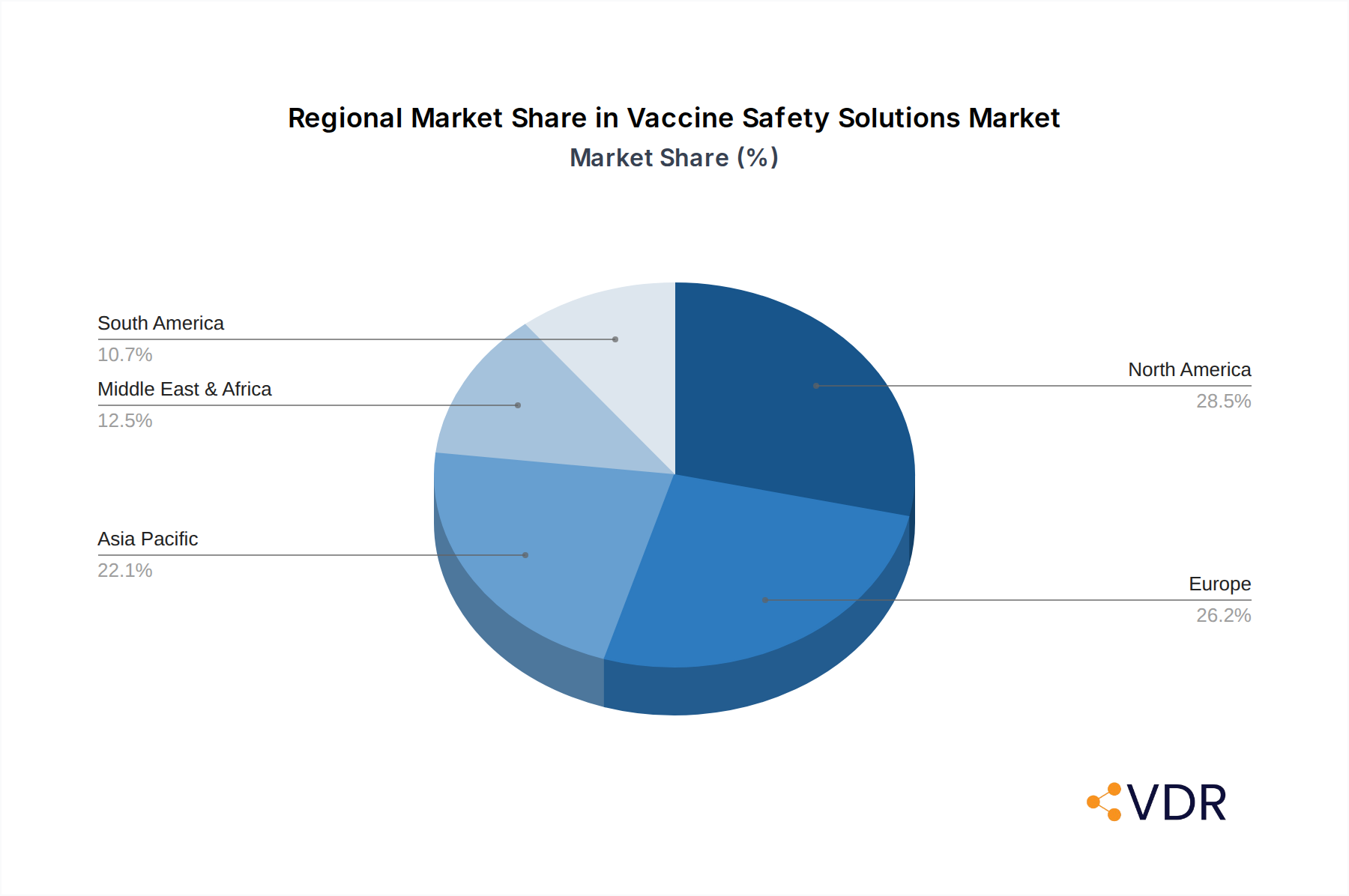
Vaccine Safety Solutions Regional Market Share

Geographic Coverage of Vaccine Safety Solutions
Vaccine Safety Solutions REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Healthcare and Hospitals
- 5.1.2. Veterinary
- 5.1.3. Other
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Equipment
- 5.2.2. Services
- 5.2.3. Other
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Vaccine Safety Solutions Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Healthcare and Hospitals
- 6.1.2. Veterinary
- 6.1.3. Other
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Equipment
- 6.2.2. Services
- 6.2.3. Other
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Vaccine Safety Solutions Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Healthcare and Hospitals
- 7.1.2. Veterinary
- 7.1.3. Other
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Equipment
- 7.2.2. Services
- 7.2.3. Other
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Vaccine Safety Solutions Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Healthcare and Hospitals
- 8.1.2. Veterinary
- 8.1.3. Other
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Equipment
- 8.2.2. Services
- 8.2.3. Other
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Vaccine Safety Solutions Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Healthcare and Hospitals
- 9.1.2. Veterinary
- 9.1.3. Other
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Equipment
- 9.2.2. Services
- 9.2.3. Other
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Vaccine Safety Solutions Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Healthcare and Hospitals
- 10.1.2. Veterinary
- 10.1.3. Other
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Equipment
- 10.2.2. Services
- 10.2.3. Other
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Vaccine Safety Solutions Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Healthcare and Hospitals
- 11.1.2. Veterinary
- 11.1.3. Other
- 11.2. Market Analysis, Insights and Forecast - by Type
- 11.2.1. Equipment
- 11.2.2. Services
- 11.2.3. Other
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Thermo Fisher Scientific
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 PHC Holdings Corporation (Panasonic Healthcare)
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Haier Biomedical
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Helmer Scientific
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 B Medical Systems
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Eppendorf AG
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Vestfrost Solutions
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Follett LLC
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Arctiko A/S
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 So-Low Environmental Equipment Co.
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.1 Thermo Fisher Scientific
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Vaccine Safety Solutions Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Vaccine Safety Solutions Revenue (million), by Application 2025 & 2033
- Figure 3: North America Vaccine Safety Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Vaccine Safety Solutions Revenue (million), by Type 2025 & 2033
- Figure 5: North America Vaccine Safety Solutions Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Vaccine Safety Solutions Revenue (million), by Country 2025 & 2033
- Figure 7: North America Vaccine Safety Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Vaccine Safety Solutions Revenue (million), by Application 2025 & 2033
- Figure 9: South America Vaccine Safety Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Vaccine Safety Solutions Revenue (million), by Type 2025 & 2033
- Figure 11: South America Vaccine Safety Solutions Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Vaccine Safety Solutions Revenue (million), by Country 2025 & 2033
- Figure 13: South America Vaccine Safety Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Vaccine Safety Solutions Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Vaccine Safety Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Vaccine Safety Solutions Revenue (million), by Type 2025 & 2033
- Figure 17: Europe Vaccine Safety Solutions Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Vaccine Safety Solutions Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Vaccine Safety Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Vaccine Safety Solutions Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Vaccine Safety Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Vaccine Safety Solutions Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa Vaccine Safety Solutions Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Vaccine Safety Solutions Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Vaccine Safety Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Vaccine Safety Solutions Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Vaccine Safety Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Vaccine Safety Solutions Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific Vaccine Safety Solutions Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Vaccine Safety Solutions Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Vaccine Safety Solutions Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Vaccine Safety Solutions Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Vaccine Safety Solutions Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global Vaccine Safety Solutions Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Vaccine Safety Solutions Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Vaccine Safety Solutions Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global Vaccine Safety Solutions Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Vaccine Safety Solutions Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Vaccine Safety Solutions Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global Vaccine Safety Solutions Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Vaccine Safety Solutions Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Vaccine Safety Solutions Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global Vaccine Safety Solutions Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Vaccine Safety Solutions Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Vaccine Safety Solutions Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global Vaccine Safety Solutions Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Vaccine Safety Solutions Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Vaccine Safety Solutions Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global Vaccine Safety Solutions Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Vaccine Safety Solutions Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Vaccine Safety Solutions?
The projected CAGR is approximately 7.4%.
2. Which companies are prominent players in the Vaccine Safety Solutions?
Key companies in the market include Thermo Fisher Scientific, PHC Holdings Corporation (Panasonic Healthcare), Haier Biomedical, Helmer Scientific, B Medical Systems, Eppendorf AG, Vestfrost Solutions, Follett LLC, Arctiko A/S, So-Low Environmental Equipment Co..
3. What are the main segments of the Vaccine Safety Solutions?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 9984 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Vaccine Safety Solutions," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Vaccine Safety Solutions report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Vaccine Safety Solutions?
To stay informed about further developments, trends, and reports in the Vaccine Safety Solutions, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
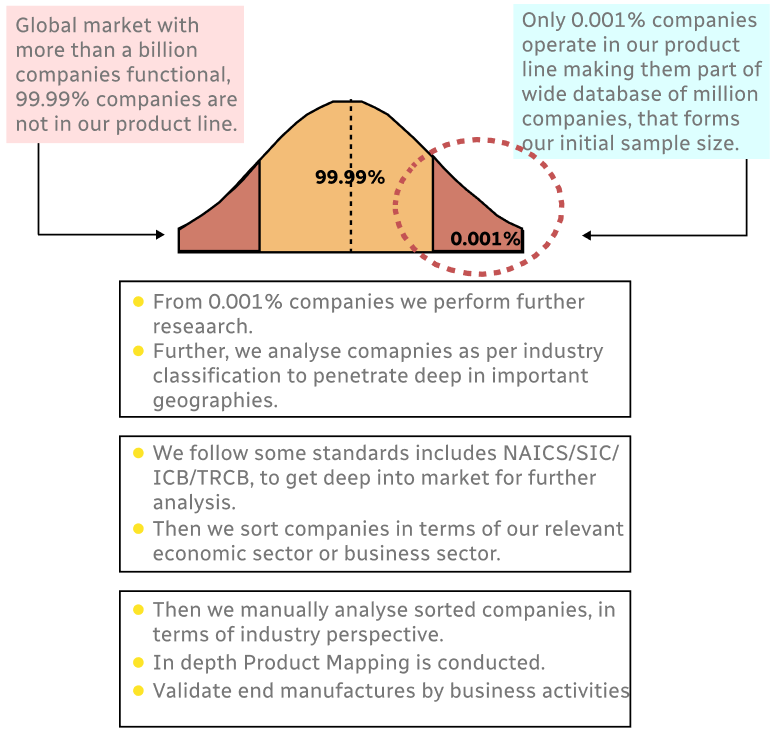
Step 1 - Identification of Relevant Samples Size from Population Database
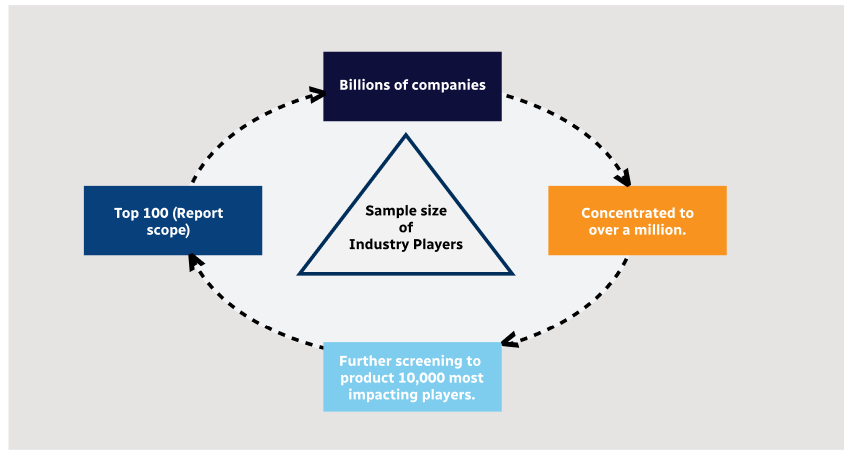
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
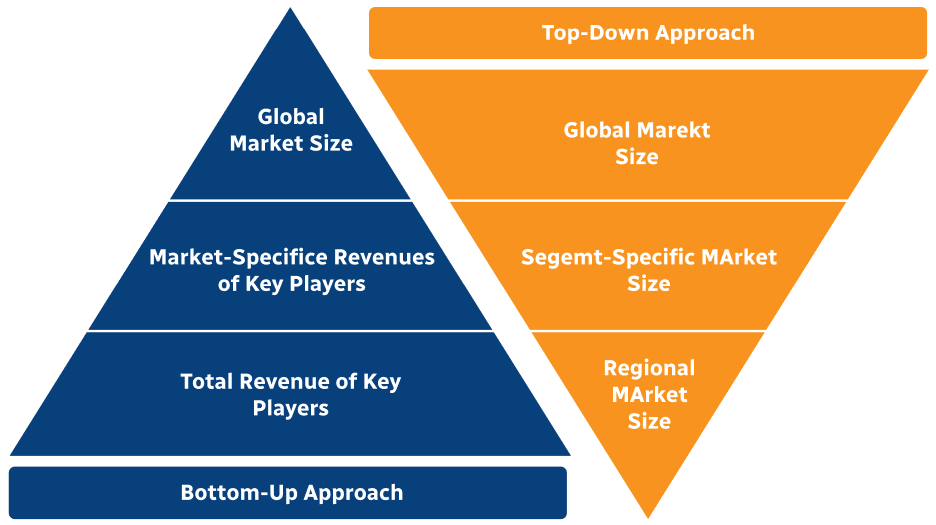
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
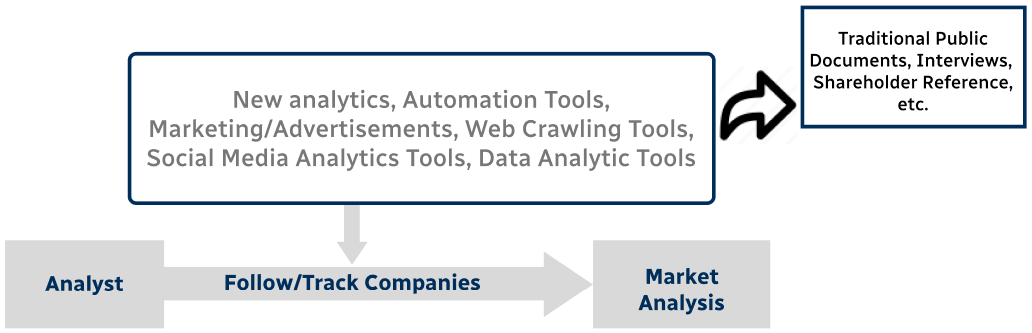
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


