Key Insights
The United States endoscopy devices market is poised for significant expansion, projected to reach $12.17 million by 2025, driven by an anticipated Compound Annual Growth Rate (CAGR) of 6.39% through 2033. This robust growth is primarily fueled by the increasing prevalence of gastrointestinal disorders, the rising demand for minimally invasive surgical procedures, and continuous technological advancements in endoscopic imaging and instrumentation. The aging population in the US, a demographic group more susceptible to chronic diseases requiring endoscopic intervention, further underpins this upward trajectory. Furthermore, increased healthcare expenditure, growing awareness among patients regarding the benefits of early diagnosis through endoscopy, and favorable reimbursement policies are also contributing factors. The market is segmented by device type, including rigid endoscopes, flexible endoscopes, capsule endoscopes, and robot-assisted endoscopes, with flexible endoscopes expected to dominate due to their versatility. Endoscopic operative devices, such as irrigation/suction systems and access devices, are also critical components of this growing market. Visualization equipment is another key segment, enabling precise diagnostics and interventions.
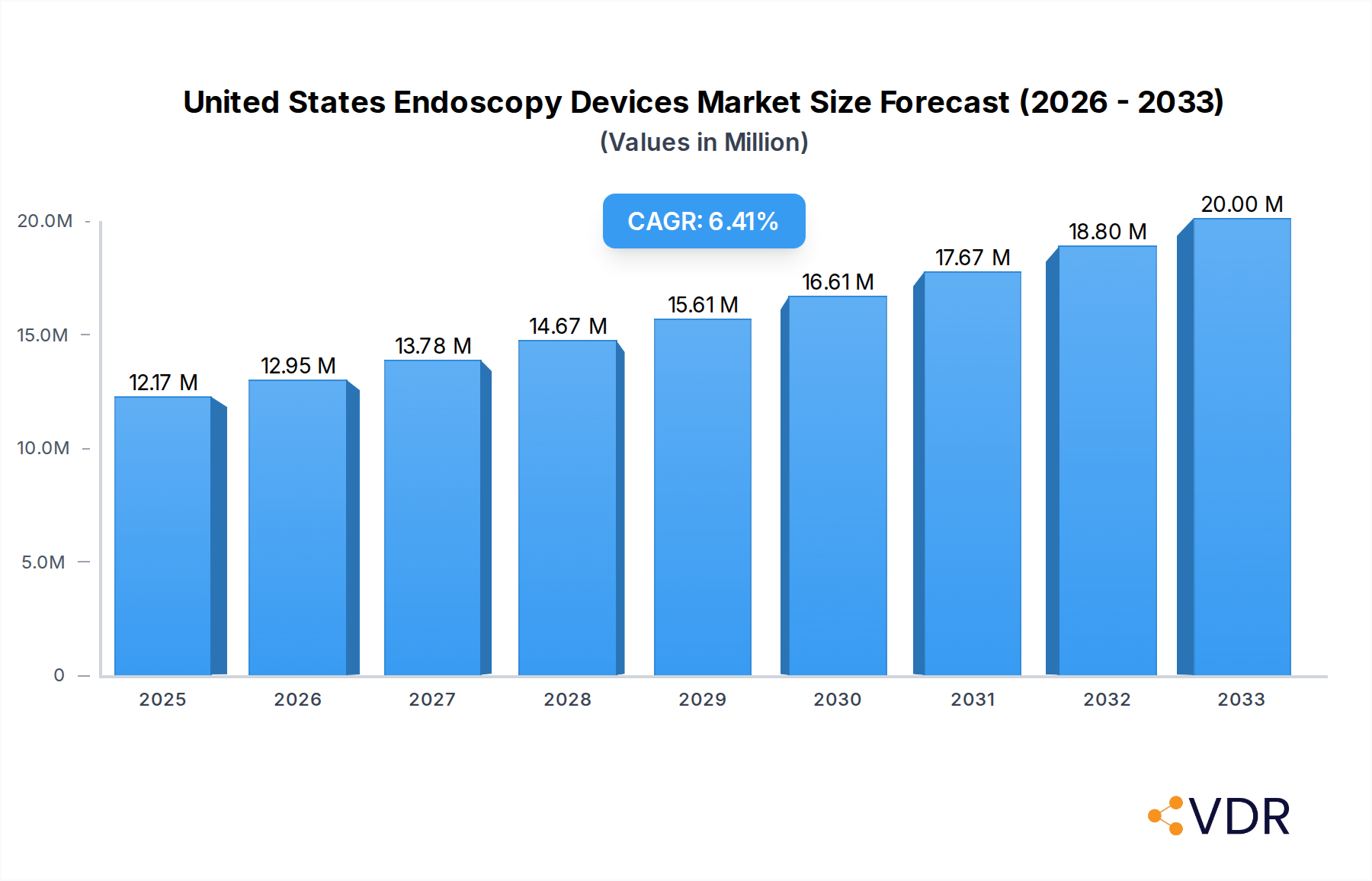
United States Endoscopy Devices Market Market Size (In Million)

The market's expansion is further propelled by advancements in AI-integrated endoscopy, robotic surgery, and the development of smaller, more sophisticated capsule endoscopes offering enhanced patient comfort and diagnostic accuracy. Gastroenterology remains the largest application segment, owing to the high incidence of digestive system diseases. However, significant growth is also anticipated in pulmonology, ENT surgery, and neurology, as endoscopic techniques become standard of care in these fields. While the market exhibits strong growth potential, certain restraints such as the high cost of advanced endoscopic equipment and the need for specialized training for healthcare professionals could present challenges. Despite these, the sustained demand for improved diagnostic and therapeutic outcomes, coupled with ongoing innovation from leading companies like Medtronic, Johnson & Johnson, and Olympus, positions the United States endoscopy devices market for a dynamic and prosperous future.
United States Endoscopy Devices Market Company Market Share

United States Endoscopy Devices Market: Comprehensive Analysis and Future Outlook (2019-2033)
Unlock critical insights into the dynamic United States endoscopy devices market with this in-depth report. Covering endoscopic procedures, gastroenterology endoscopy, pulmonology endoscopy, and urology endoscopy, this report provides a detailed market segmentation by device type and application. Explore key players like Medtronic PLC, Boston Scientific Corporation, and Olympus Corporation, and understand their strategies shaping the market. Examine the impact of technological advancements, regulatory landscapes, and evolving healthcare needs on the US medical devices market. This report is essential for stakeholders seeking to capitalize on the growth of minimally invasive diagnostic and surgical solutions.
United States Endoscopy Devices Market Market Dynamics & Structure
The United States endoscopy devices market is characterized by a moderate to high level of concentration, with major players like Medtronic PLC, Boston Scientific Corporation, and Olympus Corporation holding significant market shares. Technological innovation serves as a primary driver, pushing the development of high-definition visualization, miniaturized instruments, and robotic-assisted systems, enhancing procedural accuracy and patient outcomes. The regulatory framework, primarily overseen by the US Food and Drug Administration (FDA), plays a crucial role in ensuring device safety and efficacy, influencing product development timelines and market entry strategies. Competitive product substitutes are present, particularly in less invasive imaging techniques, yet the direct diagnostic and therapeutic capabilities of endoscopy maintain its dominance in specific applications. End-user demographics are shifting towards an aging population with a higher incidence of chronic diseases, driving demand for diagnostic and therapeutic endoscopic interventions. Mergers and acquisitions (M&A) remain a strategic tool for market consolidation and expansion, with an estimated X deal volumes observed in the historical period.
- Market Concentration: Dominated by a few key global players.
- Technological Innovation: Driven by advancements in imaging, robotics, and instrument design.
- Regulatory Framework: FDA oversight ensures safety and efficacy, impacting market access.
- Competitive Landscape: Facing competition from alternative diagnostic modalities but maintaining a strong niche.
- End-User Demographics: Aging population and rising chronic disease prevalence fuel demand.
- M&A Trends: Strategic acquisitions to enhance product portfolios and market reach.
United States Endoscopy Devices Market Growth Trends & Insights
The United States endoscopy devices market is poised for significant expansion, projected to grow at a Compound Annual Growth Rate (CAGR) of XX% from 2025 to 2033. This growth is underpinned by increasing adoption rates of minimally invasive procedures across various medical specialties, including gastroenterology, pulmonology, and urology. Technological disruptions, such as the advent of AI-powered image analysis and advanced robotic endoscopy, are revolutionizing diagnostic accuracy and therapeutic capabilities. Consumer behavior shifts are also playing a vital role, with patients increasingly seeking less invasive options for diagnosis and treatment, leading to higher procedural volumes. The market size, estimated at approximately $X million units in the base year 2025, is expected to reach $Y million units by 2033. Market penetration is enhanced by growing awareness of early disease detection and preventive healthcare. The transition from traditional endoscopy to more sophisticated systems, including capsule endoscopy and single-use endoscopes, is accelerating the market's evolution, catering to diverse clinical needs and improving infection control. Furthermore, the expanding healthcare infrastructure and increasing reimbursement for endoscopic procedures contribute to sustained market growth. The increasing prevalence of conditions like colorectal cancer, inflammatory bowel disease, and respiratory disorders will continue to be primary demand generators for diagnostic and therapeutic endoscopic interventions.
Dominant Regions, Countries, or Segments in United States Endoscopy Devices Market
Within the United States endoscopy devices market, Gastroenterology emerges as the dominant application segment, consistently driving market growth due to the high prevalence of gastrointestinal disorders and the critical role of endoscopic procedures in their diagnosis and management. The market for gastrointestinal endoscopy devices is projected to account for approximately XX% of the overall market value during the forecast period. This dominance is fueled by factors such as the rising incidence of colorectal cancer, inflammatory bowel disease, and gastroesophageal reflux disease (GERD) across all age groups, especially the aging population. The increasing demand for early detection and minimally invasive treatment options further solidifies gastroenterology's leading position.
- Type of Device Dominance: Flexible Endoscopes, specifically video endoscopes for upper and lower GI examinations, are the most widely adopted.
- Application Dominance: Gastroenterology commands the largest market share due to high procedural volumes and the critical need for diagnosis and treatment of GI conditions.
- Key Drivers in Gastroenterology:
- High prevalence of colorectal cancer and the effectiveness of colonoscopies in screening and early detection.
- Rising incidence of Inflammatory Bowel Disease (IBD) and the need for detailed visualization and biopsy during procedures.
- Increased diagnosis of GERD and related complications requiring endoscopic assessment.
- Advancements in therapeutic endoscopy for managing conditions like bleeding, strictures, and polyps.
- Growth Potential in Other Segments: While gastroenterology leads, significant growth is also anticipated in Pulmonology and Urology, driven by advancements in technology for lung cancer screening and diagnosis, as well as minimally invasive treatments for urological conditions.
United States Endoscopy Devices Market Product Landscape
The United States endoscopy devices market is characterized by a continuous stream of product innovations focused on enhancing diagnostic accuracy, therapeutic efficacy, and patient comfort. Advanced endoscopes, including high-definition flexible and rigid variants, alongside the emerging category of robot-assisted endoscopes, offer superior visualization and maneuverability. Endoscopic operative devices, such as sophisticated irrigation/suction systems and minimally invasive surgical instruments, enable complex interventions. Visualization equipment, including advanced monitors and imaging software, further elevates the procedural experience. Product developments often emphasize miniaturization for less invasive access, improved imaging resolution (e.g., 4K), and integrated AI for real-time analysis, pushing the boundaries of what can be achieved diagnostically and therapeutically through endoscopic routes.
Key Drivers, Barriers & Challenges in United States Endoscopy Devices Market
The United States endoscopy devices market is propelled by several key drivers. Technological advancements, such as improved imaging resolutions and AI integration, are enhancing diagnostic accuracy and enabling more complex therapeutic procedures. The increasing prevalence of chronic diseases, particularly gastrointestinal and respiratory conditions, along with an aging population, fuels the demand for diagnostic and therapeutic endoscopic interventions. Furthermore, the growing preference for minimally invasive procedures over traditional surgery is a significant growth accelerator.
However, the market faces notable barriers and challenges. High initial investment costs for advanced endoscopic equipment can be a restraint for smaller healthcare facilities. Stringent regulatory approval processes, while ensuring safety, can lead to longer product development cycles and increased costs. Supply chain disruptions and the need for skilled professionals to operate sophisticated equipment also pose challenges. Competitive pressures among established players and emerging technologies require continuous innovation and adaptation to maintain market share.
- Key Drivers:
- Technological Innovations (AI, HD Imaging)
- Rising Chronic Disease Prevalence
- Aging Population
- Preference for Minimally Invasive Procedures
- Key Barriers & Challenges:
- High Equipment Costs
- Stringent Regulatory Approvals
- Supply Chain Vulnerabilities
- Need for Skilled Workforce
- Intense Competition
Emerging Opportunities in United States Endoscopy Devices Market
Emerging opportunities in the United States endoscopy devices market lie in the advancement and adoption of AI-powered diagnostic tools, offering real-time analysis of endoscopic images to improve polyp detection rates and lesion characterization. The expansion of therapeutic endoscopy into new applications, such as endoscopic bariatric surgery and advanced interventions for organ failure, presents significant growth avenues. Furthermore, the development of disposable and single-use endoscopes addresses concerns regarding infection control and reprocessing costs, creating a robust market segment, with an estimated XX million units projected for the forecast period. Untapped markets in rural areas and the increasing demand for home-based diagnostic solutions also represent areas for future exploration and innovation.
Growth Accelerators in the United States Endoscopy Devices Market Industry
Long-term growth in the United States endoscopy devices market is significantly accelerated by groundbreaking technological breakthroughs, such as the integration of robotics for enhanced precision and remote surgical capabilities, and the development of advanced imaging techniques like confocal laser endomicroscopy. Strategic partnerships between device manufacturers and healthcare providers are crucial for real-world validation and wider adoption of new technologies. Market expansion strategies, including penetration into underserved patient populations and the development of integrated endoscopy platforms that combine diagnostic and therapeutic functionalities, will further fuel sustained growth and innovation within the industry.
Key Players Shaping the United States Endoscopy Devices Market Market
- Cook Medical
- Medtronic PLC
- Johnson and Johnson
- Richard Wolf GmbH
- Fujifilm Holdings
- Conmed Corporation
- Stryker Corporation
- Boston Scientific Corporation
- Becton Dickinson
- Olympus Corporation
Notable Milestones in United States Endoscopy Devices Market Sector
- May 2023: Olympus Corporation received US-FDA clearance of the EVIS X1 endoscopy system, along with two compatible gastrointestinal endoscopes: the GIF-1100 gastrointestinal videoscope and the CF-HQ1100DL/I colonovideoscope.
- January 2023: UC Davis Health launched a new endoscopy suite in the United States characterized by advanced technology and integrated care. This new suite area is more spacious to accommodate the growing volume of endoscopy procedures.
In-Depth United States Endoscopy Devices Market Market Outlook
The United States endoscopy devices market is set for robust growth driven by advancements in robotic-assisted surgery, AI-integrated imaging, and the increasing demand for minimally invasive diagnostic and therapeutic procedures. Strategic collaborations and the development of novel applications in areas like bariatric and oncological treatments will further enhance market potential. The focus on improving patient outcomes and reducing healthcare costs through advanced endoscopic solutions will continue to be a primary market driver, creating significant opportunities for innovation and market expansion in the coming years.
United States Endoscopy Devices Market Segmentation
-
1. Type of Device
-
1.1. Endoscopes
- 1.1.1. Rigid Endoscope
- 1.1.2. Flexible Endoscope
- 1.1.3. Capsule Endoscope
- 1.1.4. Robot-assisted Endoscope
-
1.2. Endoscopic Operative Devices
- 1.2.1. Irrigation/Suction Systems
- 1.2.2. Access Devices
- 1.2.3. Operative Manual Instruments
- 1.2.4. Other Endoscopic Operative Devices
- 1.3. Visualization Equipment
-
1.1. Endoscopes
-
2. Application
- 2.1. Gastroenterology
- 2.2. Pulmonology
- 2.3. ENT Surgery
- 2.4. Gynecology
- 2.5. Neurology
- 2.6. Urology
- 2.7. Other Applications
United States Endoscopy Devices Market Segmentation By Geography
- 1. United States
United States Endoscopy Devices Market Regional Market Share

Geographic Coverage of United States Endoscopy Devices Market
United States Endoscopy Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.39% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type of Device
- 5.1.1. Endoscopes
- 5.1.1.1. Rigid Endoscope
- 5.1.1.2. Flexible Endoscope
- 5.1.1.3. Capsule Endoscope
- 5.1.1.4. Robot-assisted Endoscope
- 5.1.2. Endoscopic Operative Devices
- 5.1.2.1. Irrigation/Suction Systems
- 5.1.2.2. Access Devices
- 5.1.2.3. Operative Manual Instruments
- 5.1.2.4. Other Endoscopic Operative Devices
- 5.1.3. Visualization Equipment
- 5.1.1. Endoscopes
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Gastroenterology
- 5.2.2. Pulmonology
- 5.2.3. ENT Surgery
- 5.2.4. Gynecology
- 5.2.5. Neurology
- 5.2.6. Urology
- 5.2.7. Other Applications
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. United States
- 5.1. Market Analysis, Insights and Forecast - by Type of Device
- 6. United States Endoscopy Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type of Device
- 6.1.1. Endoscopes
- 6.1.1.1. Rigid Endoscope
- 6.1.1.2. Flexible Endoscope
- 6.1.1.3. Capsule Endoscope
- 6.1.1.4. Robot-assisted Endoscope
- 6.1.2. Endoscopic Operative Devices
- 6.1.2.1. Irrigation/Suction Systems
- 6.1.2.2. Access Devices
- 6.1.2.3. Operative Manual Instruments
- 6.1.2.4. Other Endoscopic Operative Devices
- 6.1.3. Visualization Equipment
- 6.1.1. Endoscopes
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Gastroenterology
- 6.2.2. Pulmonology
- 6.2.3. ENT Surgery
- 6.2.4. Gynecology
- 6.2.5. Neurology
- 6.2.6. Urology
- 6.2.7. Other Applications
- 6.1. Market Analysis, Insights and Forecast - by Type of Device
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Cook Medical
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Medtronic PLC
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Johnson and Johnson
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Richard Wolf GmbH
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Fujifilm Holdings
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Conmed Corporation
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Stryker Corporation
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Boston Scientific Corporation
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Becton Dickinson *List Not Exhaustive
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 Olympus Corporation
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.1 Cook Medical
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: United States Endoscopy Devices Market Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: United States Endoscopy Devices Market Share (%) by Company 2025
List of Tables
- Table 1: United States Endoscopy Devices Market Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 2: United States Endoscopy Devices Market Revenue Million Forecast, by Application 2020 & 2033
- Table 3: United States Endoscopy Devices Market Revenue Million Forecast, by Region 2020 & 2033
- Table 4: United States Endoscopy Devices Market Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 5: United States Endoscopy Devices Market Revenue Million Forecast, by Application 2020 & 2033
- Table 6: United States Endoscopy Devices Market Revenue Million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the United States Endoscopy Devices Market?
The projected CAGR is approximately 6.39%.
2. Which companies are prominent players in the United States Endoscopy Devices Market?
Key companies in the market include Cook Medical, Medtronic PLC, Johnson and Johnson, Richard Wolf GmbH, Fujifilm Holdings, Conmed Corporation, Stryker Corporation, Boston Scientific Corporation, Becton Dickinson *List Not Exhaustive, Olympus Corporation.
3. What are the main segments of the United States Endoscopy Devices Market?
The market segments include Type of Device, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 12.17 Million as of 2022.
5. What are some drivers contributing to market growth?
Rising Preference for Minimally-invasive Surgeries; Increasing Prevalence of Endoscopy for Treatment and Diagnosis; Technological Advancements Leading to Enhanced Applications.
6. What are the notable trends driving market growth?
Flexible Endoscope Segment is Estimated to Register Significant Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
Infections Caused by Few Endoscopes.
8. Can you provide examples of recent developments in the market?
May 2023: Olympus Corporation received the US-FDA clearance of the EVIS X1 endoscopy system, along with two compatible gastrointestinal endoscopes: the GIF-1100 gastrointestinal videoscope and the CF-HQ1100DL/I colonovideoscope.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "United States Endoscopy Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the United States Endoscopy Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the United States Endoscopy Devices Market?
To stay informed about further developments, trends, and reports in the United States Endoscopy Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
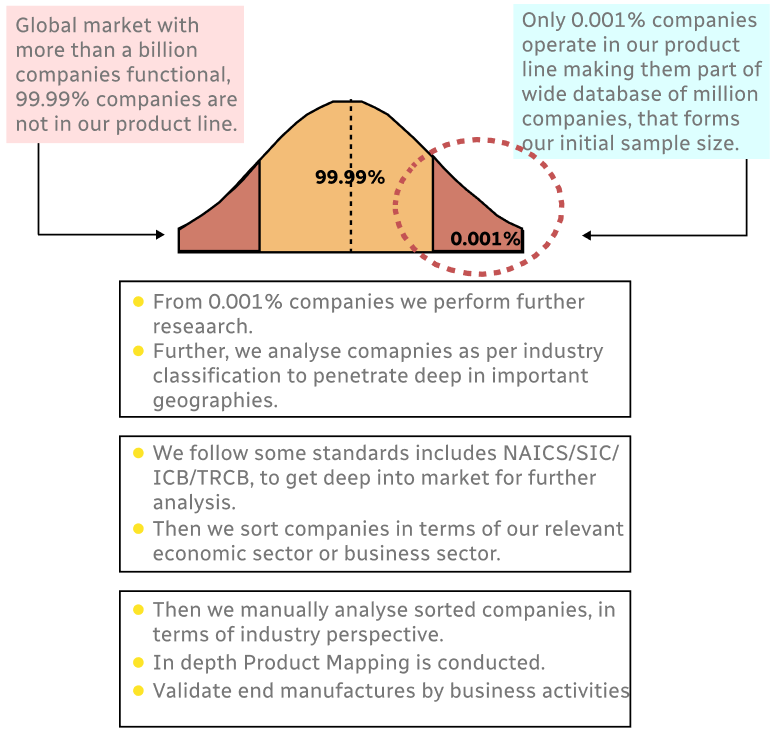
Step 1 - Identification of Relevant Samples Size from Population Database
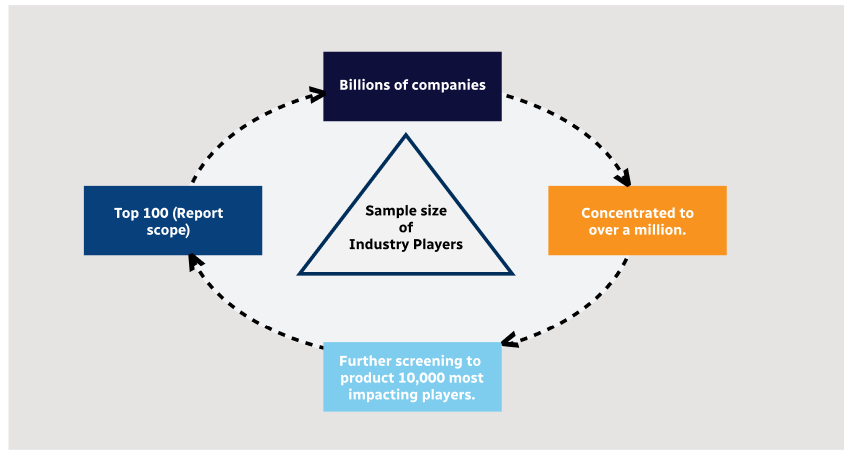
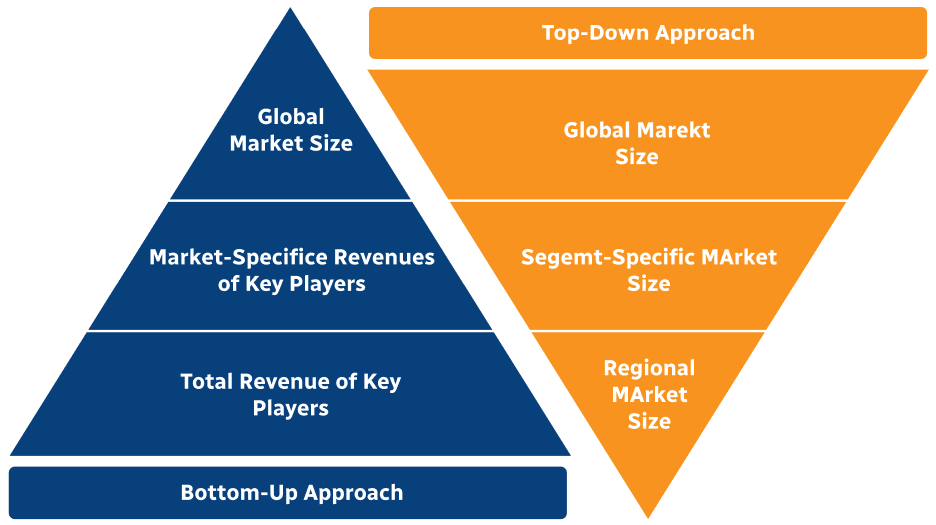
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


