Key Insights
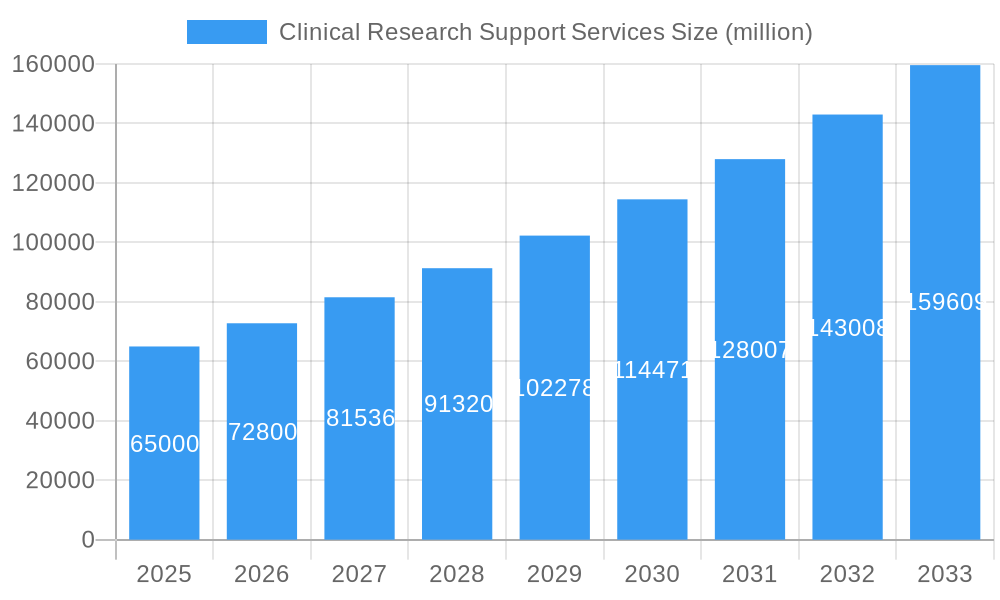
The global Clinical Research Support Services market is projected for substantial growth, estimated to reach $25.62 billion by 2033, with a Compound Annual Growth Rate (CAGR) of 7.93% from a base year of 2025. This expansion is driven by escalating demand for efficient clinical trials, spurred by the increasing prevalence of chronic diseases and continuous innovation in pharmaceuticals and biotechnology. Key growth factors include the outsourcing of clinical trial activities by life sciences companies seeking specialized expertise and cost-effectiveness, alongside advancements in data management technologies and the growing complexity of trial protocols.

Clinical Research Support Services Market Size (In Billion)

The market is segmented by application and type. Pharmaceutical and Biopharmaceutical Companies dominate the application segment due to their significant investment in drug development. Within types, Clinical Trial Site Management and Patient Recruitment Management are critical components impacting trial timelines and success. Emerging trends such as AI/ML integration, decentralized clinical trials, and real-world data utilization are reshaping the market. However, stringent regulatory requirements, high trial costs, and the availability of skilled talent present challenges. Despite these, ongoing healthcare R&D investment and the global pursuit of novel therapeutics will sustain a dynamic market for clinical research support services.
Clinical Research Support Services Company Market Share

Clinical Research Support Services Market: Dynamics, Trends, and Future Outlook (2019-2033)
This comprehensive report provides an in-depth analysis of the global Clinical Research Support Services market, offering critical insights for stakeholders navigating this dynamic sector. With a study period spanning from 2019 to 2033 and a base year of 2025, this report leverages historical data and robust forecasting to deliver actionable intelligence. We delve into market concentration, technological drivers, regulatory landscapes, competitive forces, end-user demographics, and M&A activities. The analysis extends to market size evolution, adoption rates, disruptive technologies, and shifting consumer behaviors, utilizing detailed metrics and projections. Furthermore, the report identifies dominant regions, countries, and key market segments (Application: Pharmaceutical and Biopharmaceutical Companies, Medical Device Companies, Others; Types: Clinical Trial Site Management, Patient Recruitment Management, Data Management, Administrative Staff, Others), pinpointing growth drivers and potential. Product innovations, performance metrics, and unique selling propositions are detailed, alongside a thorough examination of key drivers, barriers, challenges, and emerging opportunities. Finally, we present a list of pivotal players, notable milestones, and a future market outlook designed to guide strategic decision-making in this ever-evolving industry.
Clinical Research Support Services Market Dynamics & Structure
The Clinical Research Support Services market exhibits a moderately concentrated structure, with several large, established Contract Research Organizations (CROs) holding significant market share. The industry is driven by a confluence of technological innovations, stringent regulatory frameworks, and the increasing complexity of clinical trials. Key technological drivers include advancements in data analytics, artificial intelligence (AI) for trial design and patient recruitment, and decentralized clinical trial technologies. Regulatory frameworks, such as those established by the FDA and EMA, while creating barriers to entry, also foster a demand for specialized support services to ensure compliance. Competitive product substitutes are limited, given the highly specialized nature of these services, though in-house capabilities of larger pharmaceutical companies can be considered a form of substitution. End-user demographics are dominated by pharmaceutical and biopharmaceutical companies, followed by medical device manufacturers and academic institutions. Mergers and acquisitions (M&A) are a significant trend, aimed at expanding service portfolios, geographical reach, and technological capabilities. For instance, approximately 15-20% of market participants have been involved in M&A activities in the past two years, with deal values ranging from tens to hundreds of millions of dollars. Innovation barriers include high R&D costs, lengthy regulatory approval processes, and the need for specialized expertise.
- Market Concentration: Moderately concentrated with leading CROs dominating.
- Technological Innovation Drivers: AI in trial design, decentralized trial technologies, advanced data analytics.
- Regulatory Frameworks: FDA, EMA – driving compliance-focused service demand.
- Competitive Product Substitutes: Limited, with in-house capabilities being a minor factor.
- End-User Demographics: Pharmaceutical & Biopharmaceutical Companies (approx. 65% of demand), Medical Device Companies (approx. 25%), Others (approx. 10%).
- M&A Trends: Significant consolidation, with an estimated 15-20% of market participants engaging in M&A annually.
- Innovation Barriers: High R&D investment, regulatory hurdles, specialized talent acquisition.
Clinical Research Support Services Growth Trends & Insights
The global Clinical Research Support Services market is poised for robust growth, driven by an escalating demand for efficient and effective clinical trial execution. The market size, estimated to be approximately $75,000 million in 2025, is projected to expand at a Compound Annual Growth Rate (CAGR) of approximately 7.5% during the forecast period of 2025–2033, reaching an estimated $130,000 million by 2033. Adoption rates for specialized support services are accelerating as pharmaceutical and biopharmaceutical companies increasingly outsource non-core activities to focus on drug discovery and development. Technological disruptions are playing a pivotal role, with the integration of artificial intelligence and machine learning transforming trial design, patient recruitment, and data analysis. AI-powered platforms are enhancing predictive capabilities, identifying suitable patient cohorts more efficiently, and reducing trial timelines. The rise of decentralized clinical trials (DCTs) is another significant trend, enabling remote patient monitoring and data collection, thereby expanding access to a wider patient population and reducing geographical barriers. Consumer behavior shifts are also influencing market dynamics, with an increasing emphasis on patient-centricity in clinical trials. Patients are seeking more convenient participation options, which DCTs and sophisticated patient recruitment management services are facilitating.
Furthermore, the growing complexity of drug development pipelines, particularly in areas like oncology and rare diseases, necessitates specialized expertise and infrastructure that many research organizations may not possess in-house. This outsourcing trend is a key growth accelerator. The increasing volume of clinical trial data also demands advanced data management and analysis capabilities, a service segment that is experiencing substantial growth. Regulatory bodies are also evolving, with a greater emphasis on real-world evidence (RWE) integration, which requires specialized data handling and analysis support. The market penetration of advanced clinical trial site management solutions is increasing, as sponsors aim to optimize site selection, performance monitoring, and operational efficiency. The shift towards value-based healthcare and a greater focus on reducing the cost of drug development are also pushing for more efficient and outcome-driven clinical research support services. The increasing prevalence of chronic diseases globally is also a foundational driver for a continuous pipeline of new drug development, thereby sustaining the demand for clinical research support.
Dominant Regions, Countries, or Segments in Clinical Research Support Services
The Pharmaceutical and Biopharmaceutical Companies segment, falling under the Application category, is the undeniable leader and primary growth driver within the global Clinical Research Support Services market. This segment is projected to hold over 70% of the market share in 2025, valued at approximately $52,500 million. The dominance of this segment is attributed to the continuous and substantial investment in research and development by these entities to bring novel therapeutics to market. The high costs and inherent risks associated with drug development necessitate a strategic reliance on external expertise for efficient and compliant clinical trial execution. Countries with a robust pharmaceutical industry, such as the United States and European nations (particularly Germany, the UK, and Switzerland), are leading the charge. The United States alone accounts for an estimated 40% of the global market share in clinical research support services, driven by its large biopharmaceutical sector, advanced healthcare infrastructure, and supportive regulatory environment that encourages innovation.
Within the Types segment, Clinical Trial Site Management and Data Management are emerging as the most influential sub-segments. Clinical Trial Site Management is crucial for ensuring the smooth operation of clinical trials at investigator sites, encompassing patient recruitment, data collection, and regulatory compliance. This segment is valued at an estimated $20,000 million in 2025. Data Management, valued at approximately $18,000 million in 2025, is critical for the integrity and analysis of clinical trial data, with increasing demand for sophisticated data handling, statistical analysis, and data visualization. The growth in these segments is propelled by economic policies that foster innovation and investment in the life sciences sector, alongside the development of advanced healthcare infrastructure that supports complex trial protocols. For instance, government incentives for R&D in regions like North America and Europe significantly boost demand for these services.
- Dominant Application Segment: Pharmaceutical and Biopharmaceutical Companies (approx. 70% market share, $52,500 million in 2025).
- Key Drivers: High R&D investment, need for specialized expertise, outsourcing trends.
- Dominant Regions/Countries:
- United States: Leading market with approx. 40% market share, driven by a strong biopharmaceutical sector and supportive regulations.
- Europe (Germany, UK, Switzerland): Significant market share due to established pharmaceutical industries and advanced healthcare systems.
- Influential Types Segments:
- Clinical Trial Site Management: (approx. $20,000 million in 2025) Essential for operational efficiency and compliance at research sites.
- Data Management: (approx. $18,000 million in 2025) Critical for data integrity, analysis, and regulatory submissions.
- Growth Factors: Favorable economic policies, advanced healthcare infrastructure, increasing R&D focus.
Clinical Research Support Services Product Landscape
The product landscape of Clinical Research Support Services is characterized by a sophisticated array of integrated solutions designed to streamline every stage of clinical trial execution. These services range from comprehensive clinical trial site management platforms that optimize investigator site selection, activation, and performance monitoring, to advanced patient recruitment management tools leveraging AI and data analytics to identify and enroll eligible participants faster. Data management solutions are paramount, offering robust electronic data capture (EDC) systems, data cleansing, statistical analysis, and regulatory-compliant data archiving. Administrative staff support, encompassing project management, regulatory affairs coordination, and medical writing, further complements these offerings. Innovations are continuously being introduced, focusing on enhancing efficiency, data accuracy, and patient engagement. For example, novel AI algorithms are being integrated into data management systems for real-time anomaly detection, and patient-centric technologies are being developed to improve adherence and reduce dropout rates. The unique selling proposition often lies in the seamless integration of these services, offering a one-stop solution for sponsors.
Key Drivers, Barriers & Challenges in Clinical Research Support Services
The Clinical Research Support Services market is propelled by several key drivers. The increasing complexity and cost of drug development necessitate outsourcing to specialized CROs, optimizing efficiency and reducing time-to-market. Technological advancements, particularly in data analytics and AI, are transforming trial design and execution. The growing prevalence of chronic diseases and the demand for personalized medicine fuel the pipeline of new drug development, thus sustaining demand for research support. Furthermore, expanding global clinical trial operations and the need for diverse patient populations in trials are creating significant opportunities.
However, several barriers and challenges exist. Stringent and evolving regulatory requirements across different geographies can be complex and costly to navigate, posing a significant hurdle. The high cost of implementing advanced technologies and the need for specialized skilled personnel can also be restrictive. Supply chain issues, though less prevalent for services, can impact the availability of essential resources at trial sites. Intense competition among established CROs and emerging players can lead to pricing pressures. Data privacy and security concerns are paramount, requiring robust measures to protect sensitive patient information and maintain compliance with regulations like GDPR and HIPAA.
Emerging Opportunities in Clinical Research Support Services
Emerging opportunities in the Clinical Research Support Services market are largely centered around leveraging new technologies and adapting to evolving healthcare landscapes. The growing adoption of decentralized clinical trials (DCTs) presents a significant avenue, requiring specialized support for remote monitoring, digital data capture, and patient engagement platforms. The increasing focus on real-world evidence (RWE) generation is creating demand for services that can collect, analyze, and interpret RWE from various sources, including electronic health records and wearable devices. Furthermore, the burgeoning field of cell and gene therapies, with their unique logistical and scientific complexities, offers a niche but high-growth opportunity for specialized support services. The development of AI-driven solutions for predictive analytics in patient recruitment and trial success prediction also presents a fertile ground for innovation and market expansion.
Growth Accelerators in the Clinical Research Support Services Industry
Several catalysts are accelerating long-term growth in the Clinical Research Support Services industry. Technological breakthroughs, such as the widespread integration of artificial intelligence and machine learning for enhanced trial design, patient stratification, and predictive modeling, are significantly improving efficiency and reducing development timelines. Strategic partnerships between CROs, technology providers, and academic institutions are fostering innovation and expanding service offerings, allowing for more comprehensive solutions. Market expansion strategies, including targeting emerging markets with growing healthcare infrastructure and increasing R&D investments, are opening up new revenue streams. Furthermore, the increasing trend of pharmaceutical and biopharmaceutical companies to focus on core competencies, leading to higher outsourcing rates for clinical trial management, is a fundamental growth accelerator. The development of standardized platforms for interoperability and data sharing is also streamlining operations across the industry.
Key Players Shaping the Clinical Research Support Services Market
- Charles River Laboratories
- IQVIA
- Pharmaceutical Product Development
- WuXi AppTec
- Parexel International
- Icon PLC
- Laboratory Corp. of America Holdings
- Novotech
- Kaiser Permanente
- eukemia & Lymphoma Society
- Clinical Trial Service BV
- The Leukemia & Lymphoma Society
Notable Milestones in Clinical Research Support Services Sector
- 2019: Launch of advanced AI-powered patient recruitment platforms by several leading CROs, improving identification of eligible participants.
- 2020: Increased adoption of decentralized clinical trial (DCT) technologies and remote monitoring solutions in response to the global pandemic.
- 2021: Significant M&A activity, with major CROs acquiring smaller, specialized technology firms to enhance data analytics and digital capabilities.
- 2022: Emergence of dedicated support services for cell and gene therapy trials, catering to their unique logistical and regulatory needs.
- 2023: Growing emphasis on real-world evidence (RWE) integration, with CROs expanding services for RWE data collection and analysis.
- 2024: Increased investment in cybersecurity measures and data privacy solutions by CROs to comply with stringent regulations and protect sensitive patient data.
In-Depth Clinical Research Support Services Market Outlook
The future of the Clinical Research Support Services market is exceptionally promising, driven by ongoing innovation and increasing reliance on specialized outsourcing. Growth accelerators such as the pervasive adoption of AI for predictive analytics in trial design and patient recruitment, coupled with the expansion of decentralized clinical trial models, will continue to reshape operational efficiencies. Strategic partnerships will foster integrated service offerings, while a focus on emerging markets and novel therapeutic areas like cell and gene therapies will unlock new avenues for growth. The market is set to experience sustained expansion as life science companies prioritize their core competencies, further driving demand for comprehensive and technologically advanced clinical research support.
Clinical Research Support Services Segmentation
-
1. Application
- 1.1. Pharmaceutical and Biopharmaceutical Companies
- 1.2. Medical Device Companies
- 1.3. Others
-
2. Types
- 2.1. Clinical Trial Site Management
- 2.2. Patient Recruitment Management
- 2.3. Data Management
- 2.4. Administrative Staff
- 2.5. Others
Clinical Research Support Services Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
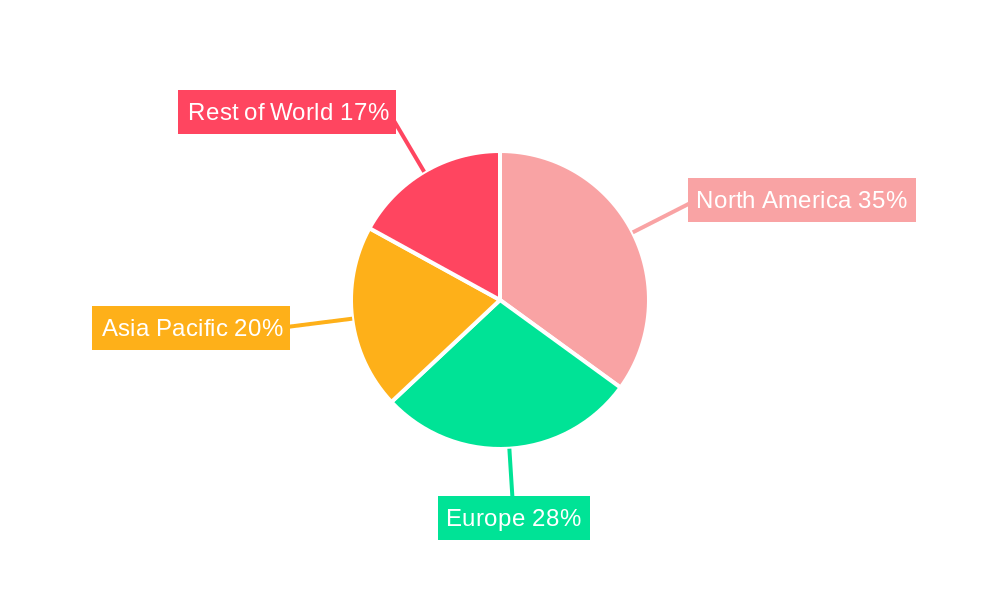
Clinical Research Support Services Regional Market Share

Geographic Coverage of Clinical Research Support Services
Clinical Research Support Services REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.93% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Clinical Research Support Services Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Pharmaceutical and Biopharmaceutical Companies
- 5.1.2. Medical Device Companies
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Clinical Trial Site Management
- 5.2.2. Patient Recruitment Management
- 5.2.3. Data Management
- 5.2.4. Administrative Staff
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Clinical Research Support Services Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Pharmaceutical and Biopharmaceutical Companies
- 6.1.2. Medical Device Companies
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Clinical Trial Site Management
- 6.2.2. Patient Recruitment Management
- 6.2.3. Data Management
- 6.2.4. Administrative Staff
- 6.2.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Clinical Research Support Services Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Pharmaceutical and Biopharmaceutical Companies
- 7.1.2. Medical Device Companies
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Clinical Trial Site Management
- 7.2.2. Patient Recruitment Management
- 7.2.3. Data Management
- 7.2.4. Administrative Staff
- 7.2.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Clinical Research Support Services Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Pharmaceutical and Biopharmaceutical Companies
- 8.1.2. Medical Device Companies
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Clinical Trial Site Management
- 8.2.2. Patient Recruitment Management
- 8.2.3. Data Management
- 8.2.4. Administrative Staff
- 8.2.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Clinical Research Support Services Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Pharmaceutical and Biopharmaceutical Companies
- 9.1.2. Medical Device Companies
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Clinical Trial Site Management
- 9.2.2. Patient Recruitment Management
- 9.2.3. Data Management
- 9.2.4. Administrative Staff
- 9.2.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Clinical Research Support Services Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Pharmaceutical and Biopharmaceutical Companies
- 10.1.2. Medical Device Companies
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Clinical Trial Site Management
- 10.2.2. Patient Recruitment Management
- 10.2.3. Data Management
- 10.2.4. Administrative Staff
- 10.2.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Charles River Laboratories
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 IQVIA
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Pharmaceutical Product Development
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 WuXi AppTec
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Parexel International
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Icon PLC
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Laboratory Corp. of America Holdings
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Novotech
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Kaiser Permanente
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 eukemia & Lymphoma Society
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Clinical Trial Service BV
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 The Leukemia & Lymphoma Society
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.1 Charles River Laboratories
List of Figures
- Figure 1: Global Clinical Research Support Services Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Clinical Research Support Services Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Clinical Research Support Services Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Clinical Research Support Services Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Clinical Research Support Services Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Clinical Research Support Services Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Clinical Research Support Services Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Clinical Research Support Services Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Clinical Research Support Services Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Clinical Research Support Services Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Clinical Research Support Services Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Clinical Research Support Services Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Clinical Research Support Services Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Clinical Research Support Services Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Clinical Research Support Services Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Clinical Research Support Services Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Clinical Research Support Services Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Clinical Research Support Services Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Clinical Research Support Services Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Clinical Research Support Services Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Clinical Research Support Services Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Clinical Research Support Services Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Clinical Research Support Services Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Clinical Research Support Services Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Clinical Research Support Services Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Clinical Research Support Services Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Clinical Research Support Services Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Clinical Research Support Services Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Clinical Research Support Services Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Clinical Research Support Services Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Clinical Research Support Services Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Clinical Research Support Services Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Clinical Research Support Services Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Clinical Research Support Services Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Clinical Research Support Services Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Clinical Research Support Services Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Clinical Research Support Services Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Clinical Research Support Services Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Clinical Research Support Services Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Clinical Research Support Services Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Clinical Research Support Services Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Clinical Research Support Services Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Clinical Research Support Services Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Clinical Research Support Services Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Clinical Research Support Services Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Clinical Research Support Services Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Clinical Research Support Services Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Clinical Research Support Services Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Clinical Research Support Services Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Clinical Research Support Services Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Clinical Research Support Services?
The projected CAGR is approximately 7.93%.
2. Which companies are prominent players in the Clinical Research Support Services?
Key companies in the market include Charles River Laboratories, IQVIA, Pharmaceutical Product Development, WuXi AppTec, Parexel International, Icon PLC, Laboratory Corp. of America Holdings, Novotech, Kaiser Permanente, eukemia & Lymphoma Society, Clinical Trial Service BV, The Leukemia & Lymphoma Society.
3. What are the main segments of the Clinical Research Support Services?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 25.62 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Clinical Research Support Services," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Clinical Research Support Services report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Clinical Research Support Services?
To stay informed about further developments, trends, and reports in the Clinical Research Support Services, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database


Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


