Key Insights
The global preclinical CRO market is poised for substantial growth, projected to reach approximately $55,000 million by 2025 and expand significantly through 2033. Driven by an estimated CAGR of 15%, this market's expansion is primarily fueled by the escalating need for specialized preclinical research services to support the development of novel therapeutics and medical devices. Pharmaceutical and biotechnology companies are increasingly outsourcing these complex and resource-intensive studies to Contract Research Organizations (CROs) to streamline drug discovery, reduce development timelines, and mitigate R&D costs. Furthermore, stringent regulatory requirements and the growing complexity of drug development pipelines are necessitating advanced bioanalysis, toxicology testing, and safety pharmacology, all key services offered by CROs. The increasing focus on precision medicine and personalized therapies also demands sophisticated preclinical models and detailed safety assessments, further bolstering market demand.
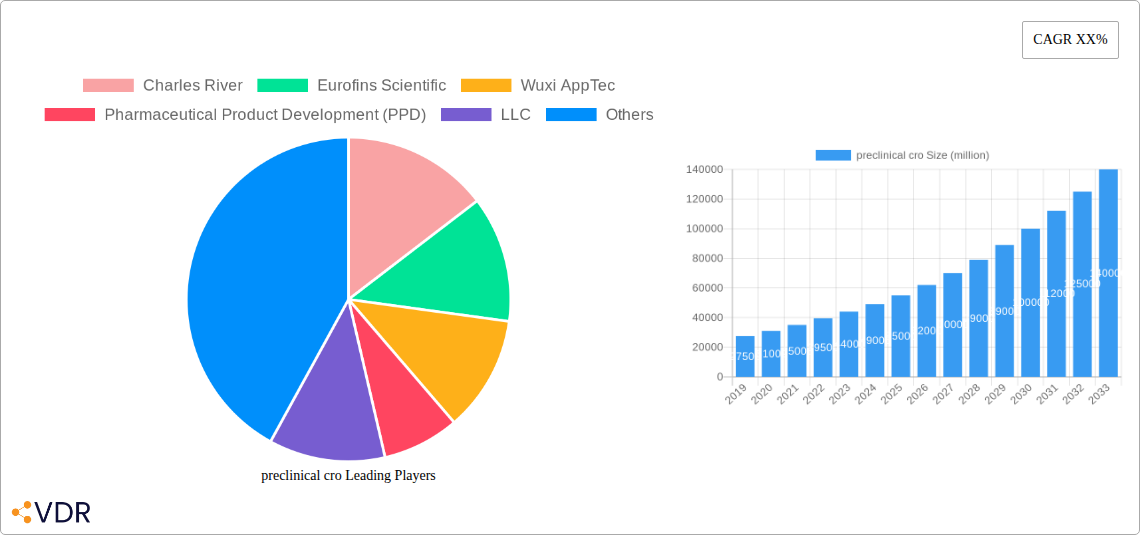
preclinical cro Market Size (In Billion)

The competitive landscape of the preclinical CRO market is characterized by the presence of major global players such as Charles River, Eurofins Scientific, Wuxi AppTec, and Labcorp, alongside a multitude of specialized niche providers. These companies are actively engaged in strategic collaborations, mergers, and acquisitions to expand their service portfolios, geographical reach, and technological capabilities. Key trends include the growing adoption of advanced technologies like AI and machine learning for data analysis and predictive modeling in toxicology, as well as the increasing demand for in vivo and in vitro testing services. However, challenges such as rising operational costs, the need for skilled scientific talent, and the long duration of some preclinical studies present potential restraints to market growth. Despite these hurdles, the robust pipeline of drug candidates across various therapeutic areas and the continuous innovation in healthcare are expected to sustain the upward trajectory of the preclinical CRO market.
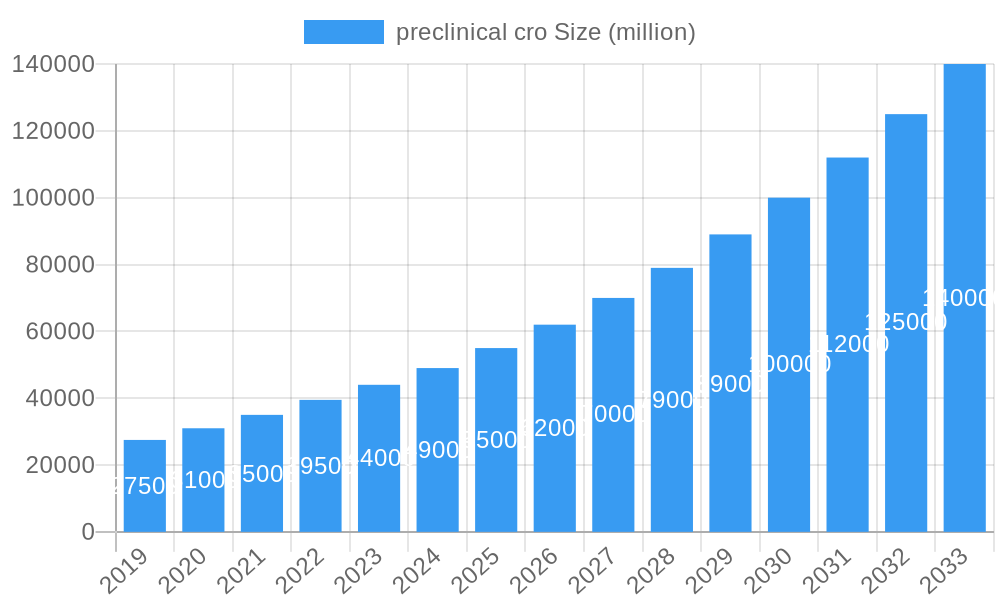
preclinical cro Company Market Share

Preclinical CRO Market Dynamics & Structure
The preclinical Contract Research Organization (CRO) market is characterized by a highly consolidated structure driven by significant technological advancements and stringent regulatory frameworks. Companies like Charles River, Eurofins Scientific, and Wuxi AppTec dominate the landscape, collectively holding an estimated 65% of the global market share. This concentration is fueled by substantial investments in cutting-edge technologies such as AI-driven drug discovery, advanced bio-imaging, and sophisticated toxicology assessment platforms. The primary drivers of innovation include the escalating demand for novel therapeutics, the increasing complexity of drug development, and the need for cost-effective outsourcing solutions by pharmaceutical and biotechnology firms.
- Market Concentration: High, with top 3 players holding ~65% market share.
- Technological Innovation: Driven by AI in drug discovery, advanced bio-imaging, high-throughput screening, and next-generation sequencing.
- Regulatory Frameworks: FDA, EMA, and other national bodies dictate rigorous standards for preclinical testing, influencing service offerings and quality benchmarks.
- Competitive Product Substitutes: In-house R&D capabilities of large pharmaceutical companies, alternative testing methods (e.g., in vitro diagnostics).
- End-User Demographics: Predominantly pharmaceutical companies (estimated 80% of market), followed by biotechnology firms, medical device companies, and academic institutions.
- M&A Trends: Active, with strategic acquisitions aimed at expanding service portfolios, geographical reach, and technological capabilities. For instance, recent M&A activity indicates a trend towards acquiring specialized expertise in areas like gene and cell therapy preclinical development.
Preclinical CRO Growth Trends & Insights
The preclinical Contract Research Organization (CRO) market is poised for substantial expansion, projected to reach an estimated \$38,500 million by 2025 and surge to an impressive \$63,200 million by 2033, exhibiting a robust Compound Annual Growth Rate (CAGR) of approximately 6.5% throughout the forecast period. This impressive growth trajectory is underpinned by a confluence of factors, including the relentless pursuit of innovative drug candidates, the increasing complexity of global regulatory requirements, and the strategic imperative for pharmaceutical and biotechnology companies to optimize their research and development (R&D) expenditures.
The historical period, spanning from 2019 to 2024, witnessed a steady upward trend, with the market size evolving from an estimated \$26,500 million in 2019 to an estimated \$35,200 million in 2024. This growth was propelled by an increasing reliance on outsourcing for specialized preclinical services, driven by the need for expertise and access to advanced technologies that many companies may not possess in-house. The base year, 2025, serves as a pivotal point, with the market estimated at \$38,500 million, setting the stage for accelerated growth in the subsequent years.
Technological disruptions are playing a pivotal role in shaping market dynamics. The integration of artificial intelligence (AI) and machine learning (ML) in drug discovery and development is revolutionizing preclinical testing, enabling faster identification of potential drug candidates and more accurate prediction of efficacy and toxicity. This translates to reduced timelines and improved success rates in early-stage drug development. Furthermore, advancements in areas like organ-on-a-chip technology and 3D bioprinting are offering more sophisticated and human-relevant in vitro models, potentially reducing the reliance on traditional animal testing and enhancing the predictive power of preclinical studies.
Consumer behavior shifts, while less direct in the preclinical CRO market, influence the overall demand for new therapeutics. The growing global health concerns, an aging population, and the prevalence of chronic diseases are fueling the demand for novel treatments, thereby increasing the pipeline of drugs requiring preclinical evaluation. Pharmaceutical companies are responding by intensifying their R&D efforts, a significant portion of which is channeled through CROs. The adoption rates of outsourcing preclinical services are expected to continue to rise as companies recognize the cost-effectiveness, speed, and specialized expertise that CROs provide, particularly for smaller biotech firms with limited internal resources. Market penetration is deepening across various therapeutic areas, indicating a broad-based demand for preclinical CRO services. The estimated market size of \$38,500 million in 2025 underscores the significant role CROs play in the global pharmaceutical value chain, acting as crucial partners in bringing life-saving medicines to market.
Dominant Regions, Countries, or Segments in Preclinical CRO
The Pharmaceutical Companies segment, within the Application category, unequivocally drives the growth of the preclinical CRO market, commanding an estimated 80% of the total market share. This dominance is a direct consequence of the fundamental role preclinical research plays in the drug development lifecycle. Pharmaceutical giants and emerging biotechs alike rely heavily on CROs to conduct a vast array of essential studies, from initial compound screening and characterization to comprehensive toxicology assessments and safety pharmacology. The sheer volume of drug candidates progressing through the pipeline, coupled with the increasing complexity and regulatory scrutiny of these early-stage studies, necessitates a substantial and sustained investment in preclinical CRO services.
The United States stands out as the leading country in the preclinical CRO market, largely due to its robust and well-established pharmaceutical and biotechnology ecosystem. This includes a high concentration of major pharmaceutical companies, a thriving venture capital landscape supporting biotech innovation, and a strong academic research base. Favorable government policies, significant R&D investments, and a well-developed regulatory framework administered by the Food and Drug Administration (FDA) further solidify its leadership position. The presence of a vast network of experienced CROs, including industry leaders like Charles River, Eurofins Scientific, and Wuxi AppTec, alongside specialized niche players, ensures a comprehensive suite of services available to meet the diverse needs of drug developers.
In terms of Types, Toxicology Testing emerges as a dominant segment within the preclinical CRO market. This is driven by the critical need to assess the safety of potential drug candidates before they can proceed to human trials. Regulatory agencies worldwide mandate rigorous toxicology studies to identify potential adverse effects, determine safe dosage levels, and evaluate the overall risk profile of a compound. The increasing complexity of therapeutic modalities, such as biologics and gene therapies, often necessitates more sophisticated and specialized toxicology testing approaches, further boosting the demand for these services. The estimated market share for toxicology testing alone accounts for a significant portion of the overall preclinical CRO market, reflecting its indispensable nature.
- Application Dominance: Pharmaceutical Companies (est. 80% market share) are the primary consumers, requiring extensive preclinical services for their R&D pipelines.
- Geographic Leadership: The United States leads due to its strong biopharmaceutical industry, robust funding, and supportive regulatory environment.
- Type Dominance: Toxicology Testing is a key segment, driven by stringent regulatory requirements and the inherent need to ensure drug safety.
- Key Drivers:
- Increasing R&D Investments: Global pharmaceutical R&D expenditure continues to rise, fueling demand for outsourced preclinical services.
- Emergence of Biologics and Advanced Therapies: These complex modalities require specialized and often novel preclinical testing.
- Stringent Regulatory Requirements: Evolving and rigorous guidelines from agencies like FDA and EMA necessitate expert preclinical evaluations.
- Cost-Effectiveness and Efficiency: Outsourcing allows companies to manage R&D budgets effectively and accelerate timelines.
- Growth Potential: North America and Europe are expected to maintain their leadership, while the Asia-Pacific region, particularly China, is exhibiting rapid growth due to expanding biopharmaceutical sectors and increasing outsourcing by global firms.
Preclinical CRO Product Landscape
The preclinical CRO market is characterized by a sophisticated and evolving product landscape focused on delivering high-quality, reliable data essential for drug development. Companies are innovating in areas such as advanced bioanalysis for complex biologics and small molecules, utilizing LC-MS/MS and other sensitive techniques to quantify drug concentrations and metabolites. DMPK (Drug Metabolism and Pharmacokinetics) studies are increasingly incorporating in silico modeling and physiologically based pharmacokinetic (PBPK) modeling to better predict human absorption, distribution, metabolism, and excretion. Toxicology testing encompasses a wide range of studies, from acute and chronic toxicity to genotoxicity, carcinogenicity, and reproductive toxicology, with an increasing emphasis on utilizing advanced in vitro and in vivo models to enhance predictivity and reduce animal usage. Safety pharmacology assessments are crucial for evaluating potential unintended effects on vital organ systems.
Key Drivers, Barriers & Challenges in Preclinical CRO
Key Drivers:
- Escalating Demand for Novel Therapeutics: The continuous need for new treatments for unmet medical needs fuels R&D and consequently, preclinical outsourcing.
- Advancements in Biotechnology and Drug Discovery: Innovations in areas like gene therapy, cell therapy, and precision medicine require specialized preclinical expertise and infrastructure.
- Cost-Effectiveness and Efficiency Gains: Pharmaceutical companies leverage CROs to optimize R&D budgets and accelerate drug development timelines by accessing specialized capabilities and avoiding in-house infrastructure costs.
- Increasing Regulatory Scrutiny: Stringent global regulatory requirements necessitate high-quality, robust preclinical data, which CROs are well-equipped to provide.
Barriers & Challenges:
- Intense Competition: The market is highly competitive, with numerous global and regional players vying for market share, leading to pricing pressures.
- Regulatory Hurdles and Compliance: Navigating diverse and evolving international regulatory landscapes can be complex and time-consuming, impacting study timelines and costs.
- Talent Acquisition and Retention: A shortage of highly skilled scientists and technical personnel can hinder operational capacity and innovation.
- Data Integrity and Security Concerns: Ensuring the accuracy, reliability, and security of sensitive preclinical data is paramount and requires robust quality management systems.
- Technological Obsolescence: Rapid advancements in technology necessitate continuous investment to remain competitive, which can be a significant financial challenge for some CROs.
Emerging Opportunities in Preclinical CRO
Emerging opportunities in the preclinical CRO market are centered around the development of advanced therapies, personalized medicine, and the integration of cutting-edge technologies. The burgeoning fields of cell and gene therapy, for instance, require highly specialized preclinical evaluation, creating a significant demand for CROs with expertise in these areas. Furthermore, the increasing focus on rare diseases and orphan drugs presents a niche but growing market for CROs that can offer tailored preclinical development strategies. The ongoing development and adoption of AI and machine learning in drug discovery and development also present opportunities for CROs to offer enhanced predictive modeling and data analysis services, leading to faster and more efficient drug candidate selection. There is also a growing demand for advanced in vitro models, such as organoids and organ-on-a-chip systems, offering more human-relevant data and potentially reducing reliance on animal testing.
Growth Accelerators in the Preclinical CRO Industry
The preclinical CRO industry's growth is significantly accelerated by the relentless pace of scientific innovation, particularly in understanding complex disease mechanisms and developing novel therapeutic modalities. The rise of biologics, cell and gene therapies, and RNA-based therapeutics necessitates sophisticated preclinical research capabilities that many pharmaceutical companies strategically outsource. Furthermore, strategic partnerships and collaborations between CROs and pharmaceutical companies, as well as between CROs themselves, are crucial for expanding service portfolios, enhancing technological capabilities, and accessing new markets. Market expansion strategies, including mergers and acquisitions aimed at consolidating market share and acquiring specialized expertise, also serve as powerful growth accelerators. The increasing investment in R&D by emerging biotechnology companies, often lacking extensive in-house preclinical infrastructure, further bolsters the demand for CRO services, acting as a sustained growth catalyst.
Key Players Shaping the Preclinical CRO Market
Charles River Eurofins Scientific Wuxi AppTec Pharmaceutical Product Development (PPD), LLC ICON Plc. Labcorp CRL Crown Bioscience Taconic Biosciences Covance EVOTEC The Jackson Laboratory MI Bioresearch Champion Oncology Xentech Living Tumor Laboratory JOINN Lab
Notable Milestones in Preclinical CRO Sector
- 2020: Wuxi AppTec’s acquisition of Aptuit, significantly expanding its drug discovery and development services.
- 2021: Charles River Laboratories' acquisition of Vigene Biosciences, strengthening its gene therapy manufacturing capabilities.
- 2022: Eurofins Scientific’s expansion of its bioanalysis services through strategic investments in advanced technologies.
- 2023: PPD, LLC (now part of Thermo Fisher Scientific) continued to enhance its clinical trial and preclinical services portfolio.
- 2024: ICON Plc. focused on integrating advanced data analytics and AI into its preclinical offerings.
In-Depth Preclinical CRO Market Outlook
The preclinical CRO market is projected for continued robust growth, driven by the fundamental need for rigorous early-stage drug development and the increasing complexity of therapeutic innovation. Key growth accelerators include the ongoing expansion of biologics and advanced therapy pipelines, a strategic shift towards outsourcing by pharmaceutical and biotech firms seeking specialized expertise and cost efficiencies, and the relentless pursuit of novel treatments for unmet medical needs. Emerging opportunities in areas like cell and gene therapy, alongside the integration of AI and advanced in vitro models, will further shape the market. Strategic partnerships and mergers and acquisitions will continue to consolidate the landscape and enhance service offerings, ensuring CROs remain indispensable partners in bringing life-saving medicines from discovery to clinic. The market outlook remains highly positive, with significant potential for innovation and expansion.
preclinical cro Segmentation
-
1. Application
- 1.1. Pharmaceutical Companies
- 1.2. Medical Device Companies
- 1.3. Other
-
2. Types
- 2.1. Bioanalysis and DMPK Studies
- 2.2. Toxicology Testing
- 2.3. Safety Pharmacology
- 2.4. Others
preclinical cro Segmentation By Geography
- 1. CA
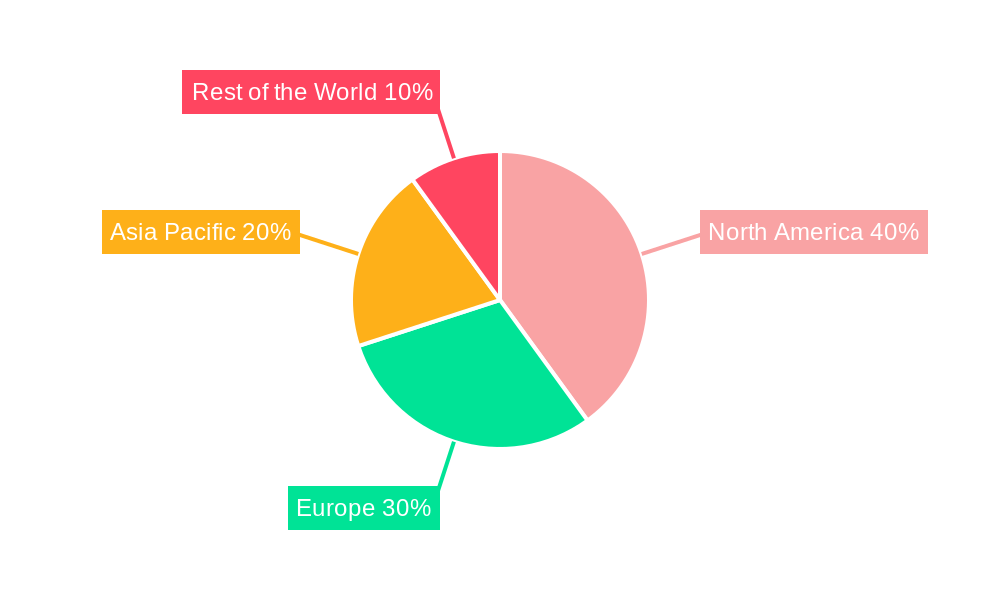
preclinical cro Regional Market Share

Geographic Coverage of preclinical cro
preclinical cro REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. preclinical cro Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Pharmaceutical Companies
- 5.1.2. Medical Device Companies
- 5.1.3. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Bioanalysis and DMPK Studies
- 5.2.2. Toxicology Testing
- 5.2.3. Safety Pharmacology
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Charles River
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Eurofins Scientific
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 Wuxi AppTec
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Pharmaceutical Product Development (PPD)
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 LLC
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 ICON Plc.
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Labcorp
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 CRL
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Crown Bioscience
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Taconic Biosciences
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 Covance
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 EVOTEC
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.13 The Jackson Laboratory
- 6.2.13.1. Overview
- 6.2.13.2. Products
- 6.2.13.3. SWOT Analysis
- 6.2.13.4. Recent Developments
- 6.2.13.5. Financials (Based on Availability)
- 6.2.14 MI Bioresearch
- 6.2.14.1. Overview
- 6.2.14.2. Products
- 6.2.14.3. SWOT Analysis
- 6.2.14.4. Recent Developments
- 6.2.14.5. Financials (Based on Availability)
- 6.2.15 Champion Oncology
- 6.2.15.1. Overview
- 6.2.15.2. Products
- 6.2.15.3. SWOT Analysis
- 6.2.15.4. Recent Developments
- 6.2.15.5. Financials (Based on Availability)
- 6.2.16 Xentech
- 6.2.16.1. Overview
- 6.2.16.2. Products
- 6.2.16.3. SWOT Analysis
- 6.2.16.4. Recent Developments
- 6.2.16.5. Financials (Based on Availability)
- 6.2.17 Living Tumor Laboratory
- 6.2.17.1. Overview
- 6.2.17.2. Products
- 6.2.17.3. SWOT Analysis
- 6.2.17.4. Recent Developments
- 6.2.17.5. Financials (Based on Availability)
- 6.2.18 JOINN Lab
- 6.2.18.1. Overview
- 6.2.18.2. Products
- 6.2.18.3. SWOT Analysis
- 6.2.18.4. Recent Developments
- 6.2.18.5. Financials (Based on Availability)
- 6.2.1 Charles River
List of Figures
- Figure 1: preclinical cro Revenue Breakdown (million, %) by Product 2025 & 2033
- Figure 2: preclinical cro Share (%) by Company 2025
List of Tables
- Table 1: preclinical cro Revenue million Forecast, by Application 2020 & 2033
- Table 2: preclinical cro Revenue million Forecast, by Types 2020 & 2033
- Table 3: preclinical cro Revenue million Forecast, by Region 2020 & 2033
- Table 4: preclinical cro Revenue million Forecast, by Application 2020 & 2033
- Table 5: preclinical cro Revenue million Forecast, by Types 2020 & 2033
- Table 6: preclinical cro Revenue million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the preclinical cro?
The projected CAGR is approximately XX%.
2. Which companies are prominent players in the preclinical cro?
Key companies in the market include Charles River, Eurofins Scientific, Wuxi AppTec, Pharmaceutical Product Development (PPD), LLC, ICON Plc., Labcorp, CRL, Crown Bioscience, Taconic Biosciences, Covance, EVOTEC, The Jackson Laboratory, MI Bioresearch, Champion Oncology, Xentech, Living Tumor Laboratory, JOINN Lab.
3. What are the main segments of the preclinical cro?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "preclinical cro," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the preclinical cro report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the preclinical cro?
To stay informed about further developments, trends, and reports in the preclinical cro, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database


Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


