Key Insights
The global LIMS (Laboratory Information Management System) market for pharmaceutical laboratories is poised for significant expansion, driven by the increasing need for robust data management, regulatory compliance, and enhanced operational efficiency within R&D and quality control processes. The market is estimated to reach USD 4.07 billion in 2025, showcasing a healthy projected Compound Annual Growth Rate (CAGR) of 4.9% over the forecast period of 2025-2033. This growth is fueled by the burgeoning pharmaceutical industry, which demands sophisticated solutions to manage vast amounts of data generated during drug discovery, development, and manufacturing. The escalating complexity of research, coupled with stringent regulatory frameworks from bodies like the FDA and EMA, necessitates the adoption of LIMS to ensure data integrity, traceability, and adherence to Good Laboratory Practices (GLP) and Good Manufacturing Practices (GMP). Furthermore, advancements in cloud-based LIMS solutions are democratizing access to powerful laboratory management tools, making them more scalable and cost-effective for a wider range of pharmaceutical organizations, from large enterprises to smaller research institutions.
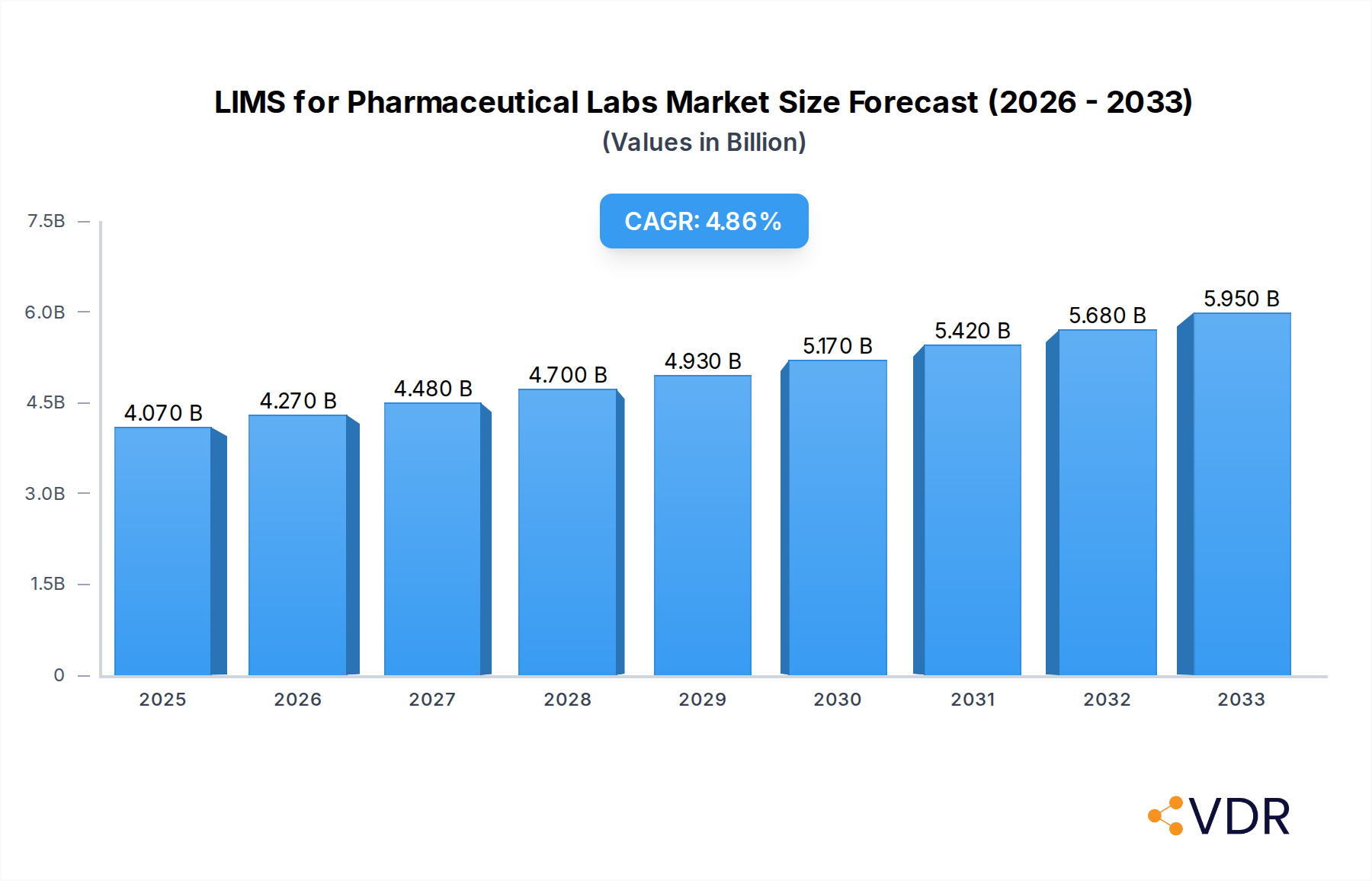
LIMS for Pharmaceutical Labs Market Size (In Billion)

Key drivers propelling this market forward include the increasing investment in pharmaceutical R&D, the growing emphasis on data analytics and AI in drug discovery, and the continuous evolution of laboratory automation. Pharmaceutical factories, in particular, are leveraging LIMS to streamline workflows, reduce manual errors, and accelerate time-to-market for new therapies. While the adoption of sophisticated LIMS solutions is a significant investment, the long-term benefits in terms of improved data accuracy, enhanced sample tracking, efficient resource allocation, and reduced compliance risks are making it an indispensable technology. The market is segmented into applications like research institutions and pharmaceutical factories, and deployment types such as on-premises and cloud-based solutions, reflecting the diverse needs of the pharmaceutical sector. Leading companies in this space are continuously innovating to offer more integrated and intelligent LIMS platforms that can adapt to the dynamic landscape of pharmaceutical research and production.
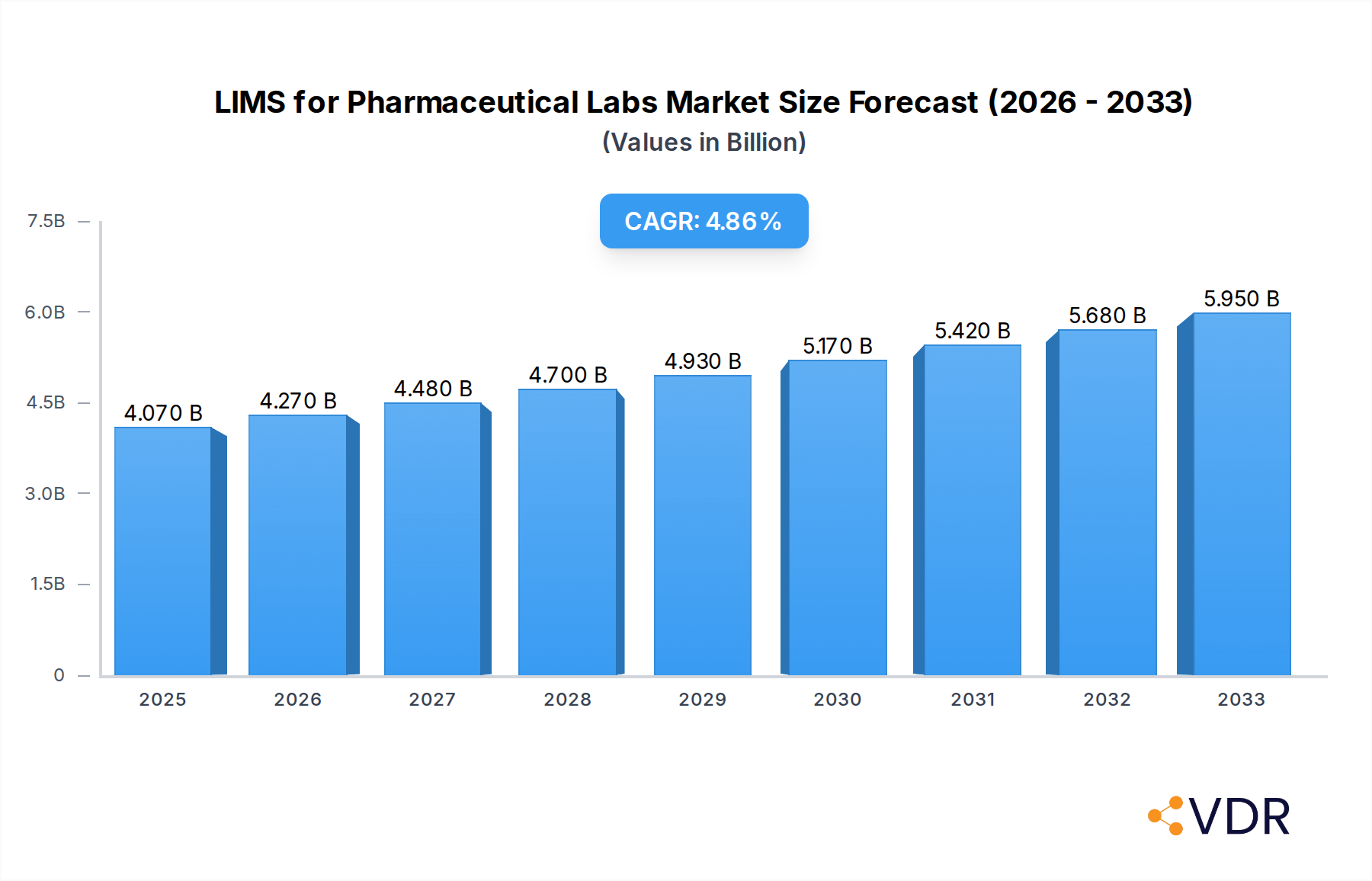
LIMS for Pharmaceutical Labs Company Market Share

This in-depth report provides a definitive analysis of the Laboratory Information Management System (LIMS) for Pharmaceutical Labs market, offering a granular view of its current state and future trajectory. Covering the study period of 2019–2033, with a base year of 2025 and a forecast period extending to 2033, this report is an indispensable resource for stakeholders seeking to understand and capitalize on the evolving landscape of pharmaceutical laboratory informatics. The market is segmented by application into Research Institutions and Pharmaceutical Factories, and by type into On-premises and Cloud Based LIMS solutions. This report integrates high-traffic keywords such as "pharmaceutical LIMS," "laboratory software," "drug development informatics," "clinical research LIMS," and "pharma quality control software" to maximize SEO visibility and engage industry professionals. We analyze parent and child market dynamics to offer comprehensive insights.
LIMS for Pharmaceutical Labs Market Dynamics & Structure
The LIMS for Pharmaceutical Labs market is characterized by a moderate to high degree of concentration, with key players like Thermo Scientific, LabWare, and LabVantage holding significant market shares. Technological innovation remains a primary driver, fueled by the increasing demand for enhanced data integrity, regulatory compliance (e.g., FDA 21 CFR Part 11, ICH guidelines), and the need for efficient sample tracking and workflow automation in drug discovery and development. Regulatory frameworks, such as those mandated by the FDA and EMA, play a crucial role in shaping LIMS adoption, pushing organizations towards robust and validated systems. Competitive product substitutes, including Electronic Lab Notebooks (ELNs) and specialized data analysis tools, are emerging but often complement rather than replace comprehensive LIMS functionalities. End-user demographics are expanding to include smaller biotech firms and contract research organizations (CROs) alongside large pharmaceutical enterprises. Mergers and acquisitions (M&A) are a notable trend, with larger vendors acquiring smaller, specialized LIMS providers to broaden their product portfolios and market reach. For instance, the historical period of 2019-2024 saw several strategic acquisitions aimed at consolidating market presence and integrating advanced functionalities like AI and machine learning.
- Market Concentration: Moderate to High, with top 3-5 players holding an estimated xx% of the market share.
- Technological Innovation Drivers: Data integrity, AI/ML integration, IoT connectivity, and automation.
- Regulatory Frameworks: Strict adherence to FDA 21 CFR Part 11, ICH Q2, and global pharmacopeia standards.
- Competitive Product Substitutes: ELNs, scientific data management systems (SDMS).
- End-User Demographics: Large pharmaceutical companies, biotech firms, CROs, academic research institutions.
- M&A Trends: Consolidation, acquisition of niche solution providers for feature expansion.
LIMS for Pharmaceutical Labs Growth Trends & Insights
The global LIMS for Pharmaceutical Labs market is projected to witness robust growth, driven by an escalating demand for streamlined laboratory operations and stringent quality control measures within the pharmaceutical industry. The market size, estimated at approximately $xx billion in 2025, is expected to reach $xx billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately xx% during the forecast period (2025–2033). This expansion is underpinned by the increasing complexity of drug development pipelines, the growing volume of research data, and the paramount importance of regulatory compliance. Adoption rates for LIMS are steadily increasing across all segments, particularly in cloud-based solutions, which offer scalability, accessibility, and cost-effectiveness. Technological disruptions, including the integration of Artificial Intelligence (AI) and Machine Learning (ML) for predictive analytics and enhanced decision-making, are transforming LIMS capabilities. Consumer behavior is shifting towards solutions that offer greater interoperability, advanced reporting, and a more intuitive user experience, pushing vendors to innovate continuously. The historical period of 2019–2024 saw steady market expansion, laying the groundwork for accelerated growth in the coming years. The market penetration for LIMS in pharmaceutical R&D is estimated to be xx% in 2025, with significant room for further growth.
Dominant Regions, Countries, or Segments in LIMS for Pharmaceutical Labs
The North America region is anticipated to continue its dominance in the LIMS for Pharmaceutical Labs market, largely driven by the United States' robust pharmaceutical and biotechnology sectors, substantial R&D investments, and a well-established regulatory environment. The Pharmaceutical Factory application segment represents the largest market share, owing to the critical need for quality control, batch release testing, and compliance in manufacturing processes. Furthermore, Cloud Based LIMS solutions are experiencing a surge in adoption, gradually eclipsing traditional on-premises deployments due to their inherent scalability, flexibility, and reduced IT overhead. Key drivers contributing to North America's supremacy include significant government funding for life sciences research, the presence of leading pharmaceutical giants, and a proactive approach to adopting advanced technologies.
- Dominant Region: North America
- Key Drivers: High R&D expenditure, presence of major pharmaceutical and biotech companies, favorable regulatory landscape, early adoption of advanced technologies.
- Market Share Contribution: Estimated at xx% of the global market in 2025.
- Growth Potential: Continued expansion driven by innovation in drug discovery and personalized medicine.
- Dominant Application Segment: Pharmaceutical Factory
- Key Drivers: Stringent quality control requirements, batch management, regulatory compliance, process optimization, sample throughput.
- Market Share Contribution: Estimated at xx% of the global market in 2025.
- Growth Potential: Increasing complexity of manufacturing, demand for automation.
- Dominant Type of LIMS: Cloud Based
- Key Drivers: Scalability, cost-effectiveness, remote accessibility, disaster recovery, faster deployment, regular updates.
- Market Share Contribution: Expected to grow from xx% in 2025 to xx% by 2033.
- Growth Potential: High adoption rates driven by preference for flexible and accessible solutions.
LIMS for Pharmaceutical Labs Product Landscape
The LIMS for Pharmaceutical Labs product landscape is dynamic, marked by continuous innovation focused on enhancing efficiency, accuracy, and compliance. Leading vendors are differentiating their offerings through advanced features such as AI-powered analytics for trend identification, real-time data dashboards for enhanced visibility, and seamless integration with other laboratory instruments and enterprise resource planning (ERP) systems. Specific applications span raw material testing, stability studies, method validation, and environmental monitoring. Performance metrics are increasingly evaluated based on speed of data retrieval, ease of use, system uptime, and the ability to generate compliant reports. Unique selling propositions often revolve around specialized modules for specific pharmaceutical workflows, robust audit trails, and customizable reporting capabilities that meet stringent global regulatory demands. Technological advancements include the integration of blockchain for enhanced data security and integrity.
Key Drivers, Barriers & Challenges in LIMS for Pharmaceutical Labs
Key Drivers: The LIMS for Pharmaceutical Labs market is primarily propelled by the escalating need for enhanced data integrity and regulatory compliance across the pharmaceutical value chain. The increasing complexity of drug discovery and development, coupled with the growing volume of research data, necessitates efficient sample management and workflow automation. Furthermore, advancements in AI and machine learning are enabling more sophisticated data analysis and predictive capabilities, driving adoption. The shift towards cloud-based solutions offers scalability and cost-effectiveness, further accelerating market growth.
Barriers & Challenges: Despite strong growth drivers, the market faces several challenges. The high initial cost of implementation and ongoing maintenance for some sophisticated LIMS can be a significant barrier, especially for smaller enterprises. Integrating LIMS with existing legacy systems can also be complex and time-consuming. Stringent validation requirements for pharmaceutical software, while ensuring quality, add to the implementation timeline and cost. Moreover, a shortage of skilled IT professionals proficient in LIMS implementation and management can hinder widespread adoption. Supply chain disruptions affecting hardware availability could also pose a temporary challenge.
Emerging Opportunities in LIMS for Pharmaceutical Labs
Emerging opportunities in the LIMS for Pharmaceutical Labs sector lie in the increasing demand for specialized LIMS solutions tailored for niche areas within drug development, such as biopharmaceutical manufacturing, personalized medicine, and cell and gene therapies. The integration of Internet of Things (IoT) devices for real-time data capture from laboratory instruments presents a significant avenue for enhanced automation and efficiency. Furthermore, the growing focus on sustainability in pharmaceutical operations opens opportunities for LIMS that can track and manage environmental impact data. Untapped markets in emerging economies with developing pharmaceutical industries also represent a substantial growth potential, provided localized solutions and support are offered.
Growth Accelerators in the LIMS for Pharmaceutical Labs Industry
Several key factors are accelerating growth in the LIMS for Pharmaceutical Labs industry. Technological breakthroughs, particularly in AI and machine learning, are enabling more intelligent data analysis, predictive modeling, and automated anomaly detection, significantly enhancing R&D and quality control processes. Strategic partnerships between LIMS vendors and other technology providers, such as those specializing in data analytics or laboratory automation, are leading to more integrated and comprehensive solutions. Market expansion strategies, including targeting emerging economies and offering tiered solutions to cater to businesses of all sizes, are also crucial growth accelerators. The increasing outsourcing of R&D activities to Contract Research Organizations (CROs) further bolsters demand for robust LIMS.
Key Players Shaping the LIMS for Pharmaceutical Labs Market
- Thermo Scientific
- LabWare
- LabVantage
- Ocimum Biosolution
- Apex Healthware
- Agilent
- Agaram
- LabSols
- Benchling
- Autoscribe Informatics
- AQ Manager
- Eusoft
- Labguru
- Blaze Systems
- Third Wave Analytics
- GUS LAB
- LabCollector
- Clinisys
- Revol LIMS
- QSI
- ATL
Notable Milestones in LIMS for Pharmaceutical Labs Sector
- 2019: Increased adoption of AI and ML capabilities within LIMS for predictive analytics and enhanced data interpretation.
- 2020: Significant surge in demand for cloud-based LIMS solutions due to remote work trends and the need for flexible data access during the global pandemic.
- 2021: Major LIMS vendors focused on integrating advanced IoT functionalities for real-time data acquisition from laboratory instruments.
- 2022: Growing emphasis on cybersecurity features and robust audit trails to meet evolving regulatory compliance standards.
- 2023: Launch of LIMS solutions with enhanced interoperability features, enabling seamless integration with other scientific software and enterprise systems.
- 2024: Increased investment in AI-driven automation and predictive maintenance modules within pharmaceutical LIMS.
In-Depth LIMS for Pharmaceutical Labs Market Outlook
The future outlook for the LIMS for Pharmaceutical Labs market is exceptionally strong, driven by ongoing technological advancements and the persistent demand for data-driven efficiency and unwavering regulatory compliance. Growth accelerators such as the pervasive integration of AI and machine learning, the strategic expansion through partnerships, and the development of tailored solutions for specialized pharmaceutical sectors will continue to fuel market expansion. The increasing adoption of cloud-based infrastructure and the penetration into emerging markets present significant untapped potential. Stakeholders can anticipate a market characterized by sophisticated, interconnected, and highly intelligent LIMS solutions that are critical for accelerating drug discovery, ensuring product quality, and navigating the complex global regulatory landscape. The market is poised for sustained growth, offering substantial strategic opportunities for innovation and market leadership.
LIMS for Pharmaceutical Labs Segmentation
-
1. Application
- 1.1. Research Institutions
- 1.2. Pharmaceutical Factory
-
2. Types
- 2.1. On-premises
- 2.2. Cloud Based
LIMS for Pharmaceutical Labs Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
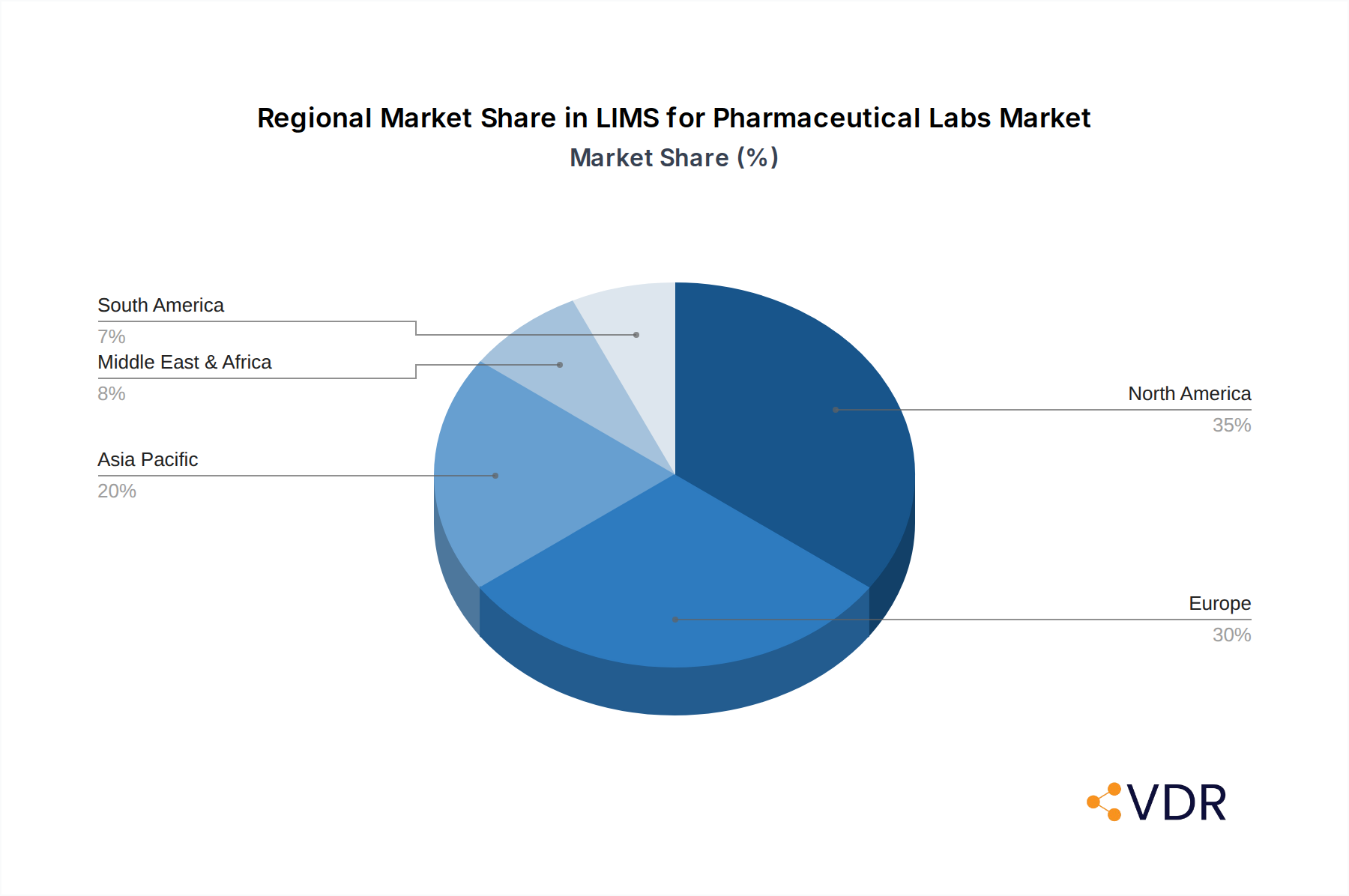
LIMS for Pharmaceutical Labs Regional Market Share

Geographic Coverage of LIMS for Pharmaceutical Labs
LIMS for Pharmaceutical Labs REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global LIMS for Pharmaceutical Labs Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Research Institutions
- 5.1.2. Pharmaceutical Factory
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. On-premises
- 5.2.2. Cloud Based
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America LIMS for Pharmaceutical Labs Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Research Institutions
- 6.1.2. Pharmaceutical Factory
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. On-premises
- 6.2.2. Cloud Based
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America LIMS for Pharmaceutical Labs Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Research Institutions
- 7.1.2. Pharmaceutical Factory
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. On-premises
- 7.2.2. Cloud Based
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe LIMS for Pharmaceutical Labs Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Research Institutions
- 8.1.2. Pharmaceutical Factory
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. On-premises
- 8.2.2. Cloud Based
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa LIMS for Pharmaceutical Labs Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Research Institutions
- 9.1.2. Pharmaceutical Factory
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. On-premises
- 9.2.2. Cloud Based
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific LIMS for Pharmaceutical Labs Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Research Institutions
- 10.1.2. Pharmaceutical Factory
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. On-premises
- 10.2.2. Cloud Based
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Thermo Scientific
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 LabWare
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 LabVantage
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Ocimum Biosolution
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Apex Healthware
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Agilent
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Agaram
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 LabSols
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Benchling
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Autoscribe Informatics
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 AQ Manager
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Eusoft
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Labguru
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Blaze Systems
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Third Wave Analytics
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 GUS LAB
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 LabCollector
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Clinisys
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 Revol LIMS
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 QSI
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.21 ATL
- 11.2.21.1. Overview
- 11.2.21.2. Products
- 11.2.21.3. SWOT Analysis
- 11.2.21.4. Recent Developments
- 11.2.21.5. Financials (Based on Availability)
- 11.2.1 Thermo Scientific
List of Figures
- Figure 1: Global LIMS for Pharmaceutical Labs Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America LIMS for Pharmaceutical Labs Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America LIMS for Pharmaceutical Labs Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America LIMS for Pharmaceutical Labs Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America LIMS for Pharmaceutical Labs Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America LIMS for Pharmaceutical Labs Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America LIMS for Pharmaceutical Labs Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America LIMS for Pharmaceutical Labs Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America LIMS for Pharmaceutical Labs Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America LIMS for Pharmaceutical Labs Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America LIMS for Pharmaceutical Labs Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America LIMS for Pharmaceutical Labs Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America LIMS for Pharmaceutical Labs Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe LIMS for Pharmaceutical Labs Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe LIMS for Pharmaceutical Labs Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe LIMS for Pharmaceutical Labs Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe LIMS for Pharmaceutical Labs Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe LIMS for Pharmaceutical Labs Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe LIMS for Pharmaceutical Labs Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa LIMS for Pharmaceutical Labs Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa LIMS for Pharmaceutical Labs Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa LIMS for Pharmaceutical Labs Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa LIMS for Pharmaceutical Labs Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa LIMS for Pharmaceutical Labs Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa LIMS for Pharmaceutical Labs Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific LIMS for Pharmaceutical Labs Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific LIMS for Pharmaceutical Labs Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific LIMS for Pharmaceutical Labs Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific LIMS for Pharmaceutical Labs Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific LIMS for Pharmaceutical Labs Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific LIMS for Pharmaceutical Labs Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global LIMS for Pharmaceutical Labs Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific LIMS for Pharmaceutical Labs Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the LIMS for Pharmaceutical Labs?
The projected CAGR is approximately 4.9%.
2. Which companies are prominent players in the LIMS for Pharmaceutical Labs?
Key companies in the market include Thermo Scientific, LabWare, LabVantage, Ocimum Biosolution, Apex Healthware, Agilent, Agaram, LabSols, Benchling, Autoscribe Informatics, AQ Manager, Eusoft, Labguru, Blaze Systems, Third Wave Analytics, GUS LAB, LabCollector, Clinisys, Revol LIMS, QSI, ATL.
3. What are the main segments of the LIMS for Pharmaceutical Labs?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "LIMS for Pharmaceutical Labs," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the LIMS for Pharmaceutical Labs report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the LIMS for Pharmaceutical Labs?
To stay informed about further developments, trends, and reports in the LIMS for Pharmaceutical Labs, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
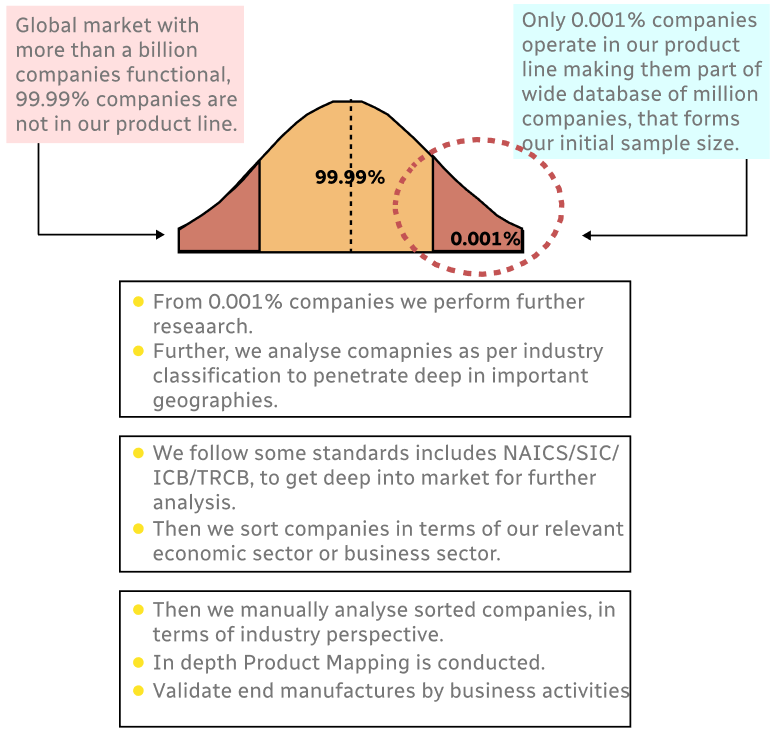
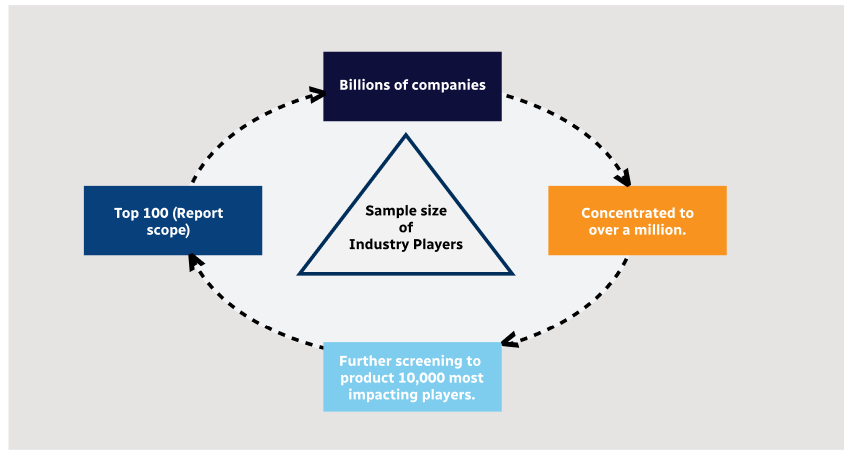
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
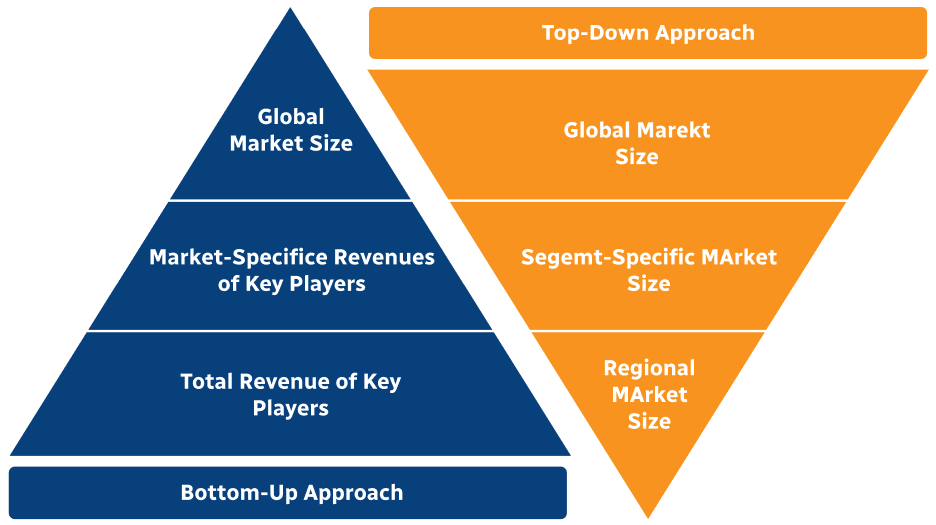
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
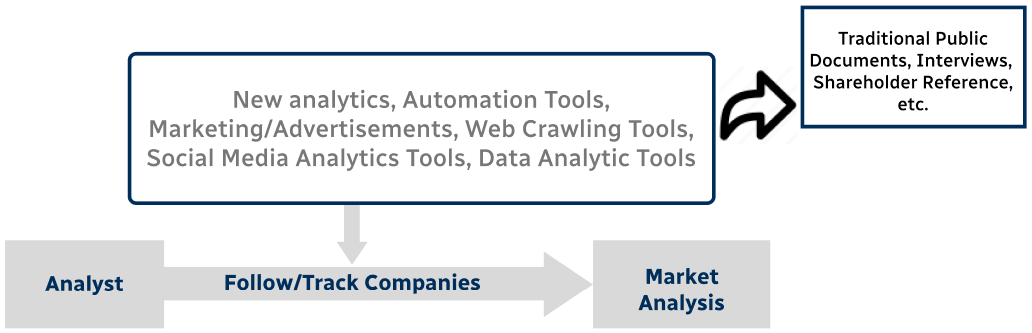
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


