Key Insights
The Swedish Minimally Invasive Surgery (MIS) Devices Market is poised for significant expansion, projected to reach an estimated USD 94.45 billion in 2025, driven by a robust Compound Annual Growth Rate (CAGR) of 16.1% throughout the forecast period of 2025-2033. This substantial growth is underpinned by increasing adoption of advanced surgical techniques, a growing prevalence of chronic diseases necessitating surgical intervention, and a strong emphasis on patient-centric care that prioritizes reduced recovery times and improved outcomes. Key market drivers include technological advancements in robotic-assisted surgery, sophisticated imaging and visualization tools, and the development of innovative handheld and endoscopic instruments. The growing demand for aesthetic, cardiovascular, and orthopedic procedures, all of which heavily rely on MIS techniques, further propels this market forward. Government initiatives promoting healthcare infrastructure development and reimbursement policies that favor less invasive procedures also contribute to this positive trajectory.
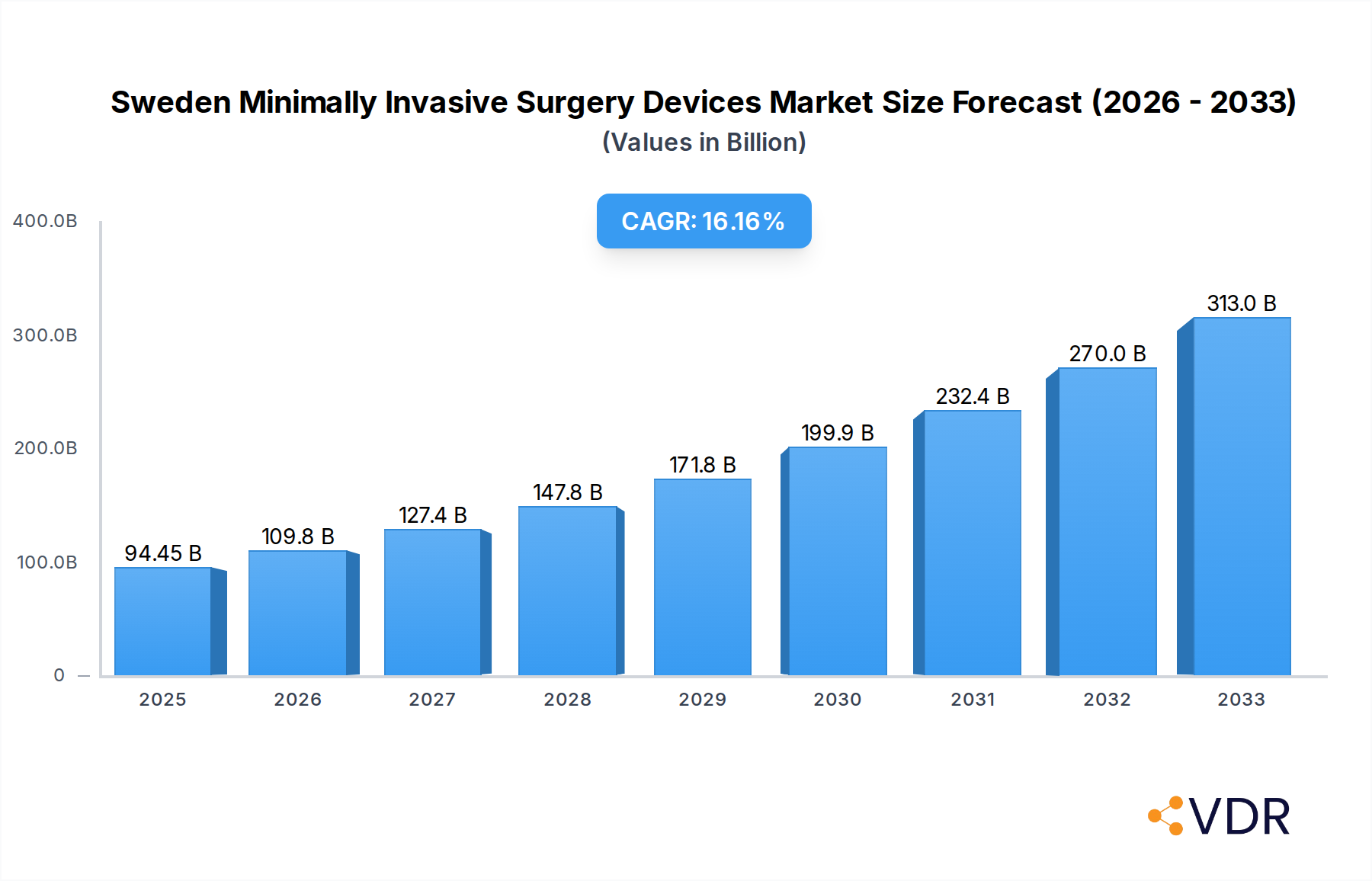
Sweden Minimally Invasive Surgery Devices Market Market Size (In Billion)

The Swedish market for Minimally Invasive Surgery Devices is characterized by a dynamic landscape of innovation and strategic partnerships among leading global players. The market is segmented across a wide array of product categories, including guiding devices (catheters and guidewires), electrosurgical devices, endoscopic and laparoscopic instruments, and advanced ablation and laser-based systems. The growing integration of artificial intelligence and robotics in surgical systems is a notable trend, promising enhanced precision and efficiency. While the market is largely driven by a push for better patient outcomes and cost-effectiveness, potential restraints such as the high initial investment for advanced robotic systems and the need for specialized training for surgical staff need to be carefully managed. Nonetheless, the overall outlook for MIS devices in Sweden remains exceptionally strong, reflecting a commitment to modernizing surgical practices and improving the quality of patient care.
Sweden Minimally Invasive Surgery Devices Market Company Market Share

This comprehensive report delves into the dynamic Sweden minimally invasive surgery (MIS) devices market, offering an in-depth analysis of its current landscape and projected future trajectory. With a study period spanning from 2019 to 2033, and a base year of 2025, this report provides actionable insights for stakeholders seeking to capitalize on the growing demand for advanced surgical technologies. Explore the intricate market structure, key growth drivers, dominant segments, and the competitive strategies of leading players. We dissect the market by product categories including handheld instruments, guiding devices (guiding catheters, guidewires), electrosurgical devices, endoscopic devices, laparoscopic devices, monitoring and visualization devices, ablation devices, laser-based devices, robotic-assisted surgical systems, and other MIS devices. Applications such as aesthetic, cardiovascular, gastrointestinal, gynecological, orthopedic, and urological procedures are also thoroughly examined, alongside emerging trends and strategic opportunities. Unlock the potential of the Swedish MIS market with detailed forecasts and expert analysis.
Sweden Minimally Invasive Surgery Devices Market Market Dynamics & Structure
The Sweden minimally invasive surgery devices market is characterized by a moderately concentrated landscape, with a few key global players holding significant market share. Technological innovation remains a primary driver, fueled by continuous research and development aimed at enhancing surgical precision, reducing patient recovery times, and expanding the scope of minimally invasive procedures. The robust regulatory framework in Sweden, aligned with European Union standards, ensures the safety and efficacy of medical devices, though it can also present certain market entry barriers for new entrants. Competitive product substitutes exist, particularly in areas where traditional open surgery remains an option, but the clear benefits of MIS are increasingly pushing adoption. End-user demographics, driven by an aging population and a growing preference for less invasive treatments, further bolster market growth. Mergers and acquisitions (M&A) are a recurring theme, as larger companies seek to consolidate their market position, acquire innovative technologies, and expand their product portfolios.
- Market Concentration: Dominated by a few large, multinational corporations, with increasing activity from specialized MIS technology providers.
- Technological Innovation Drivers: Emphasis on miniaturization, enhanced imaging capabilities, AI integration, and robotic advancements.
- Regulatory Frameworks: Stringent adherence to EU medical device regulations (MDR) ensures quality and patient safety.
- Competitive Product Substitutes: Traditional open surgery remains a substitute, but the clinical and economic advantages of MIS are diminishing this threat.
- End-User Demographics: An aging population and a growing preference for faster recovery times are key demand drivers.
- M&A Trends: Strategic acquisitions to gain market share, expand product offerings, and acquire cutting-edge technologies.
Sweden Minimally Invasive Surgery Devices Market Growth Trends & Insights
The Sweden minimally invasive surgery devices market is poised for robust expansion, driven by a confluence of factors including increasing healthcare expenditure, a growing demand for advanced surgical techniques, and a favorable reimbursement landscape for MIS procedures. The market size is projected to witness a steady Compound Annual Growth Rate (CAGR) over the forecast period. Adoption rates for MIS devices are consistently rising across various surgical specialties, reflecting the tangible benefits such as reduced patient trauma, shorter hospital stays, and faster return to normal activities. Technological disruptions, particularly in the realm of robotic-assisted surgery and advanced visualization systems, are creating new avenues for market growth. Consumer behavior shifts are also playing a significant role, with patients increasingly seeking out procedures that minimize invasiveness and accelerate recovery. The Swedish healthcare system's commitment to adopting innovative medical technologies further supports these upward trends. The projected market size for the base year of 2025 is estimated to be in the range of USD 2.5 billion to USD 3.0 billion, with a forecast for continued strong growth through 2033.
Dominant Regions, Countries, or Segments in Sweden Minimally Invasive Surgery Devices Market
Within the Sweden minimally invasive surgery devices market, the robotic-assisted surgical systems segment is emerging as a particularly dominant force, showcasing exceptional growth potential. This is largely attributed to the increasing adoption of robotic platforms in complex surgical procedures across various applications, including urological, gynecological, and general surgery. The investment in advanced healthcare infrastructure and the presence of leading academic medical centers in Sweden are instrumental in driving the uptake of these sophisticated technologies. Furthermore, the inherent advantages of robotic surgery, such as enhanced dexterity, precision, and improved ergonomics for surgeons, are key factors contributing to its market leadership.
The Cardiovascular and Orthopedic applications also represent significant market contributors. The high prevalence of cardiovascular diseases and the increasing incidence of orthopedic conditions necessitate advanced treatment modalities, with MIS offering substantial benefits over traditional open procedures. Economic policies supporting technological advancements in healthcare and the growing emphasis on patient outcomes are further accelerating the adoption of MIS devices in these fields.
- Robotic-Assisted Surgical Systems: Dominating due to precision, improved outcomes, and expanding applications in complex surgeries.
- Market share projected to increase significantly by 2033.
- Key drivers include surgeon training programs and hospital infrastructure investments.
- Cardiovascular Applications: Driven by the high burden of cardiovascular diseases and the preference for less invasive interventions.
- Advancements in catheter-based technologies and electrophysiology devices are crucial.
- Orthopedic Applications: Fueled by the rising incidence of joint degeneration and the demand for faster recovery from joint replacement surgeries.
- Growth in arthroscopic surgical instruments and imaging solutions.
- Guiding Devices (Guiding Catheters, Guidewires): Essential components for a wide range of minimally invasive procedures, exhibiting consistent demand.
- Monitoring and Visualization Devices: Critical for accurate diagnosis and procedural guidance, with continuous innovation in imaging resolution and data integration.
Sweden Minimally Invasive Surgery Devices Market Product Landscape
The Sweden minimally invasive surgery devices market is characterized by a diverse product landscape, with continuous innovation driving enhanced surgical capabilities. Robotic-assisted surgical systems represent the pinnacle of technological advancement, offering unparalleled precision and control. Endoscopic and laparoscopic devices remain foundational, with ongoing improvements in imaging clarity and instrument miniaturization. Guiding devices, including advanced guidewires and guiding catheters, are crucial for navigating complex anatomy with minimal trauma. The market also sees significant development in electrosurgical devices and ablation devices for precise tissue management and tumor removal. The focus is on developing integrated systems that offer seamless visualization, data capture, and therapeutic delivery, thereby improving patient outcomes and surgeon efficiency.
Key Drivers, Barriers & Challenges in Sweden Minimally Invasive Surgery Devices Market
Key Drivers:
- Technological Advancements: Continuous innovation in robotics, imaging, and device miniaturization.
- Aging Population: Increased prevalence of age-related diseases requiring surgical intervention.
- Growing Healthcare Expenditure: Government and private sector investments in advanced medical technologies.
- Patient Preference for Minimally Invasive Procedures: Demand for faster recovery and reduced post-operative complications.
- Favorable Reimbursement Policies: Increasing coverage for MIS procedures by insurance providers.
Barriers & Challenges:
- High Initial Cost of Robotic Systems: Significant capital investment required for robotic platforms.
- Stringent Regulatory Approval Processes: Navigating complex regulations can delay market entry.
- Surgeon Training and Adoption: The need for specialized training for new technologies can be a bottleneck.
- Reimbursement Gaps for Novel Technologies: Ensuring adequate reimbursement for cutting-edge MIS devices.
- Competition from Traditional Surgery: In some cases, open surgery may still be preferred due to cost or specific patient factors.
Emerging Opportunities in Sweden Minimally Invasive Surgery Devices Market
Emerging opportunities in the Sweden minimally invasive surgery devices market lie in the continued expansion of robotic surgery into new specialties, such as neurosurgery and complex reconstructive procedures. The integration of artificial intelligence (AI) and machine learning into surgical navigation systems presents a significant growth avenue, offering enhanced predictive analytics and real-time guidance. Furthermore, the development of advanced, single-use MIS instruments can address concerns regarding sterilization and infection control, opening up new market segments. The growing demand for personalized medicine also presents an opportunity for customized MIS solutions tailored to individual patient needs.
Growth Accelerators in the Sweden Minimally Invasive Surgery Devices Market Industry
Growth accelerators for the Sweden minimally invasive surgery devices market include the increasing focus on value-based healthcare, where MIS procedures often demonstrate superior outcomes and cost-effectiveness. Strategic partnerships between medical device manufacturers and healthcare institutions are crucial for driving adoption and innovation. Furthermore, government initiatives promoting the adoption of advanced medical technologies and fostering a conducive environment for research and development will significantly contribute to market expansion. The continuous evolution of materials science and engineering will enable the creation of even smaller, more sophisticated, and less invasive surgical tools.
Key Players Shaping the Sweden Minimally Invasive Surgery Devices Market Market
- Medtronic PLC
- Smith & Nephew
- Koninklijke Philips NV
- Intuitive Surgical Inc
- Siemens Healthineers
- Stryker Corporation
- Johnson & Johnson Inc
- GE Company (GE Healthcare)
- Olympus Corporation
- Abbott Laboratories
- Zimmer Biomet Holdings Inc
Notable Milestones in Sweden Minimally Invasive Surgery Devices Market Sector
- September 2022: Olympus Corporation launched VISERA ELITE III, its newest surgical visualization platform that addresses the needs of healthcare professionals (HCPs) for endoscopic procedures across multiple medical disciplines. VISERA ELITE III offers various imaging functions, all supported in one system, enabling minimally invasive surgeries such as laparoscopic colectomy and laparoscopic cholecystectomy.
- July 2022: Accelus, a privately held medical technology company focused on accelerating the adoption of minimally invasive surgery (MIS) as the standard of care in the spine, announced the closing of a USD 12 million lead investment in the company's Series D equity financing by Concord Innovation Fund II, LP.
In-Depth Sweden Minimally Invasive Surgery Devices Market Market Outlook
The future outlook for the Sweden minimally invasive surgery devices market is exceptionally positive, driven by ongoing technological advancements and a persistent demand for improved patient care. The increasing adoption of robotic surgery, coupled with innovations in AI-powered surgical guidance, will continue to be major growth catalysts. Investments in R&D, strategic collaborations, and supportive government policies are expected to further accelerate market penetration. The market is well-positioned to capitalize on the growing preference for outpatient procedures and same-day surgeries, further enhancing its attractiveness. Opportunities abound in the development of more compact and affordable MIS solutions, catering to a wider range of healthcare facilities and expanding access to advanced surgical care across Sweden. The projected market size is expected to reach significant figures by 2033, underscoring the robust growth trajectory.
Sweden Minimally Invasive Surgery Devices Market Segmentation
-
1. Product
- 1.1. Handheld Instruments
-
1.2. Guiding Devices
- 1.2.1. Guiding Catheters
- 1.2.2. Guidewires
- 1.3. Electrosurgical Devices
- 1.4. Endoscopic Devices
- 1.5. Laproscopic Devices
- 1.6. Monitoring and Visualization Devices
- 1.7. Ablation Devices
- 1.8. Laser-based Devices
- 1.9. Robotic-assisted Surgical Systems
- 1.10. Other MIS Devices
-
2. Application
- 2.1. Aesthetic
- 2.2. Cardiovascular
- 2.3. Gastrointestinal
- 2.4. Gynecological
- 2.5. Orthopedic
- 2.6. Urological
- 2.7. Other Applications
Sweden Minimally Invasive Surgery Devices Market Segmentation By Geography
- 1. Sweden
Sweden Minimally Invasive Surgery Devices Market Regional Market Share

Geographic Coverage of Sweden Minimally Invasive Surgery Devices Market
Sweden Minimally Invasive Surgery Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 16.1% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Handheld Instruments
- 5.1.2. Guiding Devices
- 5.1.2.1. Guiding Catheters
- 5.1.2.2. Guidewires
- 5.1.3. Electrosurgical Devices
- 5.1.4. Endoscopic Devices
- 5.1.5. Laproscopic Devices
- 5.1.6. Monitoring and Visualization Devices
- 5.1.7. Ablation Devices
- 5.1.8. Laser-based Devices
- 5.1.9. Robotic-assisted Surgical Systems
- 5.1.10. Other MIS Devices
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Aesthetic
- 5.2.2. Cardiovascular
- 5.2.3. Gastrointestinal
- 5.2.4. Gynecological
- 5.2.5. Orthopedic
- 5.2.6. Urological
- 5.2.7. Other Applications
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. Sweden
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Sweden Minimally Invasive Surgery Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Handheld Instruments
- 6.1.2. Guiding Devices
- 6.1.2.1. Guiding Catheters
- 6.1.2.2. Guidewires
- 6.1.3. Electrosurgical Devices
- 6.1.4. Endoscopic Devices
- 6.1.5. Laproscopic Devices
- 6.1.6. Monitoring and Visualization Devices
- 6.1.7. Ablation Devices
- 6.1.8. Laser-based Devices
- 6.1.9. Robotic-assisted Surgical Systems
- 6.1.10. Other MIS Devices
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Aesthetic
- 6.2.2. Cardiovascular
- 6.2.3. Gastrointestinal
- 6.2.4. Gynecological
- 6.2.5. Orthopedic
- 6.2.6. Urological
- 6.2.7. Other Applications
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Medtronic PLC
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Smith & Nephew
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Koninklijke Philips NV
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Intuitive Surgical Inc
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Siemens Healthineers
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Stryker Corporation
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Johnson & Johnson Inc
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 GE Company (GE Healthcare)
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Olympus Corporation
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 Abbott Laboratories
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.11 Zimmer Biomet Holdings Inc
- 7.1.11.1. Company Overview
- 7.1.11.2. Products
- 7.1.11.3. Company Financials
- 7.1.11.4. SWOT Analysis
- 7.1.1 Medtronic PLC
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Sweden Minimally Invasive Surgery Devices Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Sweden Minimally Invasive Surgery Devices Market Share (%) by Company 2025
List of Tables
- Table 1: Sweden Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 2: Sweden Minimally Invasive Surgery Devices Market Volume K Unit Forecast, by Product 2020 & 2033
- Table 3: Sweden Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Application 2020 & 2033
- Table 4: Sweden Minimally Invasive Surgery Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 5: Sweden Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Sweden Minimally Invasive Surgery Devices Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 7: Sweden Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 8: Sweden Minimally Invasive Surgery Devices Market Volume K Unit Forecast, by Product 2020 & 2033
- Table 9: Sweden Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Application 2020 & 2033
- Table 10: Sweden Minimally Invasive Surgery Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 11: Sweden Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Sweden Minimally Invasive Surgery Devices Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Sweden Minimally Invasive Surgery Devices Market?
The projected CAGR is approximately 16.1%.
2. Which companies are prominent players in the Sweden Minimally Invasive Surgery Devices Market?
Key companies in the market include Medtronic PLC, Smith & Nephew, Koninklijke Philips NV, Intuitive Surgical Inc, Siemens Healthineers, Stryker Corporation, Johnson & Johnson Inc , GE Company (GE Healthcare), Olympus Corporation, Abbott Laboratories, Zimmer Biomet Holdings Inc.
3. What are the main segments of the Sweden Minimally Invasive Surgery Devices Market?
The market segments include Product, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 94.45 billion as of 2022.
5. What are some drivers contributing to market growth?
Higher Acceptance Rate of Minimally Invasive Surgeries over Traditional Surgeries and Technological Advancements; Increasing Prevalence of Lifestyle-related and Chronic Disorders.
6. What are the notable trends driving market growth?
Gastrointestinal Segment is Expected to Hold the Major Market Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Shortage of Experienced Professionals and Uncertain Regulatory Framework.
8. Can you provide examples of recent developments in the market?
In September 2022, Olympus Corporation launched VISERA ELITE III, its newest surgical visualization platform that addresses the needs of healthcare professionals (HCPs) for endoscopic procedures across multiple medical disciplines. VISERA ELITE III offers various imaging functions, all supported in one system, enabling minimally invasive surgeries such as laparoscopic colectomy and laparoscopic cholecystectomy.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Sweden Minimally Invasive Surgery Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Sweden Minimally Invasive Surgery Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Sweden Minimally Invasive Surgery Devices Market?
To stay informed about further developments, trends, and reports in the Sweden Minimally Invasive Surgery Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
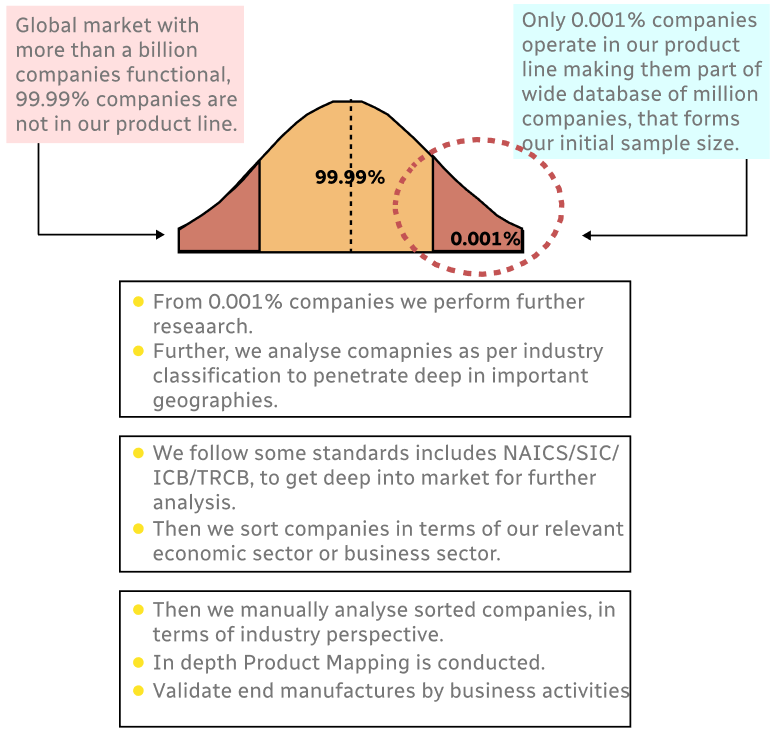
Step 1 - Identification of Relevant Samples Size from Population Database
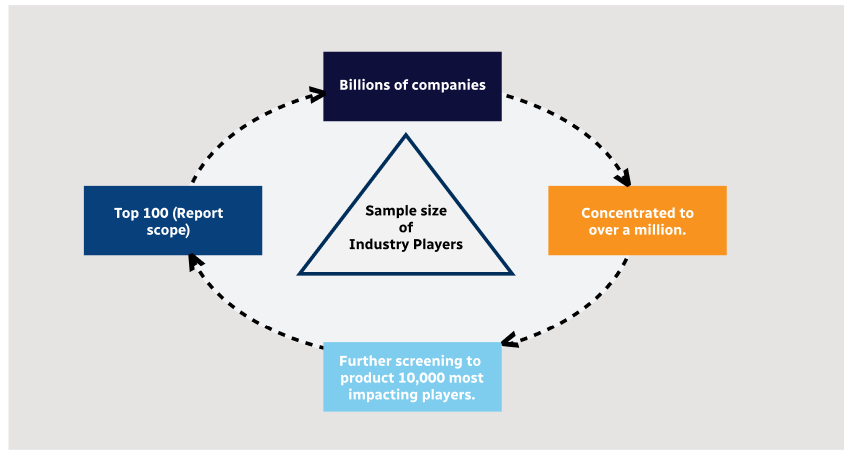
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
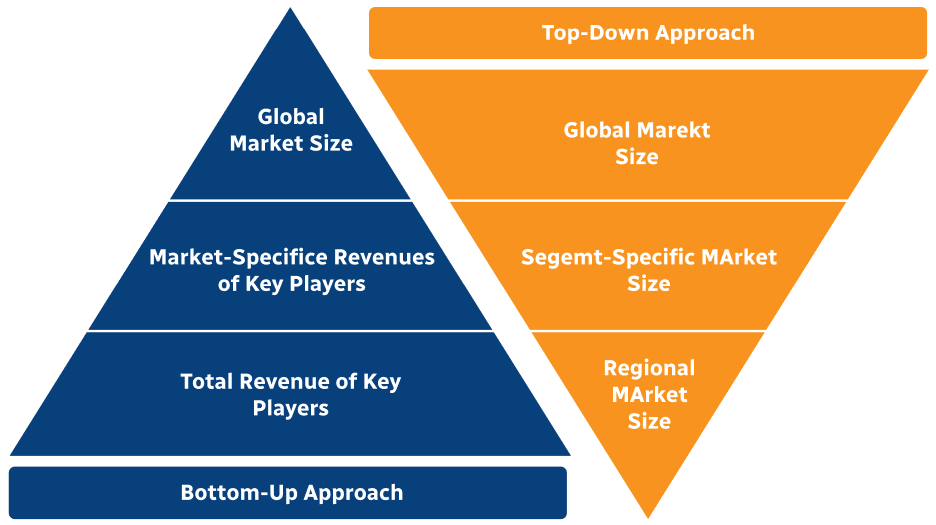
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
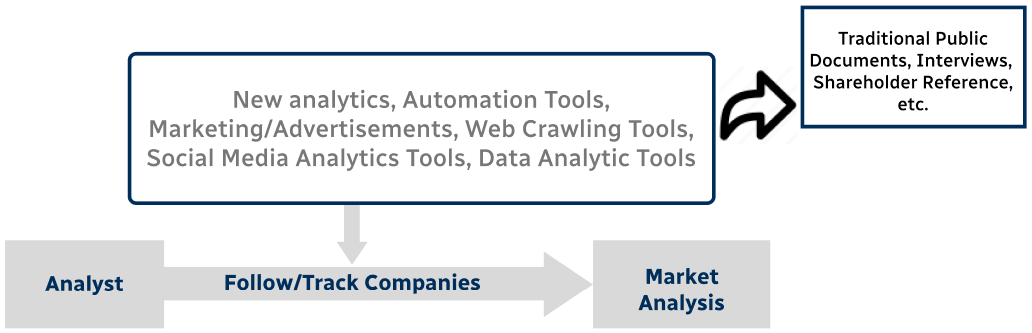
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


