Key Insights
The Thoracic Biomesh market is poised for significant expansion, projected to reach an estimated value of $262 million by 2025, growing at a robust Compound Annual Growth Rate (CAGR) of 7.1% through 2033. This growth is primarily fueled by the increasing prevalence of thoracic conditions such as hernias, pectus deformities, and trauma requiring surgical repair. Advances in biomaterial science, leading to enhanced biocompatibility and mechanical properties of thoracic biomaterials, are also significant drivers. Furthermore, the rising demand for minimally invasive surgical procedures, where biomesh is crucial for reconstruction and support, is contributing to market expansion. The market is segmented by application into public and private hospitals, with both sectors demonstrating consistent demand due to patient volume and the adoption of advanced surgical techniques. By material type, the market encompasses bovine-derived and porcine-derived materials, both of which are integral to surgical repair, with ongoing research aiming to further optimize their efficacy and reduce potential adverse reactions.
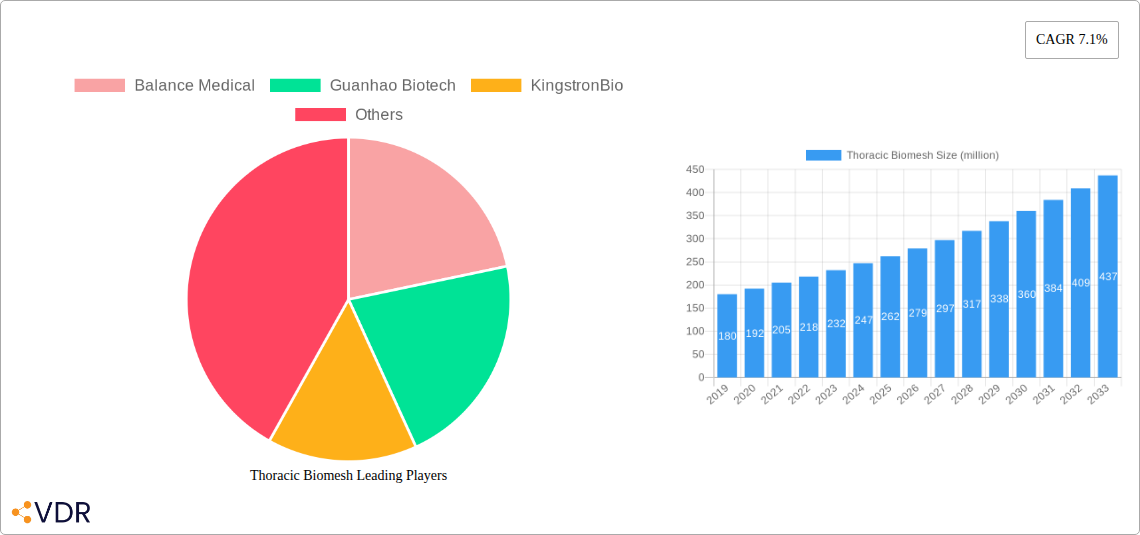
Thoracic Biomesh Market Size (In Million)

The strategic importance of the Thoracic Biomesh market is underscored by the geographical distribution of healthcare infrastructure and the incidence of relevant medical conditions. North America and Europe represent key markets, driven by advanced healthcare systems, high patient awareness, and the early adoption of innovative medical devices. Asia Pacific is anticipated to exhibit the fastest growth, owing to improving healthcare access, increasing medical tourism, and a burgeoning patient population. Restraints to market growth may include the cost of advanced biomaterials and the potential for post-operative complications, though ongoing research and development efforts are actively addressing these challenges. Key players like Balance Medical, Guanhao Biotech, and KingstronBio are actively investing in product innovation and expanding their global presence to capitalize on the expanding market opportunities. The forecast period is expected to witness an intensified competitive landscape as companies strive to offer superior biomaterials and surgical solutions for thoracic reconstruction.
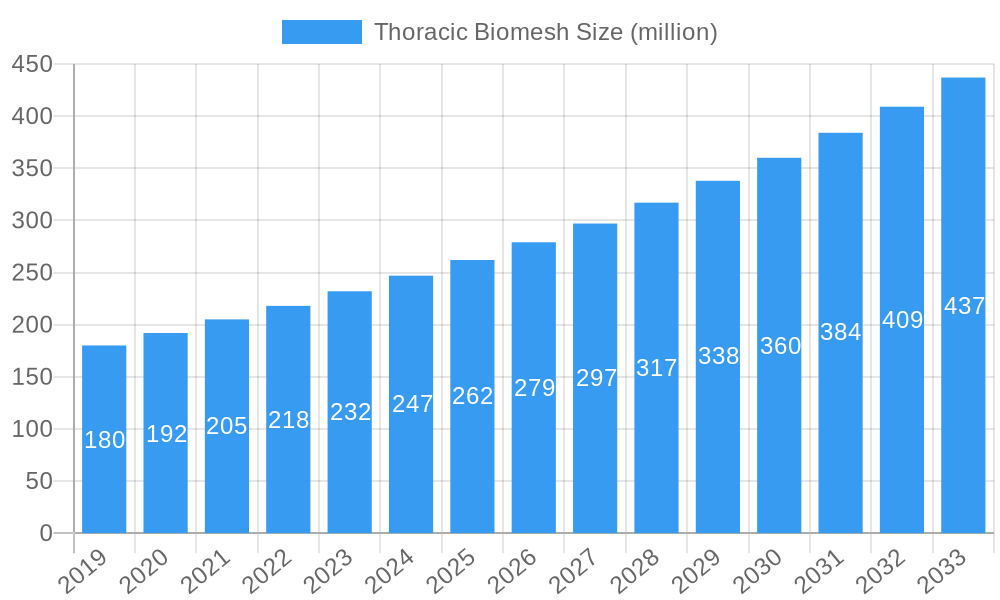
Thoracic Biomesh Company Market Share

Sure, here is a SEO-optimized report description for Thoracic Biomesh, integrating high-traffic keywords and market insights as requested.
Thoracic Biomesh Market Report: 2019-2033 – Growth, Trends, and Opportunities
This comprehensive Thoracic Biomesh market report delivers an in-depth analysis of the global market, covering the historical period (2019-2024), base year (2025), estimated year (2025), and a robust forecast period (2025-2033). We provide unparalleled insights into the thoracic defect repair, chest wall reconstruction, and sternal closure markets, driven by increasing demand for advanced medical implants and biomaterials. This report is essential for stakeholders seeking to understand thoracic biomesh applications, bovine derived materials, porcine derived materials, and the competitive landscape featuring key players like Balance Medical, Guanhao Biotech, and KingstronBio.
Thoracic Biomesh Market Dynamics & Structure
The thoracic biomesh market exhibits moderate concentration, with a few key players dominating innovation and production. Technological advancements in biomaterial science are a primary driver, pushing for enhanced biocompatibility and efficacy in thoracic surgery. Regulatory frameworks, particularly those governing medical device approvals and biocompatibility testing, play a crucial role in market access and product development. Competitive product substitutes, such as traditional sutures and advanced synthetic meshes, pose a challenge, necessitating continuous innovation in biomesh technology. End-user demographics reveal a growing preference for minimally invasive surgical techniques, increasing the demand for specialized thoracic biomesh solutions. Merger and acquisition (M&A) trends are expected to accelerate, driven by companies seeking to expand their product portfolios and market reach within the $120 million thoracic biomesh market. Key industry developments include advancements in 3D printing of medical implants and the exploration of novel regenerative biomaterials.
- Market Concentration: Moderate, with emphasis on innovation and quality.
- Technological Innovation Drivers: Advancements in biomaterial science, tissue engineering, and minimally invasive surgical tools.
- Regulatory Frameworks: Strict FDA and EMA guidelines for medical device approval and biocompatibility.
- Competitive Product Substitutes: Traditional sutures, synthetic meshes, and bioabsorbable materials.
- End-User Demographics: Aging populations with higher incidences of cardiovascular and thoracic diseases, increasing demand for advanced repair solutions.
- M&A Trends: Increasing consolidation to gain market share and access to new technologies, with an estimated 8 M&A deals in the historical period valued at over $45 million.
Thoracic Biomesh Growth Trends & Insights
The thoracic biomesh market is projected for significant growth, with an estimated market size of $135 million in 2025, poised to reach approximately $250 million by 2033, reflecting a Compound Annual Growth Rate (CAGR) of 6.5% during the forecast period. This expansion is fueled by a confluence of factors including the increasing prevalence of thoracic trauma, congenital chest wall deformities, and a rising incidence of lung cancer treatment requiring chest wall reconstruction. The adoption rate of advanced thoracic biomesh solutions is steadily increasing as surgeons recognize their superior performance in facilitating tissue integration and reducing complications compared to traditional methods. Technological disruptions, such as the development of advanced biodegradable thoracic meshes and patient-specific implant designs, are further accelerating market penetration. Consumer behavior shifts indicate a growing demand for enhanced patient outcomes and faster recovery times, directly benefiting the adoption of sophisticated biomesh products. The market penetration of advanced biomaterials in thoracic surgeries is estimated to increase from 30% in the historical period to over 45% by 2033.
- Market Size Evolution: From an estimated $120 million in 2024 to $250 million by 2033.
- Adoption Rates: Increasing adoption driven by improved patient outcomes and surgeon preference for advanced biomaterials.
- Technological Disruptions: Innovations in biodegradable materials, antimicrobial coatings, and bio-integrated meshes.
- Consumer Behavior Shifts: Growing patient awareness and demand for less invasive procedures and faster recovery.
- Market Penetration: Projected to rise significantly as awareness and clinical evidence for advanced biomaterials grow.
- CAGR: Estimated at 6.5% from 2025 to 2033.
Dominant Regions, Countries, or Segments in Thoracic Biomesh
The North America region currently dominates the thoracic biomesh market, driven by a robust healthcare infrastructure, high patient expenditure on advanced medical treatments, and a strong presence of leading medical device manufacturers. The United States, specifically, is a major contributor to this dominance, with a high prevalence of thoracic reconstructive surgery procedures. In terms of Application, Public Hospitals are anticipated to hold a larger market share due to higher patient volumes and government-backed healthcare initiatives, although Private Hospitals are increasingly adopting advanced biomesh solutions to offer premium patient care. Analyzing Type, Bovine Derived Materials currently lead the market owing to their established efficacy and biocompatibility in sternal wound closure and chest wall defect repair. However, Porcine Derived Materials are gaining traction, driven by ongoing research into enhanced processing techniques and potential cost advantages.
- Dominant Region: North America, led by the United States.
- Key Drivers: Advanced healthcare infrastructure, high R&D investment, favorable reimbursement policies, and a high incidence of thoracic pathologies.
- Market Share: Estimated to account for over 35% of the global market in 2025.
- Dominant Application Segment: Public Hospitals.
- Key Drivers: Higher patient volumes, increasing government funding for medical treatments, and a greater focus on public health initiatives.
- Growth Potential: Strong, driven by accessibility and broad patient reach.
- Dominant Type Segment: Bovine Derived Materials.
- Key Drivers: Proven clinical history, excellent biocompatibility, and established manufacturing processes for thoracic defect repair.
- Market Share: Projected to capture approximately 60% of the market in 2025.
- Emerging Type Segment: Porcine Derived Materials.
- Growth Potential: Significant, driven by ongoing research, potential for cost optimization, and development of enhanced material properties.
Thoracic Biomesh Product Landscape
The thoracic biomesh product landscape is characterized by continuous innovation focused on enhancing patient outcomes and simplifying surgical procedures. Key product advancements include the development of biologically active meshes that promote tissue regeneration, as well as patient-specific 3D-printed biomesh implants tailored to individual anatomical needs for chest wall reconstruction. These innovations offer superior integration with surrounding tissues, reduced risk of infection, and improved mechanical strength for complex thoracic repairs. Unique selling propositions often lie in the biomaterial source (e.g., highly purified bovine pericardium or porcine submucosa), inherent biomechanical properties, and the incorporation of antimicrobial agents or growth factors. Technological advancements are also focusing on creating meshes with varying pore sizes and degradation rates to optimize healing for diverse surgical applications in thoracic surgery.
Key Drivers, Barriers & Challenges in Thoracic Biomesh
Several key drivers are propelling the thoracic biomesh market forward. The increasing incidence of thoracic trauma, congenital chest wall deformities, and the rising demand for minimally invasive surgery are significant catalysts. Technological advancements in biomaterial science and tissue engineering are enabling the development of more effective and biocompatible meshes for chest wall reconstruction. Furthermore, supportive government initiatives and increasing healthcare expenditure globally are creating a favorable environment for market growth.
Conversely, the market faces several barriers and challenges. High research and development costs associated with novel biomaterial creation and clinical trials can be prohibitive. Stringent regulatory approval processes for medical devices can lead to delays in market entry. The availability of cost-effective traditional surgical techniques also presents a competitive challenge, particularly in price-sensitive markets. Supply chain disruptions and the need for specialized surgical expertise can also impede wider adoption of advanced thoracic biomesh solutions.
Emerging Opportunities in Thoracic Biomesh
Emerging opportunities in the thoracic biomesh market lie in the development of regenerative medicine-based biomaterials that actively promote tissue regrowth and integration. The expansion of thoracic surgery procedures in emerging economies, coupled with increasing healthcare investment, presents significant untapped market potential. Moreover, the exploration of smart biomaterials with embedded sensors for real-time monitoring of healing processes and the customization of biomesh designs through advanced 3D printing technology for complex thoracic reconstruction scenarios are promising avenues for innovation and market growth.
Growth Accelerators in the Thoracic Biomesh Industry
Growth accelerators in the thoracic biomesh industry are primarily driven by ongoing technological breakthroughs in biomaterial science, leading to the creation of meshes with enhanced biocompatibility and regenerative capabilities. Strategic partnerships between medical device manufacturers and research institutions are fostering innovation and accelerating the translation of new technologies into clinical practice. Market expansion strategies, including the penetration of developing economies and the introduction of novel product applications for pulmonary surgery and cardiac support, are also vital growth catalysts. The increasing focus on patient-centric care and the demand for improved surgical outcomes further amplify the adoption of advanced thoracic biomesh solutions.
Key Players Shaping the Thoracic Biomesh Market
- Balance Medical
- Guanhao Biotech
- KingstronBio
Notable Milestones in Thoracic Biomesh Sector
- 2021/08: Balance Medical launched its new generation of bovine-derived thoracic biomesh with enhanced tensile strength.
- 2022/03: Guanhao Biotech secured significant funding to expand its research into bioabsorbable porcine-derived materials for thoracic applications.
- 2023/01: KingstronBio announced a strategic partnership aimed at developing advanced 3D-printed thoracic implants.
- 2023/09: Regulatory approval granted for a novel antimicrobial-coated thoracic biomesh in key European markets.
- 2024/05: A major clinical study published highlighting superior outcomes with bovine-derived biomesh in complex sternal closures.
In-Depth Thoracic Biomesh Market Outlook
The thoracic biomesh market is poised for sustained growth, driven by an aging global population, increasing prevalence of thoracic pathologies, and continuous technological advancements. Future market potential will be significantly shaped by the development of next-generation biomaterials, including bioresorbable scaffolds and bio-integrated implants that actively promote tissue regeneration. Strategic opportunities for market players include expanding product portfolios to address a wider range of thoracic surgical procedures, investing in research and development for innovative biomaterial applications, and focusing on market penetration in emerging economies. The increasing emphasis on patient outcomes and minimally invasive techniques will continue to fuel the demand for advanced thoracic biomesh solutions, solidifying their position as essential tools in modern thoracic surgery.
Thoracic Biomesh Segmentation
-
1. Application
- 1.1. Public Hospital
- 1.2. Private Hospital
-
2. Type
- 2.1. Bovine Derived Materials
- 2.2. Porcine Derived Materials
Thoracic Biomesh Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
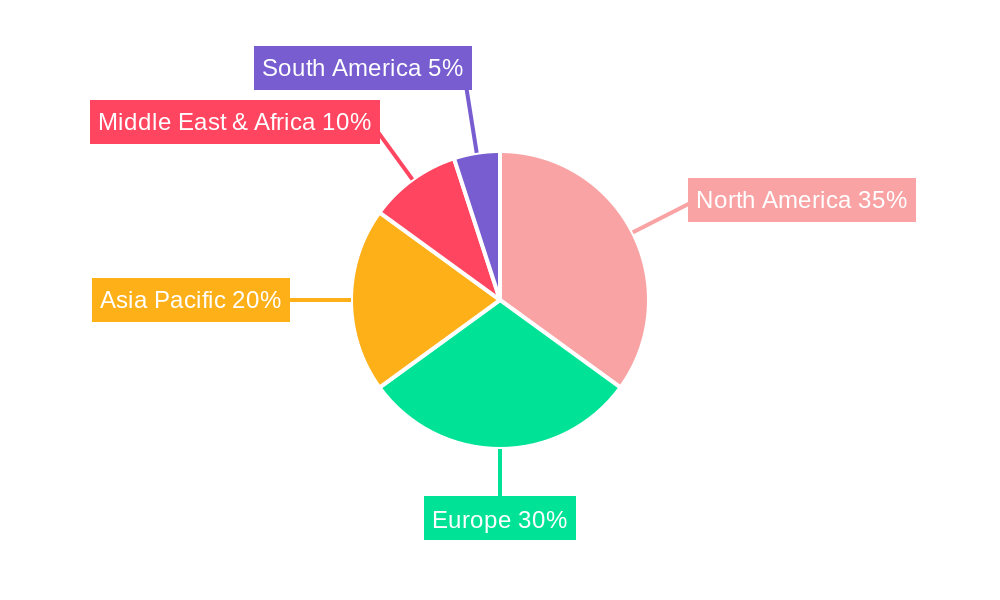
Thoracic Biomesh Regional Market Share

Geographic Coverage of Thoracic Biomesh
Thoracic Biomesh REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Thoracic Biomesh Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Public Hospital
- 5.1.2. Private Hospital
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Bovine Derived Materials
- 5.2.2. Porcine Derived Materials
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Thoracic Biomesh Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Public Hospital
- 6.1.2. Private Hospital
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Bovine Derived Materials
- 6.2.2. Porcine Derived Materials
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Thoracic Biomesh Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Public Hospital
- 7.1.2. Private Hospital
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Bovine Derived Materials
- 7.2.2. Porcine Derived Materials
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Thoracic Biomesh Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Public Hospital
- 8.1.2. Private Hospital
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Bovine Derived Materials
- 8.2.2. Porcine Derived Materials
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Thoracic Biomesh Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Public Hospital
- 9.1.2. Private Hospital
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Bovine Derived Materials
- 9.2.2. Porcine Derived Materials
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Thoracic Biomesh Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Public Hospital
- 10.1.2. Private Hospital
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Bovine Derived Materials
- 10.2.2. Porcine Derived Materials
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Balance Medical
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Guanhao Biotech
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 KingstronBio
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.1 Balance Medical
List of Figures
- Figure 1: Global Thoracic Biomesh Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Thoracic Biomesh Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Thoracic Biomesh Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Thoracic Biomesh Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Thoracic Biomesh Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Thoracic Biomesh Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Thoracic Biomesh Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Thoracic Biomesh Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Thoracic Biomesh Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Thoracic Biomesh Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Thoracic Biomesh Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Thoracic Biomesh Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Thoracic Biomesh Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Thoracic Biomesh Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Thoracic Biomesh Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Thoracic Biomesh Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Thoracic Biomesh Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Thoracic Biomesh Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Thoracic Biomesh Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Thoracic Biomesh Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Thoracic Biomesh Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Thoracic Biomesh Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Thoracic Biomesh Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Thoracic Biomesh Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Thoracic Biomesh Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Thoracic Biomesh Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Thoracic Biomesh Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Thoracic Biomesh Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Thoracic Biomesh Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Thoracic Biomesh Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Thoracic Biomesh Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Thoracic Biomesh Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Thoracic Biomesh Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Thoracic Biomesh Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Thoracic Biomesh Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Thoracic Biomesh Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Thoracic Biomesh Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Thoracic Biomesh Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Thoracic Biomesh Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Thoracic Biomesh Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Thoracic Biomesh Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Thoracic Biomesh Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Thoracic Biomesh Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Thoracic Biomesh Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Thoracic Biomesh Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Thoracic Biomesh Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Thoracic Biomesh Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Thoracic Biomesh Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Thoracic Biomesh Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Thoracic Biomesh Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Thoracic Biomesh?
The projected CAGR is approximately 7.3%.
2. Which companies are prominent players in the Thoracic Biomesh?
Key companies in the market include Balance Medical, Guanhao Biotech, KingstronBio.
3. What are the main segments of the Thoracic Biomesh?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Thoracic Biomesh," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Thoracic Biomesh report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Thoracic Biomesh?
To stay informed about further developments, trends, and reports in the Thoracic Biomesh, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
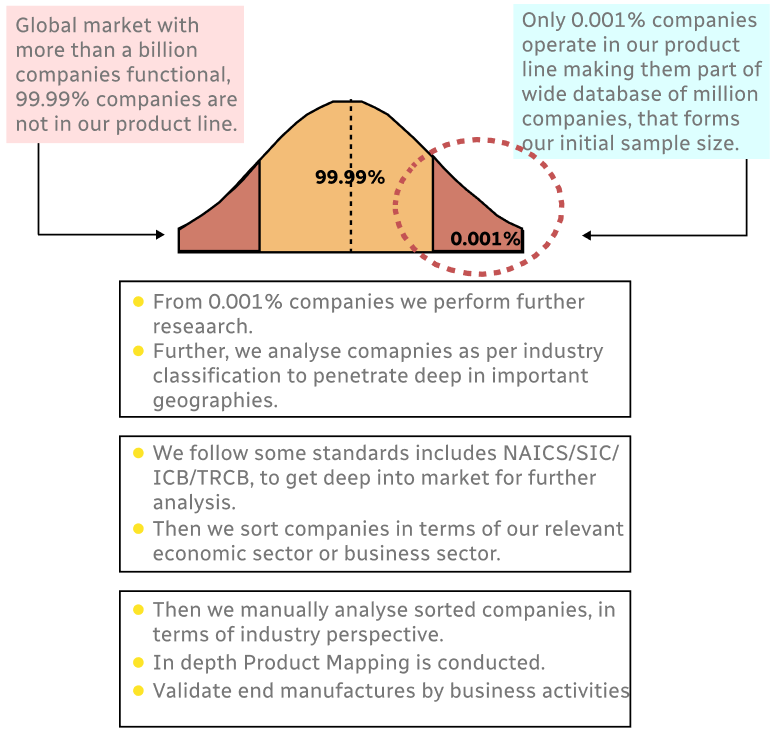
Step 1 - Identification of Relevant Samples Size from Population Database
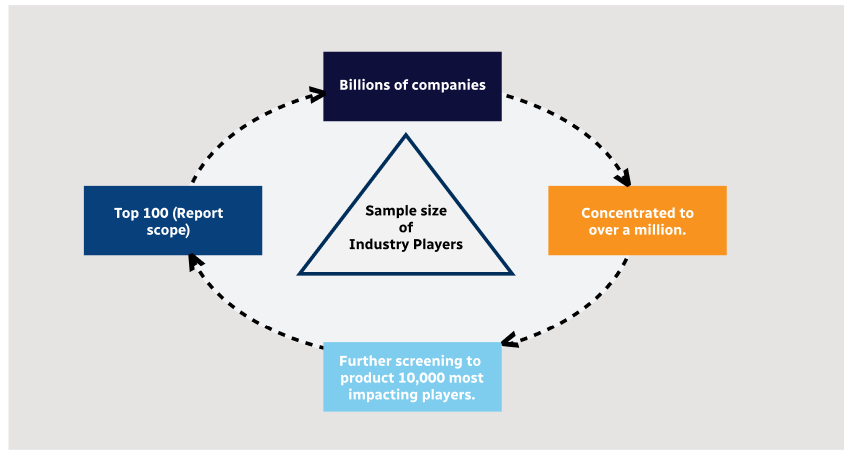
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
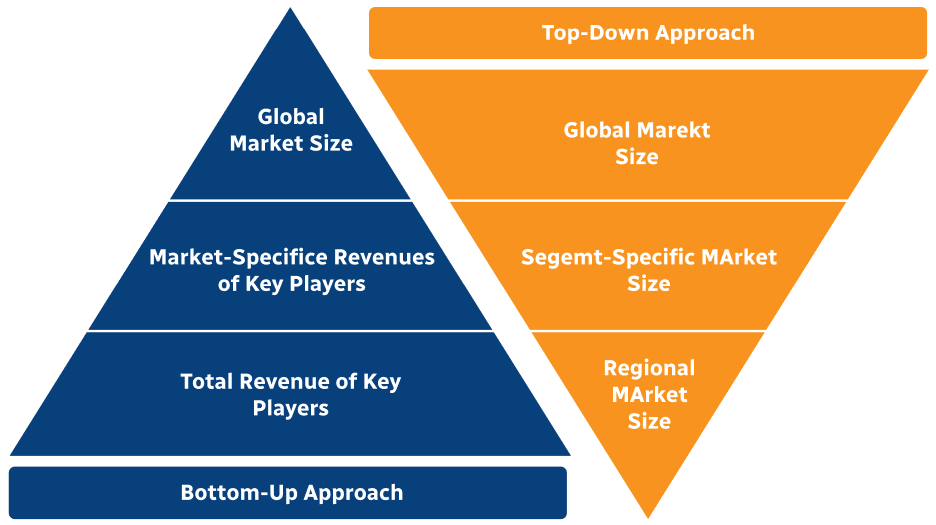
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
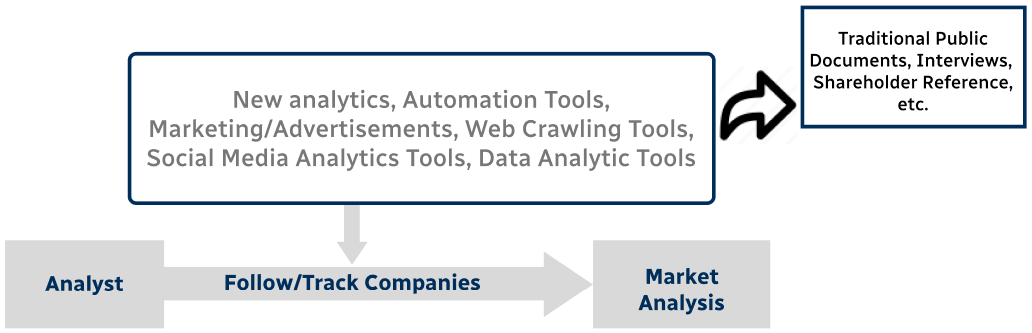
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


