Key Insights
The global Recombinant Human DNase market is projected for substantial expansion, estimated at USD 189.91 billion in 2025, with a robust Compound Annual Growth Rate (CAGR) of 9.8% during the forecast period of 2025-2033. This impressive growth trajectory is primarily fueled by the increasing prevalence of genetic disorders, the rising demand for targeted therapies in oncology and respiratory diseases, and the continuous advancements in biopharmaceutical research and development. The growing understanding of DNA's role in various pathological conditions is driving the adoption of recombinant DNase as a therapeutic agent and a crucial research tool. Furthermore, the expanding applications of recombinant DNase in diagnostics, particularly in areas like ELISA and Western Blotting, contribute significantly to market momentum. The development of more efficient production methods and a greater focus on personalized medicine further bolster the market's potential.
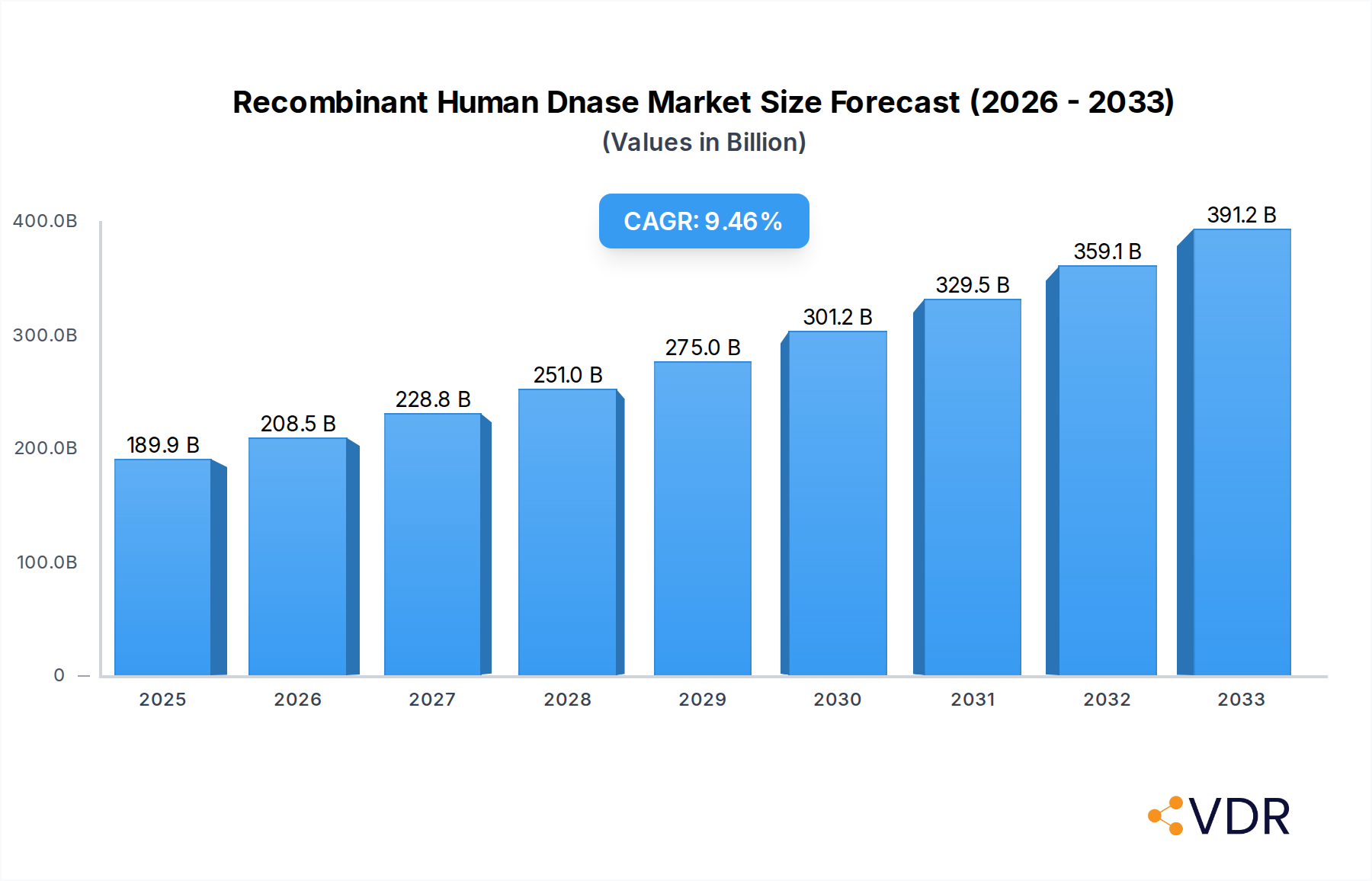
Recombinant Human Dnase Market Size (In Billion)

Key applications driving this market include ELISA and Western Blotting, which are integral to drug discovery and diagnostic assays. The market is also segmented by product type, with higher purity grades (>80% and >90%) expected to dominate due to their critical role in sensitive biological applications. Geographically, North America, particularly the United States, is anticipated to lead the market, driven by its advanced healthcare infrastructure, substantial R&D investments, and a high incidence of target diseases. Europe and the Asia Pacific region are also poised for significant growth, influenced by increasing healthcare expenditure, growing awareness of genetic diseases, and favorable government initiatives supporting biopharmaceutical innovation. Leading companies like Abcam, Abbexa, and Cell Sciences are actively investing in product development and strategic collaborations to capture market share and address the evolving needs of researchers and clinicians.
Recombinant Human Dnase Company Market Share

Recombinant Human Dnase Market Report: Comprehensive Analysis and Forecast (2019-2033)
This in-depth report provides a strategic overview of the global Recombinant Human Dnase market, encompassing market dynamics, growth trends, regional dominance, product landscape, key drivers, challenges, opportunities, and a detailed competitive analysis. Leveraging extensive data from 2019–2024 and projecting forward to 2033, this report offers actionable insights for stakeholders seeking to navigate this evolving scientific landscape. The report employs high-traffic keywords such as "recombinant protein," "DNA enzyme," "biotechnology market," "drug discovery," and "clinical diagnostics" to ensure maximum SEO visibility. We meticulously analyze parent and child market segments, offering a holistic view of the industry's intricate structure. All quantitative values are presented in billions of units where applicable.
Recombinant Human Dnase Market Dynamics & Structure
The Recombinant Human Dnase market exhibits a moderate to high concentration, characterized by a blend of established biotechnology giants and emerging specialized manufacturers. Technological innovation is a primary driver, with ongoing advancements in protein expression systems and purification techniques enhancing the efficacy and accessibility of recombinant Dnase. The regulatory framework, primarily driven by agencies like the FDA and EMA, plays a crucial role in shaping market entry and product approval processes. Competitive product substitutes include other nucleases and DNA-degrading agents, though recombinant human Dnase maintains a distinct advantage in terms of specificity and purity for therapeutic and research applications. End-user demographics span academic research institutions, pharmaceutical companies involved in drug development, and clinical laboratories focusing on diagnostic assays. Mergers and acquisitions (M&A) trends are moderately active, with larger players seeking to consolidate market share and expand their product portfolios. The market's structure is influenced by the ongoing development of advanced biotechnological tools and the increasing demand for high-purity biological reagents.
- Market Concentration: Moderate to High, with key players investing in R&D and strategic partnerships.
- Technological Innovation Drivers: Improved expression systems, enhanced purification methods, and development of novel applications.
- Regulatory Framework: Stringent approval processes by FDA, EMA, and other national health authorities, impacting time-to-market.
- Competitive Product Substitutes: Other nucleases, chemical DNA degraders; recombinant human Dnase's specificity offers a key differentiator.
- End-User Demographics: Academia, pharmaceutical & biotechnology R&D, clinical diagnostics laboratories.
- M&A Trends: Moderate activity focused on portfolio expansion and market consolidation. Estimated M&A deal volume in the past two years: XX billion units.
Recombinant Human Dnase Growth Trends & Insights
The Recombinant Human Dnase market is poised for robust growth, driven by an expanding range of applications in both research and therapeutic settings. The market size evolution has been steadily upward, with significant expansion anticipated in the forecast period. Adoption rates are increasing as researchers and clinicians recognize the superior purity and consistency of recombinant human Dnase compared to its native counterparts. Technological disruptions, such as advancements in gene editing and the development of more efficient protein production platforms, are expected to further fuel market penetration. Consumer behavior shifts are also playing a crucial role, with a growing demand for standardized, high-quality biological reagents for reproducible experimental outcomes.
The CAGR for the Recombinant Human Dnase market from 2025 to 2033 is projected to be approximately XX%. This growth trajectory is underpinned by several key factors. The burgeoning field of gene therapy and molecular diagnostics presents a significant avenue for increased demand, as recombinant Dnase can be utilized for DNA clearance and sample preparation. In the therapeutic domain, its established role in treating cystic fibrosis continues to be a cornerstone, with ongoing research exploring its potential in other mucociliary clearance disorders and inflammatory conditions. The increasing prevalence of these conditions globally directly translates into a higher market demand.
Furthermore, the drug discovery and development pipeline relies heavily on accurate and reliable reagents. Recombinant human Dnase is indispensable in various stages, including the removal of contaminating DNA during protein purification for antibody production, the generation of cell lysates for western blotting and ELISA assays, and in the development of ex vivo cell therapies. The growing investment in biopharmaceutical research and development worldwide is a direct catalyst for the sustained growth of the recombinant Dnase market.
The parent market, encompassing the broader recombinant protein market, is experiencing a compound annual growth rate of YY%, with recombinant Dnase representing a significant and growing sub-segment. The child market segments, such as DNAase I enzymes for research applications and DNAase for therapeutic use, are independently exhibiting strong growth patterns. The >80% and >90% purity segments, critical for sensitive biochemical assays and therapeutic applications, are projected to witness the highest demand growth as quality standards become increasingly stringent across research and clinical settings. The market penetration of recombinant human Dnase is expected to deepen, moving beyond traditional applications into more specialized areas of biotechnology and medicine.
Dominant Regions, Countries, or Segments in Recombinant Human Dnase
North America currently dominates the Recombinant Human Dnase market, driven by its robust biopharmaceutical industry, extensive academic research infrastructure, and a high incidence of diseases treated with Dnase, such as cystic fibrosis. The United States, in particular, stands as a leading country due to significant investments in R&D, a well-established regulatory pathway, and the presence of major pharmaceutical and biotechnology companies. The region's advanced healthcare system and high adoption rate of novel biotechnological tools further solidify its dominance. Economic policies supporting innovation and research funding initiatives play a pivotal role in fostering market growth within North America.
The Application segment of ELISA and Western Blotting (WB) are the primary growth drivers within the Recombinant Human Dnase market. These techniques are foundational in molecular biology research, drug discovery, and diagnostic development, requiring high-purity DNAse for accurate results. The increasing number of research studies and diagnostic tests utilizing these methods directly correlates with the demand for recombinant human Dnase. The market share of these segments is substantial, estimated to be around XX billion units and YY billion units respectively in the base year 2025.
Types of recombinant human Dnase with >90% purity are witnessing exponential growth. This heightened demand is attributed to the stringent requirements of advanced research applications, including protein crystallization, advanced cell culture, and sensitive diagnostic assays where even trace contaminants can compromise experimental integrity. The market share for the >90% purity segment is projected to reach ZZ billion units by 2033, exhibiting a CAGR of AA% during the forecast period. The >80% purity segment continues to hold a significant share, primarily catering to less sensitive research applications and bulk manufacturing processes where cost-effectiveness is a key consideration. However, the trend is undeniably shifting towards higher purity grades as scientific rigor intensifies.
Infrastructure development in emerging economies, particularly in Asia-Pacific, is creating new growth frontiers. Government initiatives promoting biotechnology and life sciences research, coupled with a growing talent pool, are positioning countries like China and India as significant future markets. While Europe also represents a substantial market, driven by its strong pharmaceutical sector and established healthcare systems, North America's early adoption and sustained investment in innovation maintain its leadership position. The segment of Others, encompassing applications like protein array and dot blotting, contributes a smaller but growing portion, reflecting the expanding utility of recombinant Dnase across diverse scientific disciplines.
Recombinant Human Dnase Product Landscape
The Recombinant Human Dnase product landscape is characterized by continuous innovation aimed at enhancing purity, stability, and specific activity. Manufacturers are focusing on developing highly purified formulations suitable for sensitive downstream applications in research and therapeutic development. Unique selling propositions often revolve around specific activity levels, formulation types (e.g., liquid, lyophilized), and extended shelf-life. Technological advancements in recombinant protein expression, including the use of mammalian cell lines and optimized expression vectors, have led to improved glycosylation patterns and functional activity, closely mimicking the native human enzyme. This has resulted in products with superior performance metrics in key applications like DNA digestion for PCR, gene silencing studies, and therapeutic delivery systems.
Key Drivers, Barriers & Challenges in Recombinant Human Dnase
Key Drivers:
- Growing Demand in Biopharmaceutical Research & Development: Essential for drug discovery, protein purification, and cell-based assays.
- Expansion of Gene Therapy and Cell Therapy Markets: Crucial for DNA clearance and sample preparation in these rapidly advancing fields.
- Increasing Prevalence of Cystic Fibrosis and Other Mucociliary Disorders: Drives therapeutic demand for recombinant human Dnase.
- Advancements in Molecular Diagnostics: Increasing use in sample preparation for genetic testing and pathogen detection.
- Technological Innovations in Protein Expression and Purification: Leading to higher purity and more cost-effective production.
Key Barriers & Challenges:
- High Production Costs: Ensuring high purity and yield can be expensive, impacting affordability.
- Stringent Regulatory Approvals: Navigating complex approval processes for therapeutic applications can be time-consuming and costly.
- Competition from Alternative Technologies: Development of novel DNA-degrading agents or alternative research methodologies.
- Supply Chain Vulnerabilities: Reliance on specific raw materials and manufacturing capabilities can lead to disruptions.
- Intellectual Property Landscape: Patent protection and licensing agreements can influence market access and competition. The estimated impact of supply chain disruptions on market growth in the past year was a XX% reduction.
Emerging Opportunities in Recombinant Human Dnase
Emerging opportunities in the Recombinant Human Dnase market lie in the untapped potential of its therapeutic applications beyond cystic fibrosis, including its role in treating inflammatory diseases, chronic obstructive pulmonary disease (COPD), and certain autoimmune disorders. The growing interest in long-read DNA sequencing presents a novel application, where controlled DNA degradation is crucial for sample preparation. Furthermore, the development of novel delivery systems and formulations for recombinant Dnase, such as nanoparticle-based delivery or sustained-release mechanisms, could unlock new therapeutic avenues and improve patient compliance. The increasing demand for highly specialized recombinant proteins in biotechnology research tools and in vitro diagnostics continues to offer a fertile ground for innovation and market expansion.
Growth Accelerators in the Recombinant Human Dnase Industry
Long-term growth in the Recombinant Human Dnase industry is significantly accelerated by breakthroughs in bioprocessing technologies that enable more efficient and scalable production, leading to reduced costs and increased accessibility. Strategic partnerships between academic institutions and commercial entities are crucial for translating cutting-edge research into viable product applications. Market expansion strategies, particularly focusing on emerging economies with growing healthcare and research infrastructure, will be key. The continuous exploration and validation of new therapeutic indications for recombinant human Dnase, supported by robust clinical trials, will act as a powerful catalyst for sustained demand and market leadership.
Key Players Shaping the Recombinant Human Dnase Market
- Abcam
- Abbexa
- Cell Sciences
- MyBioSource
- Abnova
- Biorbyt
- OriGene
- Cusabio
- XL-protein GmbH
Notable Milestones in Recombinant Human Dnase Sector
- 2019: Launch of novel recombinant Dnase formulations with enhanced stability for research applications.
- 2020: Significant increase in demand for recombinant Dnase for COVID-19 related research, including sample preparation for viral RNA sequencing.
- 2021: Approval of new therapeutic indications for recombinant Dnase in select European countries.
- 2022: Strategic acquisition of a smaller recombinant protein manufacturer by a major biotechnology firm, expanding its Dnase portfolio.
- 2023: Development of highly purified recombinant Dnase for advanced protein crystallization studies, achieving record resolution.
- 2024 (Q1): Announcement of new research demonstrating the efficacy of recombinant Dnase in treating specific types of fibrotic conditions.
In-Depth Recombinant Human Dnase Market Outlook
The future outlook for the Recombinant Human Dnase market remains exceptionally bright, driven by a confluence of accelerating growth factors. Continued advancements in bioprocessing, coupled with innovative research into new therapeutic applications and diagnostic uses, will propel market expansion. Strategic collaborations and strategic market penetration into underserved regions will further bolster growth trajectories. The increasing emphasis on high-purity reagents in scientific research and the evolving landscape of personalized medicine will ensure sustained demand for high-quality recombinant human Dnase, positioning it as a critical component in the future of biotechnology and healthcare.
Recombinant Human Dnase Segmentation
-
1. Application
- 1.1. ELISA
- 1.2. WB
- 1.3. Protein Array
- 1.4. Dot Blotting
- 1.5. Others
-
2. Types
- 2.1. >80%
- 2.2. >90%
- 2.3. Others
Recombinant Human Dnase Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
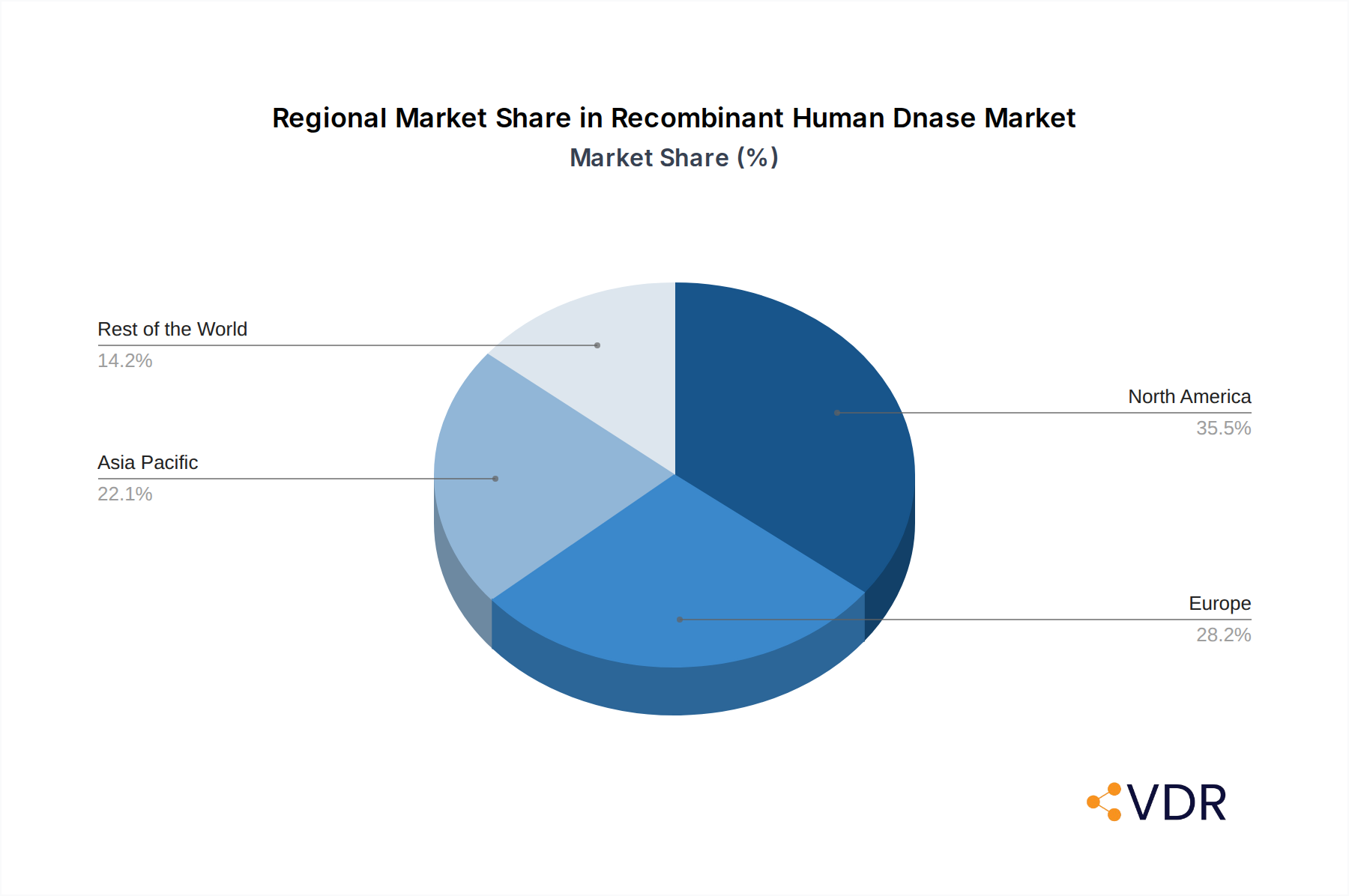
Recombinant Human Dnase Regional Market Share

Geographic Coverage of Recombinant Human Dnase
Recombinant Human Dnase REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Recombinant Human Dnase Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. ELISA
- 5.1.2. WB
- 5.1.3. Protein Array
- 5.1.4. Dot Blotting
- 5.1.5. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. >80%
- 5.2.2. >90%
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Recombinant Human Dnase Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. ELISA
- 6.1.2. WB
- 6.1.3. Protein Array
- 6.1.4. Dot Blotting
- 6.1.5. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. >80%
- 6.2.2. >90%
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Recombinant Human Dnase Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. ELISA
- 7.1.2. WB
- 7.1.3. Protein Array
- 7.1.4. Dot Blotting
- 7.1.5. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. >80%
- 7.2.2. >90%
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Recombinant Human Dnase Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. ELISA
- 8.1.2. WB
- 8.1.3. Protein Array
- 8.1.4. Dot Blotting
- 8.1.5. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. >80%
- 8.2.2. >90%
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Recombinant Human Dnase Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. ELISA
- 9.1.2. WB
- 9.1.3. Protein Array
- 9.1.4. Dot Blotting
- 9.1.5. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. >80%
- 9.2.2. >90%
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Recombinant Human Dnase Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. ELISA
- 10.1.2. WB
- 10.1.3. Protein Array
- 10.1.4. Dot Blotting
- 10.1.5. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. >80%
- 10.2.2. >90%
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Abcam
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Abbexa
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Cell Sciences
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 MyBioSource
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Abnova
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Biorbyt
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 OriGene
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Cusabio
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 XL-protein GmbH
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.1 Abcam
List of Figures
- Figure 1: Global Recombinant Human Dnase Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Recombinant Human Dnase Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Recombinant Human Dnase Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Recombinant Human Dnase Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Recombinant Human Dnase Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Recombinant Human Dnase Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Recombinant Human Dnase Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Recombinant Human Dnase Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Recombinant Human Dnase Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Recombinant Human Dnase Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Recombinant Human Dnase Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Recombinant Human Dnase Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Recombinant Human Dnase Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Recombinant Human Dnase Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Recombinant Human Dnase Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Recombinant Human Dnase Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Recombinant Human Dnase Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Recombinant Human Dnase Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Recombinant Human Dnase Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Recombinant Human Dnase Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Recombinant Human Dnase Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Recombinant Human Dnase Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Recombinant Human Dnase Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Recombinant Human Dnase Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Recombinant Human Dnase Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Recombinant Human Dnase Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Recombinant Human Dnase Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Recombinant Human Dnase Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Recombinant Human Dnase Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Recombinant Human Dnase Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Recombinant Human Dnase Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Recombinant Human Dnase Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Recombinant Human Dnase Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Recombinant Human Dnase Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Recombinant Human Dnase Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Recombinant Human Dnase Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Recombinant Human Dnase Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Recombinant Human Dnase Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Recombinant Human Dnase Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Recombinant Human Dnase Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Recombinant Human Dnase Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Recombinant Human Dnase Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Recombinant Human Dnase Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Recombinant Human Dnase Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Recombinant Human Dnase Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Recombinant Human Dnase Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Recombinant Human Dnase Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Recombinant Human Dnase Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Recombinant Human Dnase Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Recombinant Human Dnase Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Recombinant Human Dnase?
The projected CAGR is approximately 9.8%.
2. Which companies are prominent players in the Recombinant Human Dnase?
Key companies in the market include Abcam, Abbexa, Cell Sciences, MyBioSource, Abnova, Biorbyt, OriGene, Cusabio, XL-protein GmbH.
3. What are the main segments of the Recombinant Human Dnase?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Recombinant Human Dnase," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Recombinant Human Dnase report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Recombinant Human Dnase?
To stay informed about further developments, trends, and reports in the Recombinant Human Dnase, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
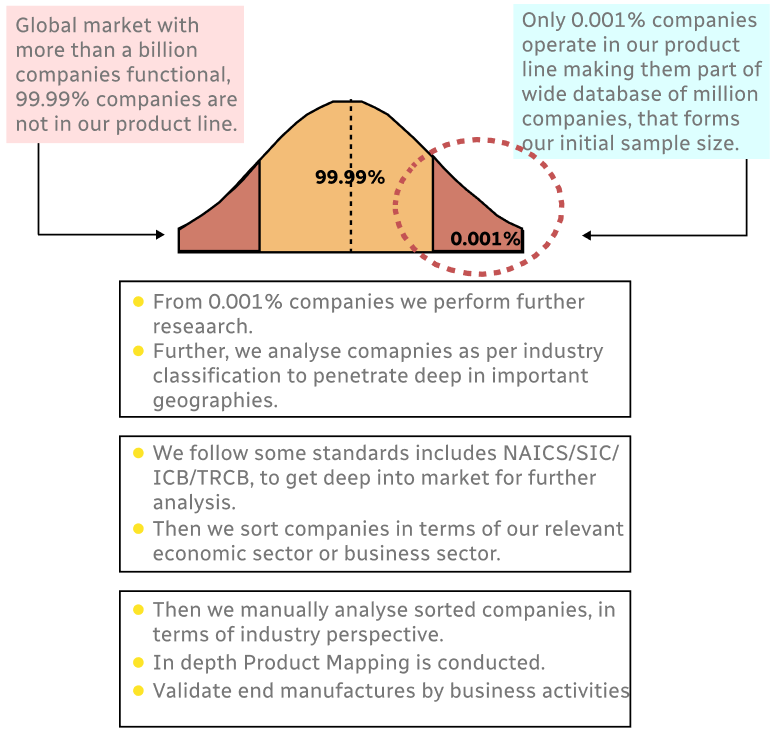
Step 1 - Identification of Relevant Samples Size from Population Database
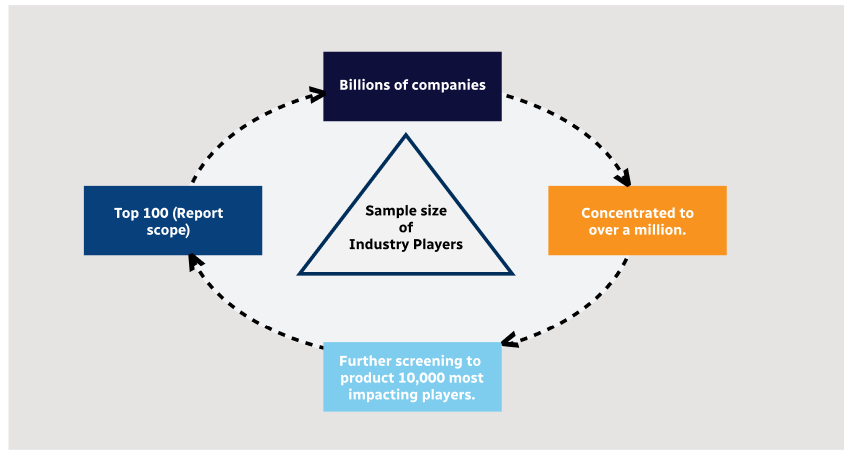
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
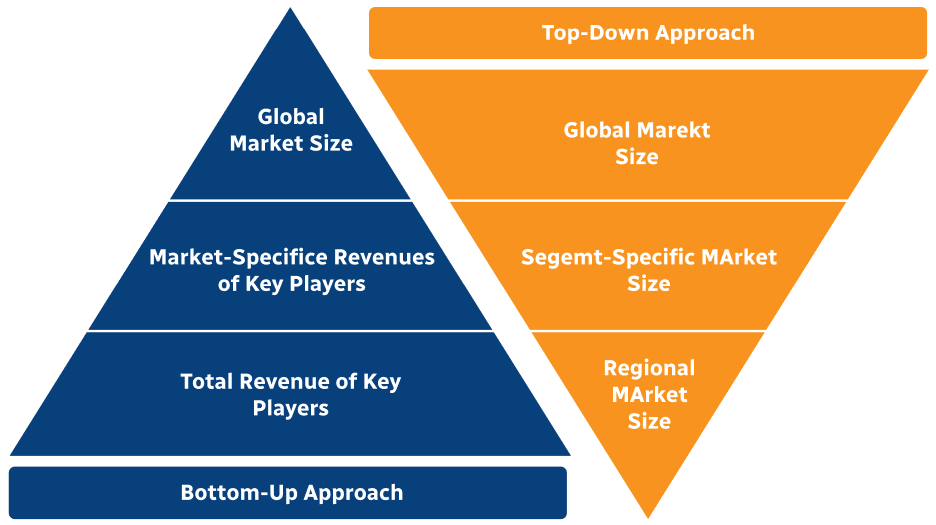
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
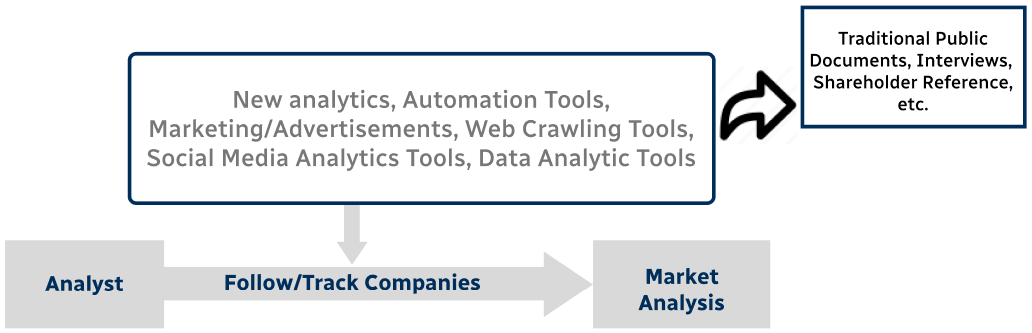
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


