Key Insights
The Biopharmaceutical Testing Services market is poised for significant expansion, projected to reach an estimated $7.48 billion in 2025. This robust growth is driven by a confluence of factors, including the escalating demand for novel biopharmaceutical drugs, stringent regulatory requirements for product quality and safety, and the increasing complexity of drug development processes. The market's CAGR of 10.8% underscores a dynamic landscape characterized by continuous innovation and a strong emphasis on ensuring the efficacy and safety of biopharmaceutical products. Key applications within this market encompass a wide spectrum, from essential raw materials and in-process testing to the critical analysis of finished pharmaceutical products, environmental sample monitoring, and other specialized testing needs. Leading players such as Eurofins Scientific, SGS, Envigo, and PPD Inc. are instrumental in shaping this market through their comprehensive service offerings and advancements in analytical technologies.

Biopharmaceutical Testing Services Market Size (In Billion)

The trajectory of the biopharmaceutical testing services market is further shaped by emerging trends such as the rise of personalized medicine, which necessitates more sophisticated and tailored testing protocols. Advancements in analytical techniques, including high-throughput screening, omics technologies, and advanced cell-based assays, are enhancing the precision and efficiency of testing. Conversely, challenges such as the high cost of advanced testing equipment and the need for specialized expertise can act as moderating forces. Geographically, North America and Europe currently lead the market due to established biopharmaceutical hubs and robust regulatory frameworks, but the Asia Pacific region is exhibiting rapid growth, fueled by increasing investments in R&D and a burgeoning biopharmaceutical manufacturing base. The forecast period from 2025 to 2033 indicates sustained expansion as the global healthcare industry continues to prioritize the development and reliable testing of life-saving biopharmaceutical interventions.

Biopharmaceutical Testing Services Company Market Share

This comprehensive report provides an in-depth analysis of the global Biopharmaceutical Testing Services market, offering critical insights into market dynamics, growth trends, regional dominance, product landscape, and key players. Spanning the historical period of 2019-2024, the base year of 2025, and extending to a forecast period of 2025-2033, this report is an indispensable resource for stakeholders seeking to understand and capitalize on the evolving biopharmaceutical testing sector. The market is segmented by Application (Hospitals, Clinics, Others) and Type (Raw Materials Testing, In-Process and Product Release Testing, Finished Pharmaceutical Products Testing, Environmental Samples, Others), providing a granular view of demand drivers and innovation frontiers.
Biopharmaceutical Testing Services Market Dynamics & Structure
The biopharmaceutical testing services market exhibits a moderately concentrated structure, with a few key players holding significant market share. Technological innovation serves as a primary driver, propelled by advancements in analytical techniques, genomics, proteomics, and data analytics, enabling more precise and efficient drug development and quality control. Stringent regulatory frameworks, such as those established by the FDA and EMA, mandate rigorous testing at every stage of biopharmaceutical development, fostering a continuous demand for specialized testing services. Competitive product substitutes are limited within core biopharmaceutical testing due to the highly specialized and regulated nature of the field, though advancements in in-house testing capabilities by large pharmaceutical companies can represent indirect competition. End-user demographics are primarily driven by pharmaceutical and biotechnology companies, contract research organizations (CROs), and academic institutions. Mergers and acquisitions (M&A) are a notable trend, consolidating market power and expanding service portfolios. For instance, the historical period saw several strategic acquisitions aimed at broadening geographic reach and specialized service offerings.
- Market Concentration: Moderately concentrated with key players like Eurofins Scientific and SGS dominating.
- Technological Innovation: Driven by advancements in omics technologies, high-throughput screening, and AI-powered data analysis.
- Regulatory Frameworks: Driven by strict compliance requirements from bodies like the FDA, EMA, and PMDA.
- Competitive Landscape: Characterized by specialized service providers and a growing trend of strategic partnerships.
- End-User Base: Primarily pharmaceutical and biotechnology companies, CROs, and academic research institutions.
- M&A Activity: Active consolidation to enhance capabilities, expand service offerings, and gain market share.
Biopharmaceutical Testing Services Growth Trends & Insights
The global Biopharmaceutical Testing Services market is poised for substantial growth, projected to expand significantly over the forecast period. This growth is underpinned by several interconnected trends, including an accelerating pipeline of novel biologics, increasing complexity in drug formulations, and a heightened emphasis on product safety and efficacy. The escalating investment in biopharmaceutical research and development (R&D) by both established and emerging companies directly translates into a higher demand for comprehensive testing solutions. Furthermore, the growing prevalence of chronic diseases and an aging global population are fueling the demand for innovative biopharmaceutical treatments, thereby expanding the need for robust testing services throughout the drug lifecycle.
Technological disruptions are profoundly influencing the market. The adoption of advanced analytical instrumentation, such as mass spectrometry and next-generation sequencing, allows for more accurate characterization of complex biomolecules and the detection of trace impurities. The integration of artificial intelligence (AI) and machine learning (ML) in data analysis and predictive modeling is revolutionizing the efficiency and effectiveness of biopharmaceutical testing. These technologies are not only accelerating the identification of potential drug candidates but also optimizing quality control processes, leading to faster market entry for life-saving therapies.
Consumer behavior shifts, particularly the increasing patient and regulatory demand for transparency and accountability in drug manufacturing, are also significant growth catalysts. This necessitates meticulous testing and validation at every stage, from raw material sourcing to finished product release. The expansion of contract testing services, driven by the need for specialized expertise and cost-effectiveness, is another key trend. Many pharmaceutical companies are increasingly outsourcing their testing needs to specialized service providers to streamline operations and access cutting-edge technologies. The market penetration of these services is expected to deepen as the benefits of outsourcing become more widely recognized. The estimated market size for biopharmaceutical testing services in the base year of 2025 is projected to be in the range of $XX billion, with a projected Compound Annual Growth Rate (CAGR) of approximately XX% during the forecast period. This robust growth trajectory is indicative of the critical and expanding role of these services in bringing safe and effective biopharmaceutical products to market.
Dominant Regions, Countries, or Segments in Biopharmaceutical Testing Services
The North America region consistently emerges as a dominant force in the global Biopharmaceutical Testing Services market, driven by a confluence of factors that foster innovation, investment, and regulatory oversight. The United States, in particular, boasts a highly developed biopharmaceutical industry with a significant concentration of leading pharmaceutical and biotechnology companies, a robust venture capital ecosystem supporting startups, and a well-established network of Contract Research Organizations (CROs). This robust ecosystem creates an incessant demand for a wide array of testing services.
Within the Application segment, Hospitals play a crucial, albeit indirectly, role by being major consumers of finished biopharmaceutical products. Their demand drives the production volumes, which in turn necessitates extensive testing services. However, the Others application segment, encompassing Contract Research Organizations (CROs) and specialized biotechnology firms, is a more direct and significant driver of testing service demand. These entities are at the forefront of drug discovery and development, requiring comprehensive testing at every stage.
From the Type perspective, In-Process and Product Release Testing stands out as a segment with substantial and consistent growth. This is due to the critical need for quality assurance during the manufacturing of biologics, ensuring consistency, purity, and potency. Finished Pharmaceutical Products Testing also holds a dominant position, reflecting the regulatory mandates for final product verification before market release. The increasing complexity of biopharmaceutical products, such as monoclonal antibodies, vaccines, and gene therapies, further accentuates the need for specialized and advanced testing solutions in this category.
Key drivers for North America's dominance include favorable economic policies that encourage R&D investment, well-developed healthcare infrastructure, and a highly educated workforce with expertise in life sciences. The region’s proactive regulatory agencies, like the FDA, set high standards for drug safety and efficacy, compelling companies to invest heavily in rigorous testing. The market share held by North America is estimated to be around XX% of the global market in the base year of 2025, with a projected growth potential that is expected to outpace many other regions. The strong presence of academic research institutions also contributes to a continuous flow of innovation and demand for specialized testing services for novel research projects.
- Dominant Region: North America, primarily driven by the United States.
- Key Application Segment: The "Others" segment (CROs, Biotech Firms) is a direct demand driver, while Hospitals indirectly influence through end-product consumption.
- Dominant Type Segment: "In-Process and Product Release Testing" and "Finished Pharmaceutical Products Testing" are paramount due to regulatory and quality control imperatives.
- Growth Drivers: Robust R&D investment, advanced healthcare infrastructure, stringent regulatory oversight, and a strong presence of pharmaceutical and biotech companies.
- Market Share (2025 Estimate): North America estimated to hold approximately XX% of the global market.
Biopharmaceutical Testing Services Product Landscape
The product landscape of biopharmaceutical testing services is characterized by sophisticated, multi-faceted offerings designed to address the complex needs of drug development and manufacturing. Innovations are centered around enhancing sensitivity, specificity, and throughput. This includes the development of advanced analytical platforms for protein characterization, impurity profiling, and potency assays. Emerging applications are focusing on cell and gene therapy testing, requiring specialized assays for viral vector integrity, vector potency, and off-target effects. Performance metrics are continuously being improved, with faster turnaround times, lower detection limits for impurities, and greater automation becoming standard expectations. Unique selling propositions often lie in specialized expertise in niche therapeutic areas or complex molecule testing.
Key Drivers, Barriers & Challenges in Biopharmaceutical Testing Services
Key Drivers: The biopharmaceutical testing services market is propelled by the relentless pursuit of novel drug discovery and development, fueled by significant R&D investments. Stringent regulatory requirements worldwide mandate comprehensive testing for safety and efficacy, creating a consistent demand. The increasing complexity of biologics, including biosimilars and advanced therapies, necessitates specialized analytical capabilities. Furthermore, the outsourcing trend, driven by cost efficiencies and access to specialized expertise, is a major growth accelerator.
Barriers & Challenges: High capital investment in advanced instrumentation and skilled personnel acts as a significant barrier to entry. The evolving and often complex regulatory landscape can pose challenges, requiring continuous adaptation and expertise. Supply chain disruptions for critical reagents and consumables can impact service delivery timelines and costs. Intense competition among established players and emerging niche providers can lead to pricing pressures. Lastly, the need for specialized scientific expertise and the challenge of retaining top talent are ongoing hurdles.
Emerging Opportunities in Biopharmaceutical Testing Services
Emerging opportunities lie in the burgeoning fields of cell and gene therapies, which require highly specialized and novel testing methodologies for viral vectors, gene editing efficacy, and immunogenicity. The growing market for biosimilars presents a significant opportunity, demanding extensive comparability testing. Expansion into emerging economies with expanding biopharmaceutical sectors also offers untapped potential. Furthermore, the integration of AI and machine learning in data interpretation and predictive analytics for testing offers avenues for enhanced efficiency and novel service development. The increasing focus on personalized medicine will also drive demand for companion diagnostic testing and associated services.
Growth Accelerators in the Biopharmaceutical Testing Services Industry
Growth accelerators in the biopharmaceutical testing services industry are multifaceted. Technological breakthroughs in areas like CRISPR gene editing, single-cell analysis, and advanced mass spectrometry are opening new frontiers for testing. Strategic partnerships and collaborations between testing service providers and pharmaceutical companies, as well as between CROs and academic institutions, foster innovation and market penetration. Market expansion strategies, including geographical diversification into Asia-Pacific and other developing regions, are crucial for long-term growth. The increasing adoption of automation and digital solutions within testing laboratories further enhances efficiency and scalability, acting as significant growth catalysts.
Key Players Shaping the Biopharmaceutical Testing Services Market
- Eurofins Scientific
- SGS
- Envigo
- Exova Group
- PPD Inc.
- Pace Analytical Services
- Intertek Group
- DYNALABS
- RD Laboratories
- West Pharmaceutical Services
- Polymer Solutions
- Boston Analytical
- Accuratus Labs
- Microbac
- ARLBioPharma
- Lapuck Laboratories
- BioScreen
Notable Milestones in Biopharmaceutical Testing Services Sector
- 2019: Significant M&A activities, with larger CROs acquiring specialized testing firms to expand service portfolios.
- 2020: Increased demand for viral testing services due to the COVID-19 pandemic, accelerating investments in relevant technologies and capacity.
- 2021: Advancements in AI and machine learning integration for biopharmaceutical data analysis and quality control become more prevalent.
- 2022: Growing focus on environmental monitoring and sustainability testing within biopharmaceutical manufacturing.
- 2023: Increased regulatory scrutiny on impurities, particularly nitrosamines, driving demand for advanced analytical testing.
- 2024: Emergence of dedicated testing solutions for mRNA-based therapeutics and advanced cell & gene therapies.
In-Depth Biopharmaceutical Testing Services Market Outlook
The future outlook for the biopharmaceutical testing services market is exceptionally promising, driven by sustained innovation and the increasing complexity of therapeutic modalities. Growth accelerators such as the expansion of cell and gene therapy pipelines, the continuous demand for biosimilar development, and the globalization of biopharmaceutical manufacturing will ensure robust market expansion. Strategic opportunities lie in further developing specialized testing capabilities for novel modalities, enhancing digital integration for faster data turnaround, and expanding service offerings in underserved emerging markets. The commitment to quality, safety, and efficacy in biopharmaceutical development will continue to solidify the indispensable role of comprehensive testing services, leading to significant market potential and continued growth.
Biopharmaceutical Testing Services Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Others
-
2. Type
- 2.1. Raw Materials Testing
- 2.2. In-Process and Product Release Testing
- 2.3. Finished Pharmaceutical Products Testing
- 2.4. Environmental Samples
- 2.5. Others
Biopharmaceutical Testing Services Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
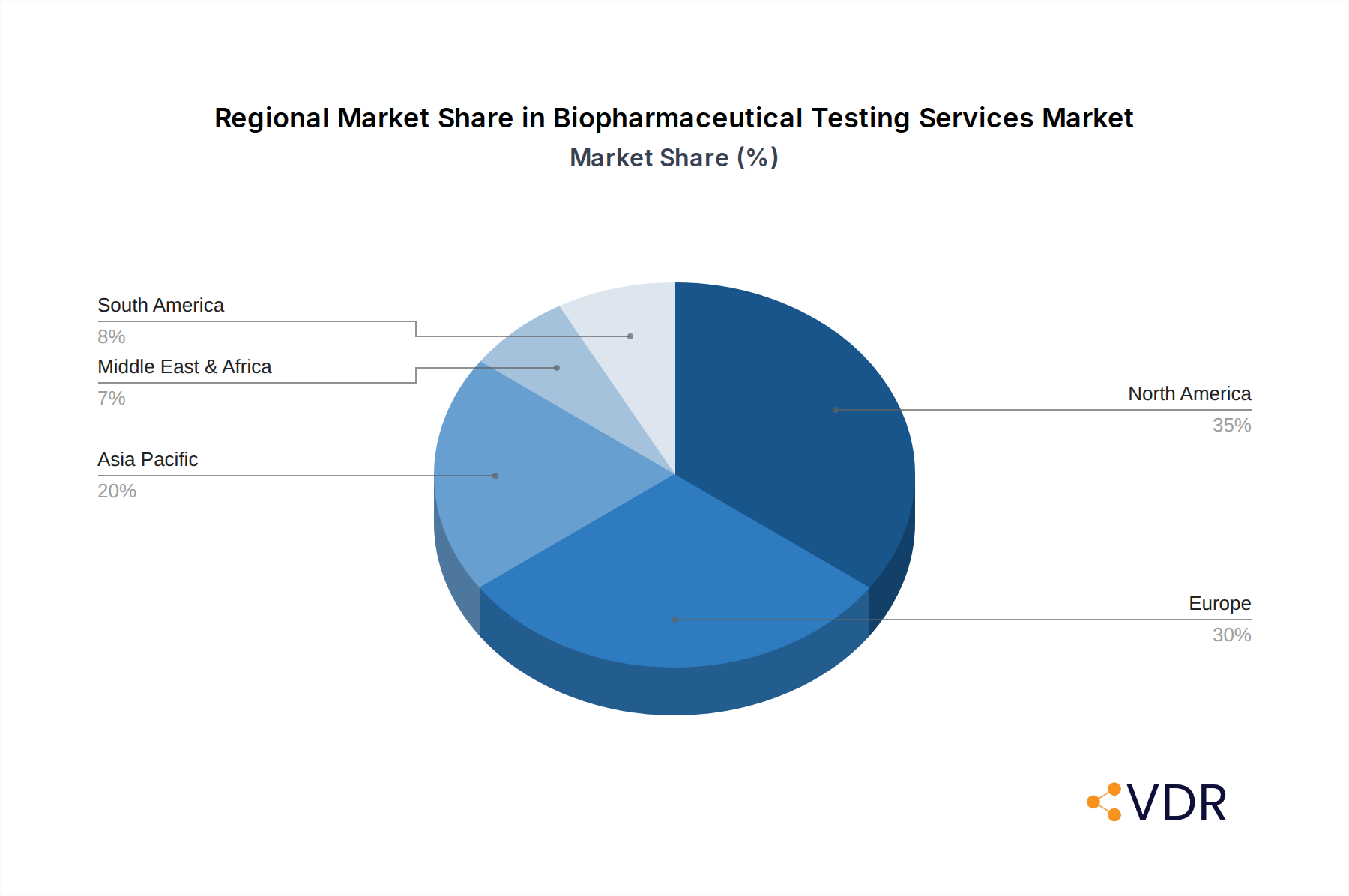
Biopharmaceutical Testing Services Regional Market Share

Geographic Coverage of Biopharmaceutical Testing Services
Biopharmaceutical Testing Services REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Biopharmaceutical Testing Services Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Raw Materials Testing
- 5.2.2. In-Process and Product Release Testing
- 5.2.3. Finished Pharmaceutical Products Testing
- 5.2.4. Environmental Samples
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Biopharmaceutical Testing Services Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Raw Materials Testing
- 6.2.2. In-Process and Product Release Testing
- 6.2.3. Finished Pharmaceutical Products Testing
- 6.2.4. Environmental Samples
- 6.2.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Biopharmaceutical Testing Services Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Raw Materials Testing
- 7.2.2. In-Process and Product Release Testing
- 7.2.3. Finished Pharmaceutical Products Testing
- 7.2.4. Environmental Samples
- 7.2.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Biopharmaceutical Testing Services Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Raw Materials Testing
- 8.2.2. In-Process and Product Release Testing
- 8.2.3. Finished Pharmaceutical Products Testing
- 8.2.4. Environmental Samples
- 8.2.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Biopharmaceutical Testing Services Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Raw Materials Testing
- 9.2.2. In-Process and Product Release Testing
- 9.2.3. Finished Pharmaceutical Products Testing
- 9.2.4. Environmental Samples
- 9.2.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Biopharmaceutical Testing Services Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Raw Materials Testing
- 10.2.2. In-Process and Product Release Testing
- 10.2.3. Finished Pharmaceutical Products Testing
- 10.2.4. Environmental Samples
- 10.2.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Eurofins Scientific
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 SGS
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Envigo
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Exova Group
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 PPD Inc.
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Pace Analytical Services
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Intertek Group
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 DYNALABS
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 RD Laboratories
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 West Pharmaceutical Services
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Polymer Solutions
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Boston Analytical
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Accuratus Labs
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Microbac
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 ARLBioPharma
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Lapuck Laboratories
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 BioScreen
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.1 Eurofins Scientific
List of Figures
- Figure 1: Global Biopharmaceutical Testing Services Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Biopharmaceutical Testing Services Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Biopharmaceutical Testing Services Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Biopharmaceutical Testing Services Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Biopharmaceutical Testing Services Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Biopharmaceutical Testing Services Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Biopharmaceutical Testing Services Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Biopharmaceutical Testing Services Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Biopharmaceutical Testing Services Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Biopharmaceutical Testing Services Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Biopharmaceutical Testing Services Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Biopharmaceutical Testing Services Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Biopharmaceutical Testing Services Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Biopharmaceutical Testing Services Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Biopharmaceutical Testing Services Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Biopharmaceutical Testing Services Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Biopharmaceutical Testing Services Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Biopharmaceutical Testing Services Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Biopharmaceutical Testing Services Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Biopharmaceutical Testing Services Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Biopharmaceutical Testing Services Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Biopharmaceutical Testing Services Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Biopharmaceutical Testing Services Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Biopharmaceutical Testing Services Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Biopharmaceutical Testing Services Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Biopharmaceutical Testing Services Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Biopharmaceutical Testing Services Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Biopharmaceutical Testing Services Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Biopharmaceutical Testing Services Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Biopharmaceutical Testing Services Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Biopharmaceutical Testing Services Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Biopharmaceutical Testing Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Biopharmaceutical Testing Services Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Biopharmaceutical Testing Services?
The projected CAGR is approximately 10.8%.
2. Which companies are prominent players in the Biopharmaceutical Testing Services?
Key companies in the market include Eurofins Scientific, SGS, Envigo, Exova Group, PPD Inc., Pace Analytical Services, Intertek Group, DYNALABS, RD Laboratories, West Pharmaceutical Services, Polymer Solutions, Boston Analytical, Accuratus Labs, Microbac, ARLBioPharma, Lapuck Laboratories, BioScreen.
3. What are the main segments of the Biopharmaceutical Testing Services?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Biopharmaceutical Testing Services," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Biopharmaceutical Testing Services report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Biopharmaceutical Testing Services?
To stay informed about further developments, trends, and reports in the Biopharmaceutical Testing Services, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
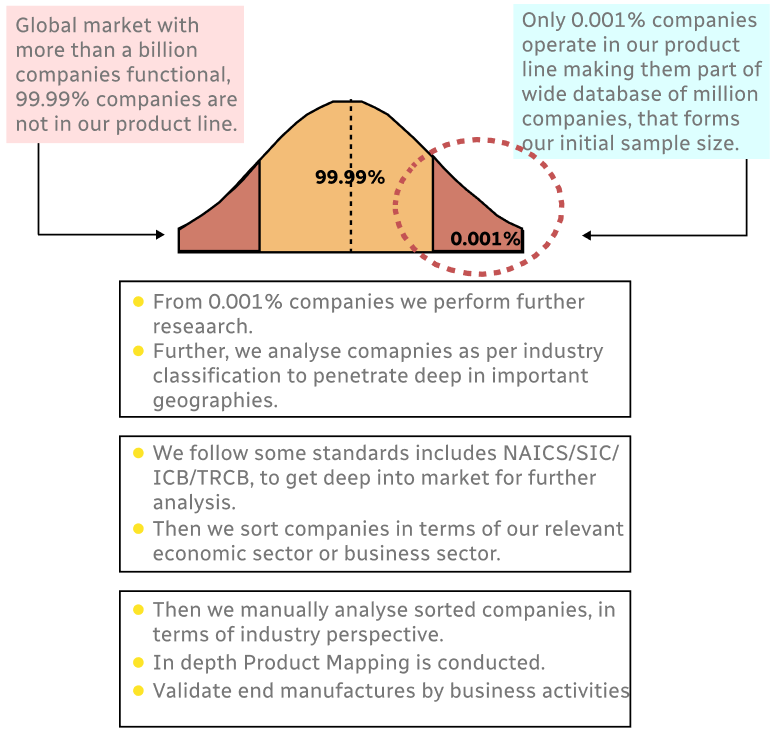
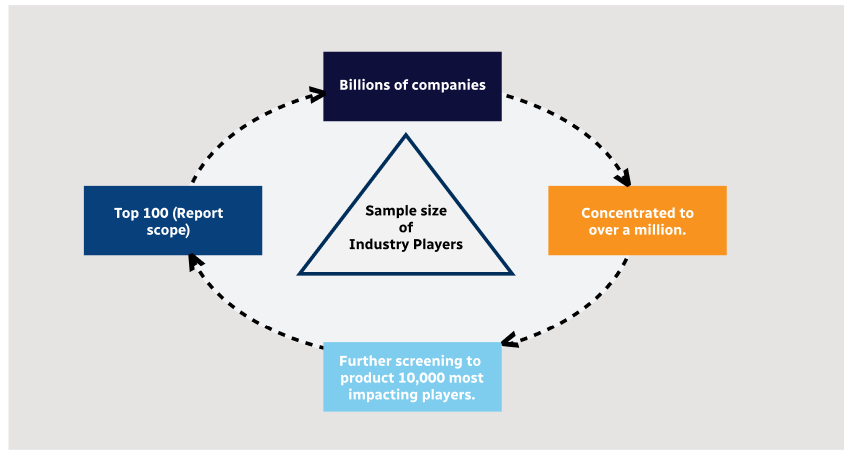
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
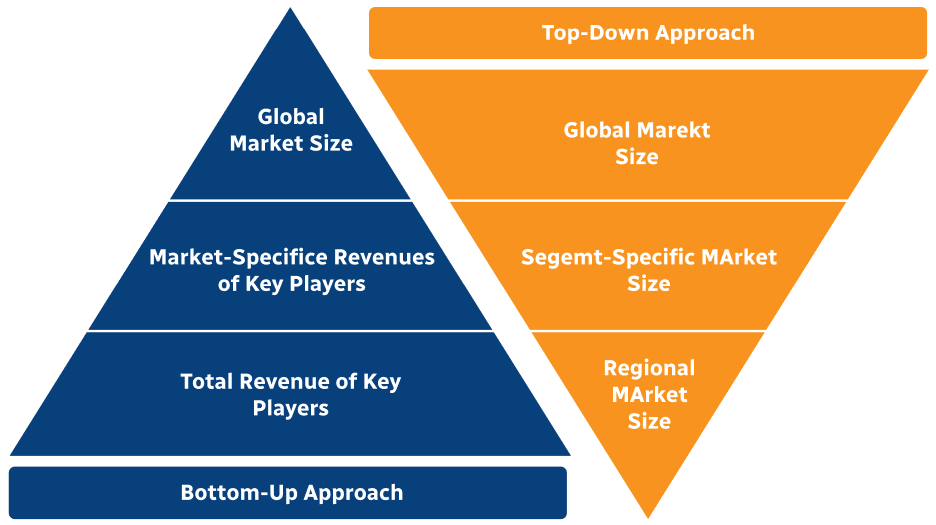
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
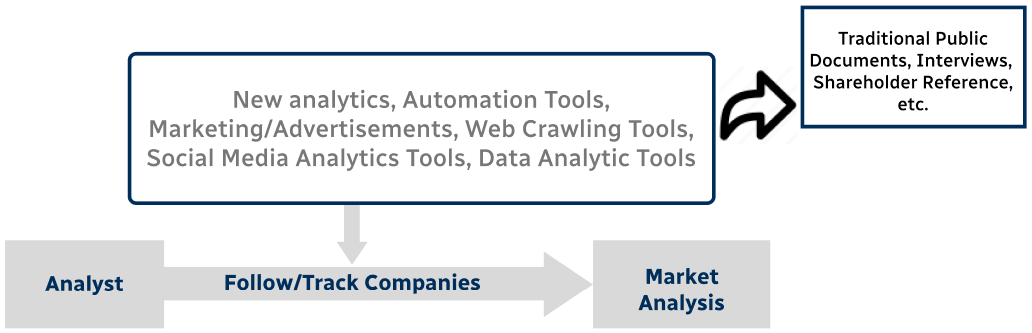
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


