Key Insights
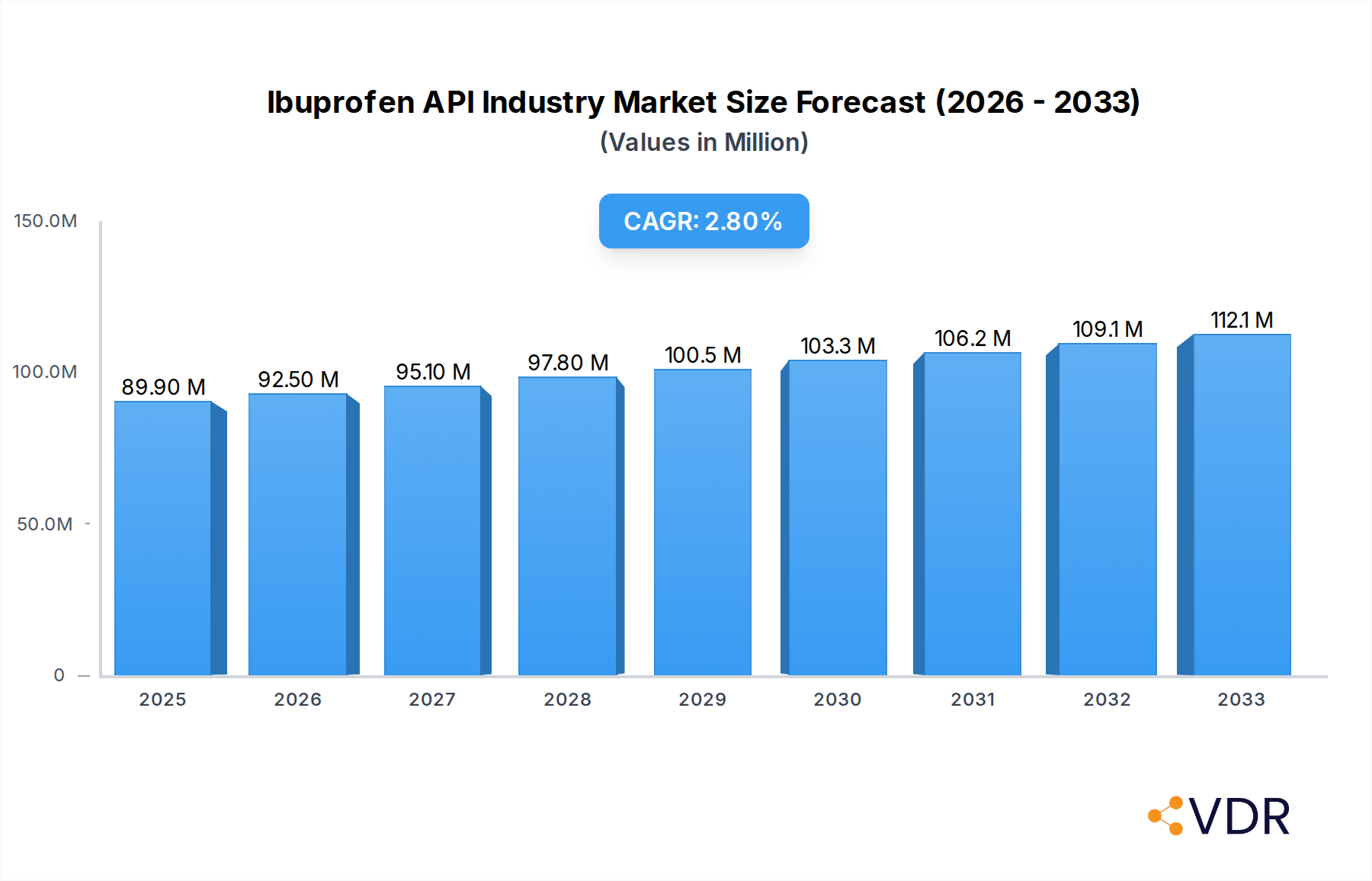
The global Ibuprofen API market is poised for steady growth, projected to reach $89.9 million in 2025, driven by an increasing prevalence of pain-related conditions and a growing demand for effective anti-inflammatory treatments. The market is expected to expand at a Compound Annual Growth Rate (CAGR) of 2.9% from 2025 to 2033, reflecting sustained consumer need and pharmaceutical innovation. Key market drivers include the rising incidence of chronic pain, arthritis, and headaches, all of which necessitate consistent access to reliable ibuprofen Active Pharmaceutical Ingredients (APIs). Furthermore, an aging global population, more susceptible to inflammatory and pain conditions, contributes significantly to this upward trajectory. The pharmaceutical and biopharmaceutical industries, alongside Contract Research Organizations (CROs) and Contract Manufacturing Organizations (CMOs), represent the primary end-users, highlighting the critical role of these sectors in the ibuprofen supply chain. The application segment for headaches is particularly strong, demonstrating a broad consumer base, while arthritis applications underscore the market's importance in managing chronic inflammatory diseases.
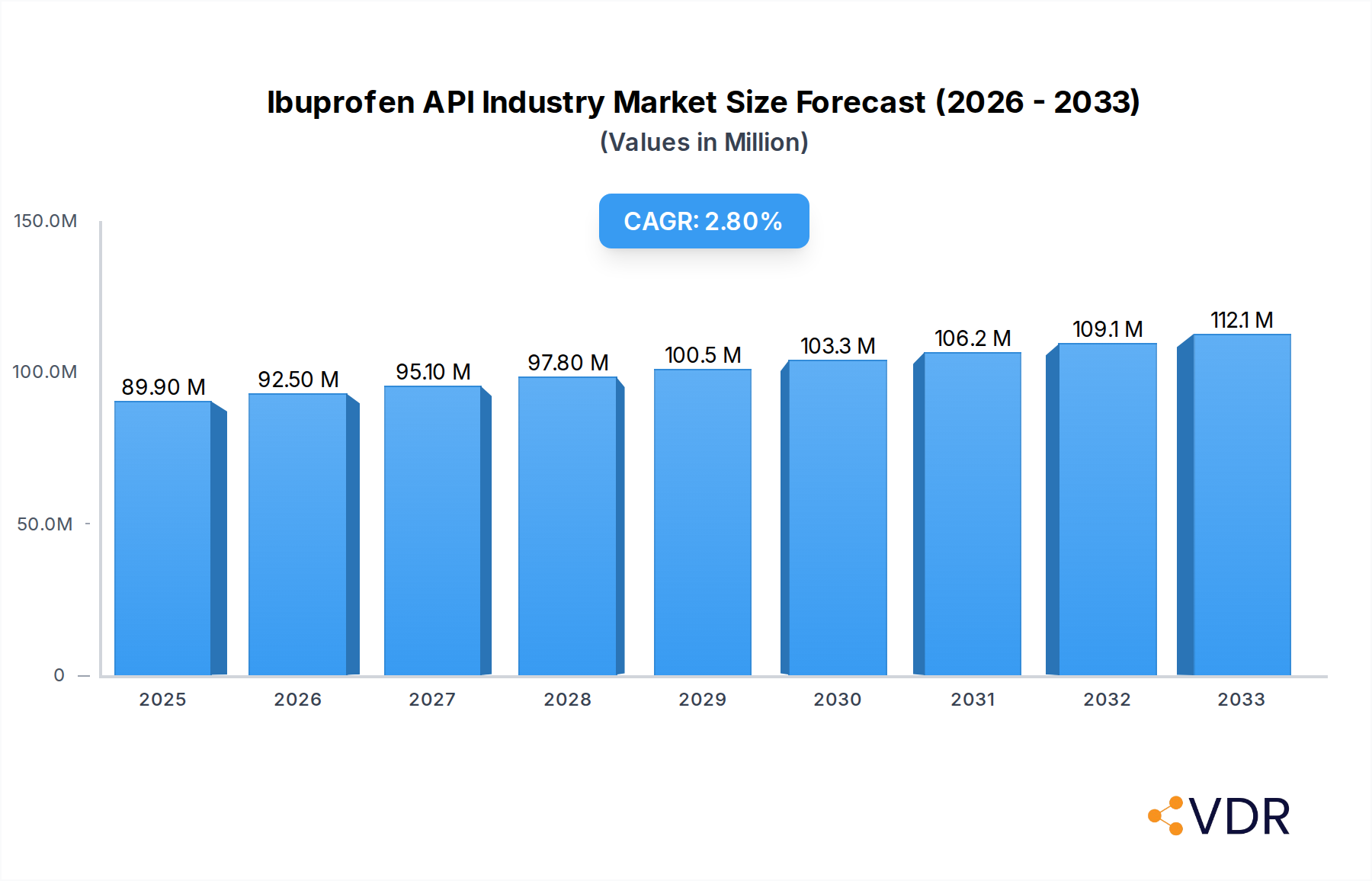
Ibuprofen API Industry Market Size (In Million)

The market landscape is characterized by a competitive environment featuring established players like BASF SE, Dr. Reddy's Laboratories Ltd, and Teva Pharmaceutical Industries Ltd, alongside emerging entities. These companies are actively engaged in research and development to enhance production efficiency and maintain stringent quality standards, crucial for API manufacturing. While market growth is robust, certain restraints such as fluctuating raw material prices and stringent regulatory hurdles for API manufacturing can pose challenges. However, the expanding pharmaceutical manufacturing capabilities in regions like Asia Pacific, coupled with increasing healthcare expenditure globally, are expected to mitigate these restraints. Emerging trends such as the focus on sustainable manufacturing practices and the development of novel drug delivery systems for ibuprofen may also shape the future market dynamics, offering opportunities for innovation and market differentiation. The geographical distribution of the market indicates significant presence and potential in North America and Europe, with Asia Pacific emerging as a key growth region due to its expanding manufacturing base and growing healthcare accessibility.
Ibuprofen API Industry Company Market Share

Ibuprofen API Industry: Market Analysis, Growth Forecast, and Key Player Insights (2019-2033)
This comprehensive report provides an in-depth analysis of the global Ibuprofen Active Pharmaceutical Ingredient (API) market, forecasting its trajectory from 2019 to 2033, with a base and estimated year of 2025. Delving into market dynamics, growth trends, regional dominance, product landscape, key drivers, challenges, opportunities, and pivotal industry players, this report offers actionable intelligence for stakeholders navigating the competitive ibuprofen API landscape. We explore both parent and child market segments, providing granular insights into applications, end-users, and industry developments. All values are presented in million units.
Ibuprofen API Industry Market Dynamics & Structure
The Ibuprofen API market exhibits a moderate to high concentration, with a significant share held by a few leading global manufacturers. Technological innovation remains a crucial driver, focusing on process optimization for cost-efficiency and enhanced purity. Regulatory frameworks, particularly stringent quality control measures mandated by agencies like the EDQM, significantly influence market entry and operational standards. Competitive product substitutes, though present, have a limited impact on the established demand for ibuprofen due to its efficacy and widespread use.
- Market Concentration: Dominated by a mix of large multinational corporations and specialized API manufacturers.
- Technological Innovation Drivers: Process intensification, green chemistry initiatives, and continuous manufacturing technologies.
- Regulatory Frameworks: Strict adherence to Good Manufacturing Practices (GMP), ICH guidelines, and regional pharmacopoeial standards.
- Competitive Product Substitutes: Limited by ibuprofen's established therapeutic profile and cost-effectiveness for its primary applications.
- End-User Demographics: Primarily pharmaceutical and biopharmaceutical companies, with a growing presence of Contract Research Organizations (CROs) and Contract Manufacturing Organizations (CMOs).
- M&A Trends: Strategic acquisitions and partnerships aimed at expanding manufacturing capacity, enhancing R&D capabilities, and securing supply chains. For instance, Lonza's substantial investment in new sites highlights this trend.
Ibuprofen API Industry Growth Trends & Insights
The Ibuprofen API market is poised for steady growth, driven by the escalating prevalence of pain management needs and inflammatory conditions globally. Market size evolution is characterized by consistent demand from established pharmaceutical sectors and emerging markets. Adoption rates for ibuprofen API remain high due to its favorable safety profile and extensive use in over-the-counter (OTC) and prescription medications. Technological disruptions are primarily focused on improving synthesis efficiency and reducing environmental impact, rather than fundamental product innovation. Consumer behavior shifts indicate a growing preference for reliable and affordable pain relief solutions, further bolstering the demand for ibuprofen.
The market penetration of ibuprofen API is substantial, underscoring its role as a cornerstone in pain and inflammation management. The Compound Annual Growth Rate (CAGR) for the forecast period is anticipated to be robust, reflecting the sustained demand from diverse applications. Insights into market evolution reveal a dynamic interplay between generic drug manufacturing and the development of novel drug delivery systems incorporating ibuprofen. Analysis of market size evolution shows a consistent upward trend, supported by increasing healthcare expenditure and an aging global population.
Technological advancements are continuously refining production processes, leading to improved yields and reduced manufacturing costs. This efficiency gain is crucial for maintaining competitive pricing in a market segment with a significant volume of generic products. The adoption rates are further amplified by the expanding healthcare infrastructure in developing economies, increasing access to essential medicines. Consumer behavior, while influenced by factors like brand loyalty for finished dosage forms, ultimately relies on the consistent availability and efficacy of the active ingredient, ibuprofen API.
The market size is projected to reach approximately $XXX million by 2033, up from an estimated $XXX million in 2025. This growth trajectory is supported by a CAGR of approximately X.X% during the forecast period. Key factors contributing to this growth include the rising incidence of chronic pain, the increasing demand for anti-inflammatory drugs, and the growing awareness of the benefits of ibuprofen in various therapeutic areas. Furthermore, the expanding global pharmaceutical industry and the continuous demand for generic drugs are expected to sustain the market's upward momentum. The market penetration remains high, as ibuprofen is a widely prescribed and over-the-counter medication for a broad spectrum of pain and inflammatory conditions.
Dominant Regions, Countries, or Segments in Ibuprofen API Industry
The Arthritis segment, within the Application category, is a primary driver of growth in the global Ibuprofen API market. Its dominance is attributed to the escalating global burden of arthritis, characterized by chronic pain and inflammation, necessitating widespread and continuous use of effective analgesics and anti-inflammatories like ibuprofen.
- North America emerges as a leading region, driven by high per capita healthcare spending, a large aging population prone to inflammatory conditions, and a well-established pharmaceutical manufacturing base.
- Key Drivers: Robust R&D infrastructure, stringent quality standards promoting high-purity API production, and a significant market for both OTC and prescription pain relief medications.
- Market Share: Holds a substantial share of the global ibuprofen API market due to extensive consumption and advanced manufacturing capabilities.
- Growth Potential: Continues to exhibit steady growth, fueled by advancements in pain management therapies and the demand for generics.
- Europe follows closely, with a similar demographic profile and a strong regulatory environment that favors high-quality API production.
- Key Drivers: Favorable reimbursement policies for pain management, advanced healthcare systems, and a consolidated pharmaceutical industry.
- Market Share: A significant contributor to global demand, with a strong presence of API manufacturers adhering to EDQM standards.
- Growth Potential: Stable growth expected, driven by an aging population and the ongoing need for effective arthritis and pain management solutions.
- The Arthritis application segment’s dominance is further solidified by the increasing prevalence of osteoarthritis and rheumatoid arthritis worldwide. Pharmaceutical and Biopharmaceutical Companies are major end-users, consistently procuring ibuprofen API to meet the demand for pain relief formulations targeting these conditions. The economic policies in developed nations often support research and development in pain management, further boosting the segment. Infrastructure for API manufacturing and distribution in regions like North America and Europe is highly developed, ensuring efficient supply chains. The market share for ibuprofen API in the arthritis segment is significant due to its established efficacy and cost-effectiveness compared to newer, more specialized therapies.
Ibuprofen API Industry Product Landscape
The Ibuprofen API product landscape is characterized by high purity and consistent quality, adhering to stringent pharmacopoeial standards. Manufacturers focus on producing ibuprofen in various grades suitable for different pharmaceutical formulations, from solid oral dosage forms to topical applications. Technological advancements in crystallization and purification processes are continuously enhancing product performance metrics, ensuring minimal impurities and optimal bioavailability. The unique selling proposition of ibuprofen API lies in its proven efficacy as a non-steroidal anti-inflammatory drug (NSAID), its broad therapeutic applications, and its cost-effectiveness, making it a staple ingredient for global pharmaceutical manufacturers.
Key Drivers, Barriers & Challenges in Ibuprofen API Industry
Key Drivers:
- Rising Incidence of Pain and Inflammatory Conditions: Increasing global prevalence of arthritis, musculoskeletal disorders, and post-operative pain drives consistent demand.
- Growing Demand for Generic Drugs: Cost-effectiveness and widespread availability of generic ibuprofen formulations fuel API consumption.
- Technological Advancements in API Manufacturing: Process optimization and efficiency improvements lead to reduced production costs and higher yields.
- Increasing Healthcare Expenditure: Global rise in healthcare spending enhances access to pain management solutions.
Barriers & Challenges:
- Stringent Regulatory Compliance: Adherence to evolving quality standards and complex approval processes from regulatory bodies worldwide.
- Raw Material Price Volatility: Fluctuations in the cost of key precursor chemicals can impact profit margins.
- Intense Competition and Price Pressure: The presence of numerous manufacturers leads to competitive pricing and potential margin erosion.
- Supply Chain Disruptions: Geopolitical factors, natural disasters, and pandemics can disrupt the sourcing of raw materials and the distribution of finished APIs.
- Environmental Regulations: Increasing focus on sustainable manufacturing practices and waste reduction presents operational challenges.
Emerging Opportunities in Ibuprofen API Industry
Emerging opportunities in the Ibuprofen API industry lie in the development of novel drug delivery systems that enhance ibuprofen's efficacy, reduce side effects, and improve patient compliance. There is significant potential in exploring combination therapies where ibuprofen is formulated with other active ingredients to target complex pain conditions. Untapped markets in developing economies, with improving healthcare infrastructure and increasing disposable incomes, offer substantial growth prospects. Furthermore, innovations in green chemistry for API synthesis can provide a competitive edge and align with growing environmental consciousness.
Growth Accelerators in the Ibuprofen API Industry Industry
Several catalysts are accelerating long-term growth in the Ibuprofen API industry. Technological breakthroughs in continuous manufacturing processes are enhancing production efficiency and reducing costs, making ibuprofen more accessible globally. Strategic partnerships between API manufacturers and finished dosage form companies are streamlining supply chains and fostering market expansion. Furthermore, ongoing research into ibuprofen's potential therapeutic benefits beyond pain and inflammation, coupled with market expansion strategies targeting underserved populations, are significant growth accelerators.
Key Players Shaping the Ibuprofen API Industry Market
- Solara Active Pharma Sciences Limited
- SI Group Inc
- BASF SE
- Granules India Limited
- Athenex Inc
- Strides Pharma Science Limited
- Octavius Pharma Pvt Ltd
- SX Pharma
- Teva Pharmaceutical Industries Ltd
- Rochem International Inc
- Sino-US Zibo Xinhua-Perrigo Pharmaceutical Co Ltd
- IOL Chemicals and Pharmaceuticals Limited
- Dr Reddy's Laboratories Ltd
Notable Milestones in Ibuprofen API Industry Sector
- November 2022: Solara Active Pharma Sciences Limited's new cutting-edge, multipurpose API manufacturing facility in Vishakhapatnam, Andhra Pradesh, received Certificate of Suitability (CEP) approval for manufacturing ibuprofen API from the European Directorate for the Quality of Medicine (EDQM), enhancing its European market access.
- March 2022: Lonza invested USD 935 million to build sites in Portsmouth, New Hampshire, and Visp, Switzerland, signaling significant investment in API manufacturing capacity and potentially impacting global supply dynamics. The company completed a laboratory expansion project at its API manufacturing facility in Nansha, China, in August 2021, indicating a strategic focus on enhancing R&D and production capabilities in key regions.
In-Depth Ibuprofen API Industry Market Outlook
The outlook for the Ibuprofen API industry remains highly positive, driven by sustained global demand for effective and affordable pain management solutions. Growth accelerators, including advancements in manufacturing technologies like continuous processing and the strategic expansion of production capacities by key players such as Lonza, are poised to further optimize supply chains and reduce costs. Emerging opportunities in untapped markets and the development of innovative drug delivery systems present significant potential for market expansion. Stakeholders can leverage these insights to capitalize on the robust future market potential by focusing on regulatory compliance, cost-effective production, and strategic market penetration.
Ibuprofen API Industry Segmentation
-
1. Application
- 1.1. Headache
- 1.2. Arthritis
- 1.3. Other Applications
-
2. End-user
- 2.1. CROs and CMOs
- 2.2. Pharmaceutical and Biopharmaceutical Companies
Ibuprofen API Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
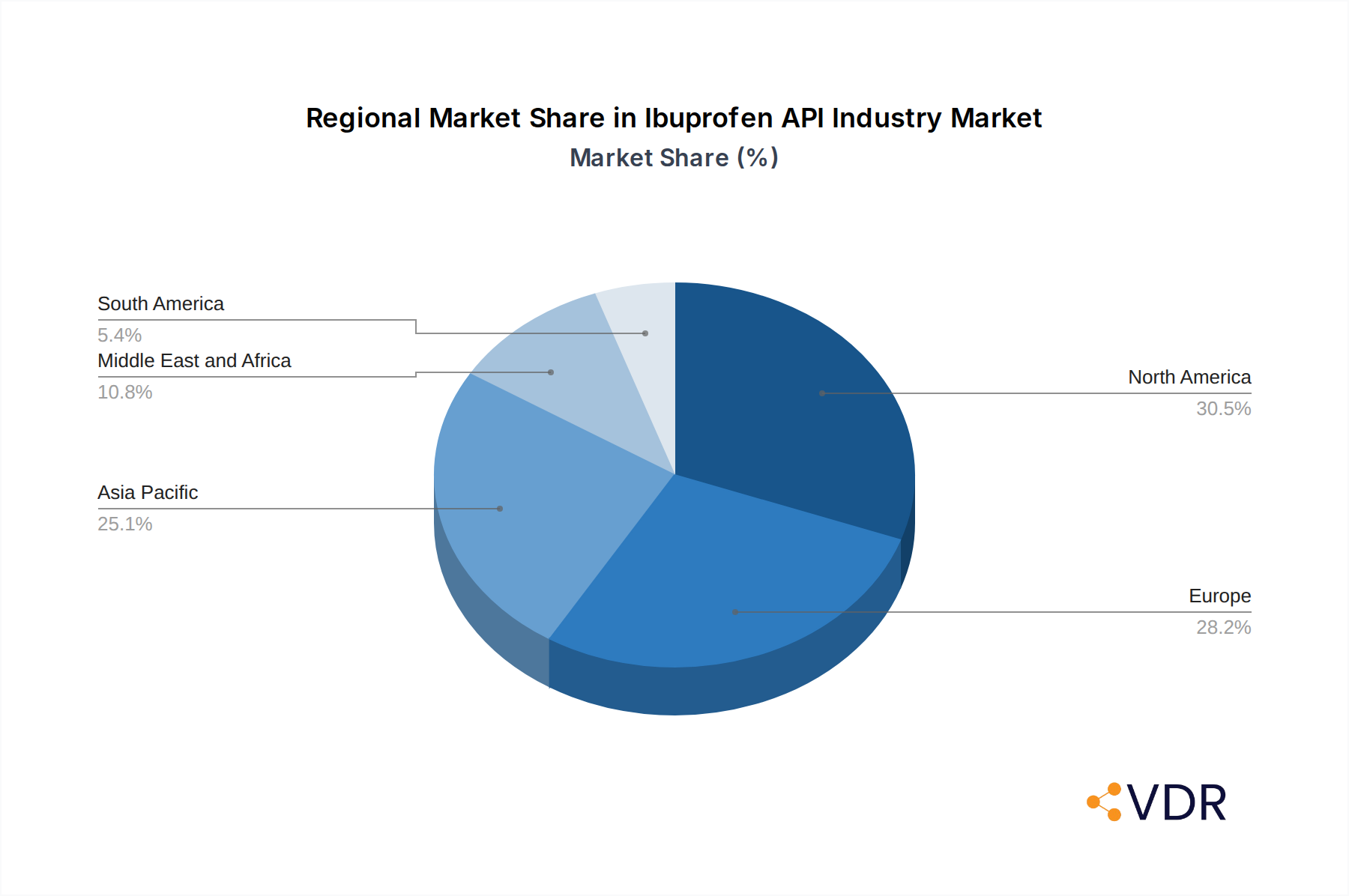
Ibuprofen API Industry Regional Market Share

Geographic Coverage of Ibuprofen API Industry
Ibuprofen API Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 2.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Headache
- 5.1.2. Arthritis
- 5.1.3. Other Applications
- 5.2. Market Analysis, Insights and Forecast - by End-user
- 5.2.1. CROs and CMOs
- 5.2.2. Pharmaceutical and Biopharmaceutical Companies
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East and Africa
- 5.3.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Ibuprofen API Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Headache
- 6.1.2. Arthritis
- 6.1.3. Other Applications
- 6.2. Market Analysis, Insights and Forecast - by End-user
- 6.2.1. CROs and CMOs
- 6.2.2. Pharmaceutical and Biopharmaceutical Companies
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Ibuprofen API Industry Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Headache
- 7.1.2. Arthritis
- 7.1.3. Other Applications
- 7.2. Market Analysis, Insights and Forecast - by End-user
- 7.2.1. CROs and CMOs
- 7.2.2. Pharmaceutical and Biopharmaceutical Companies
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Ibuprofen API Industry Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Headache
- 8.1.2. Arthritis
- 8.1.3. Other Applications
- 8.2. Market Analysis, Insights and Forecast - by End-user
- 8.2.1. CROs and CMOs
- 8.2.2. Pharmaceutical and Biopharmaceutical Companies
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Asia Pacific Ibuprofen API Industry Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Headache
- 9.1.2. Arthritis
- 9.1.3. Other Applications
- 9.2. Market Analysis, Insights and Forecast - by End-user
- 9.2.1. CROs and CMOs
- 9.2.2. Pharmaceutical and Biopharmaceutical Companies
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East and Africa Ibuprofen API Industry Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Headache
- 10.1.2. Arthritis
- 10.1.3. Other Applications
- 10.2. Market Analysis, Insights and Forecast - by End-user
- 10.2.1. CROs and CMOs
- 10.2.2. Pharmaceutical and Biopharmaceutical Companies
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. South America Ibuprofen API Industry Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Headache
- 11.1.2. Arthritis
- 11.1.3. Other Applications
- 11.2. Market Analysis, Insights and Forecast - by End-user
- 11.2.1. CROs and CMOs
- 11.2.2. Pharmaceutical and Biopharmaceutical Companies
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Solara Active Pharma Sciences Limited
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 SI Group Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 BASF SE
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Granules India Limited
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Athenex Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Strides Pharma Science Limited
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Octavius Pharma Pvt Ltd
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 SX Pharma
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Teva Pharmaceutical Industries Ltd*List Not Exhaustive
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Rochem International Inc
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Sino-US Zibo Xinhua-Perrigo Pharmaceutical Co Ltd
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 IOL Chemicals and Pharmaceuticals Limited
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 Dr Reddy's Laboratories Ltd
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.1 Solara Active Pharma Sciences Limited
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Ibuprofen API Industry Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Ibuprofen API Industry Revenue (million), by Application 2025 & 2033
- Figure 3: North America Ibuprofen API Industry Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Ibuprofen API Industry Revenue (million), by End-user 2025 & 2033
- Figure 5: North America Ibuprofen API Industry Revenue Share (%), by End-user 2025 & 2033
- Figure 6: North America Ibuprofen API Industry Revenue (million), by Country 2025 & 2033
- Figure 7: North America Ibuprofen API Industry Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe Ibuprofen API Industry Revenue (million), by Application 2025 & 2033
- Figure 9: Europe Ibuprofen API Industry Revenue Share (%), by Application 2025 & 2033
- Figure 10: Europe Ibuprofen API Industry Revenue (million), by End-user 2025 & 2033
- Figure 11: Europe Ibuprofen API Industry Revenue Share (%), by End-user 2025 & 2033
- Figure 12: Europe Ibuprofen API Industry Revenue (million), by Country 2025 & 2033
- Figure 13: Europe Ibuprofen API Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Pacific Ibuprofen API Industry Revenue (million), by Application 2025 & 2033
- Figure 15: Asia Pacific Ibuprofen API Industry Revenue Share (%), by Application 2025 & 2033
- Figure 16: Asia Pacific Ibuprofen API Industry Revenue (million), by End-user 2025 & 2033
- Figure 17: Asia Pacific Ibuprofen API Industry Revenue Share (%), by End-user 2025 & 2033
- Figure 18: Asia Pacific Ibuprofen API Industry Revenue (million), by Country 2025 & 2033
- Figure 19: Asia Pacific Ibuprofen API Industry Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East and Africa Ibuprofen API Industry Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East and Africa Ibuprofen API Industry Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East and Africa Ibuprofen API Industry Revenue (million), by End-user 2025 & 2033
- Figure 23: Middle East and Africa Ibuprofen API Industry Revenue Share (%), by End-user 2025 & 2033
- Figure 24: Middle East and Africa Ibuprofen API Industry Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East and Africa Ibuprofen API Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Ibuprofen API Industry Revenue (million), by Application 2025 & 2033
- Figure 27: South America Ibuprofen API Industry Revenue Share (%), by Application 2025 & 2033
- Figure 28: South America Ibuprofen API Industry Revenue (million), by End-user 2025 & 2033
- Figure 29: South America Ibuprofen API Industry Revenue Share (%), by End-user 2025 & 2033
- Figure 30: South America Ibuprofen API Industry Revenue (million), by Country 2025 & 2033
- Figure 31: South America Ibuprofen API Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Ibuprofen API Industry Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Ibuprofen API Industry Revenue million Forecast, by End-user 2020 & 2033
- Table 3: Global Ibuprofen API Industry Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Ibuprofen API Industry Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Ibuprofen API Industry Revenue million Forecast, by End-user 2020 & 2033
- Table 6: Global Ibuprofen API Industry Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Ibuprofen API Industry Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Ibuprofen API Industry Revenue million Forecast, by End-user 2020 & 2033
- Table 12: Global Ibuprofen API Industry Revenue million Forecast, by Country 2020 & 2033
- Table 13: Germany Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: United Kingdom Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: France Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Italy Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 17: Spain Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 18: Rest of Europe Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 19: Global Ibuprofen API Industry Revenue million Forecast, by Application 2020 & 2033
- Table 20: Global Ibuprofen API Industry Revenue million Forecast, by End-user 2020 & 2033
- Table 21: Global Ibuprofen API Industry Revenue million Forecast, by Country 2020 & 2033
- Table 22: China Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Japan Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: India Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Australia Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: South Korea Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Asia Pacific Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Ibuprofen API Industry Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Ibuprofen API Industry Revenue million Forecast, by End-user 2020 & 2033
- Table 30: Global Ibuprofen API Industry Revenue million Forecast, by Country 2020 & 2033
- Table 31: GCC Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: South Africa Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: Rest of Middle East and Africa Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: Global Ibuprofen API Industry Revenue million Forecast, by Application 2020 & 2033
- Table 35: Global Ibuprofen API Industry Revenue million Forecast, by End-user 2020 & 2033
- Table 36: Global Ibuprofen API Industry Revenue million Forecast, by Country 2020 & 2033
- Table 37: Brazil Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 38: Argentina Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
- Table 39: Rest of South America Ibuprofen API Industry Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Ibuprofen API Industry?
The projected CAGR is approximately 2.9%.
2. Which companies are prominent players in the Ibuprofen API Industry?
Key companies in the market include Solara Active Pharma Sciences Limited, SI Group Inc, BASF SE, Granules India Limited, Athenex Inc, Strides Pharma Science Limited, Octavius Pharma Pvt Ltd, SX Pharma, Teva Pharmaceutical Industries Ltd*List Not Exhaustive, Rochem International Inc, Sino-US Zibo Xinhua-Perrigo Pharmaceutical Co Ltd, IOL Chemicals and Pharmaceuticals Limited, Dr Reddy's Laboratories Ltd.
3. What are the main segments of the Ibuprofen API Industry?
The market segments include Application, End-user.
4. Can you provide details about the market size?
The market size is estimated to be USD 89.9 million as of 2022.
5. What are some drivers contributing to market growth?
Upsurge in Pharmaceutical and Biopharmaceutical Production; Growing Prevalence of Chronic Disorders.
6. What are the notable trends driving market growth?
Arthritis Holds a Significant Share in the Ibuprofen API Market.
7. Are there any restraints impacting market growth?
Rising Production Costs.
8. Can you provide examples of recent developments in the market?
November 2022: Solara Active Pharma Sciences Limited's new cutting-edge, multipurpose API manufacturing facility in Vishakhapatnam, Andhra Pradesh, received Certificate of Suitability (CEP) approval for manufacturing ibuprofen API from the European Directorate for the Quality of Medicine (EDQM).
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Ibuprofen API Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Ibuprofen API Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Ibuprofen API Industry?
To stay informed about further developments, trends, and reports in the Ibuprofen API Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
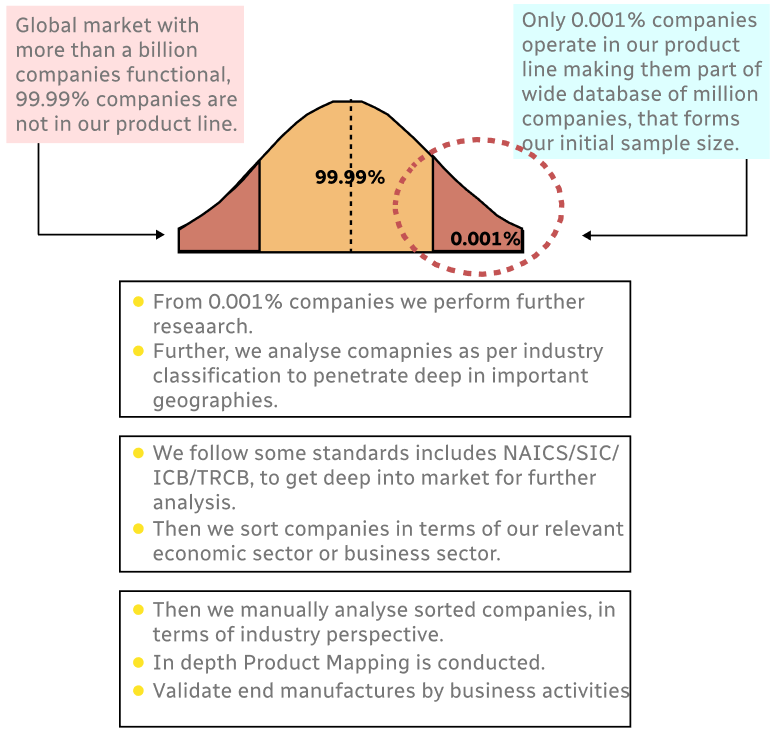
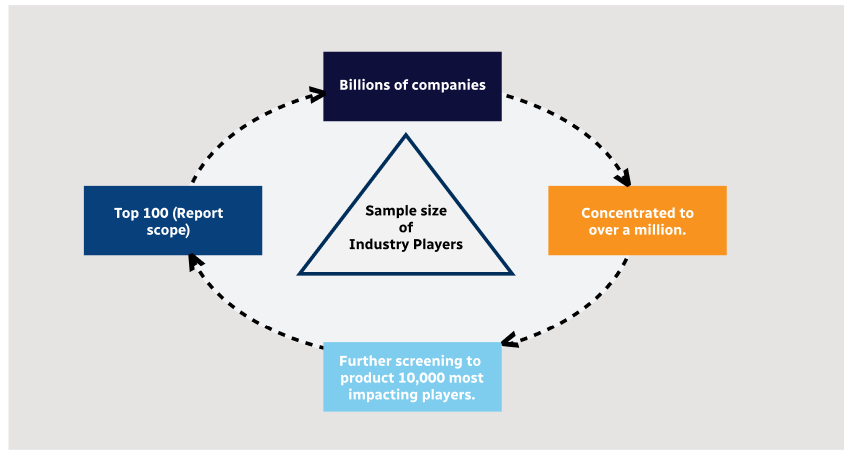
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
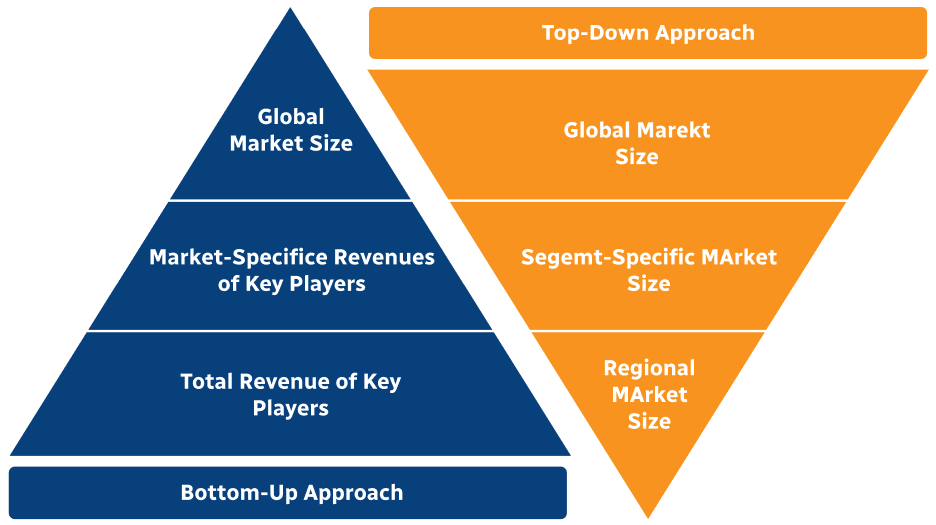
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


