Key Insights
The Viral Packaging Services market is poised for robust expansion, projected to reach an estimated $1.68 billion in 2025. This impressive growth is fueled by a CAGR of 15.3%, indicating a dynamic and rapidly evolving sector. The increasing demand for advanced gene and cell therapies, particularly for treating complex diseases like cancer and genetic disorders, is a primary driver. The fundamental research segment is experiencing significant traction as scientists explore novel therapeutic avenues, while the clinical trial segment is escalating due to the growing number of drug candidates entering advanced testing phases. Innovations in viral vector technology, including improved efficiency and safety profiles for AAV, adenovirus, and lentivirus packaging, are also critical enablers of this market surge.
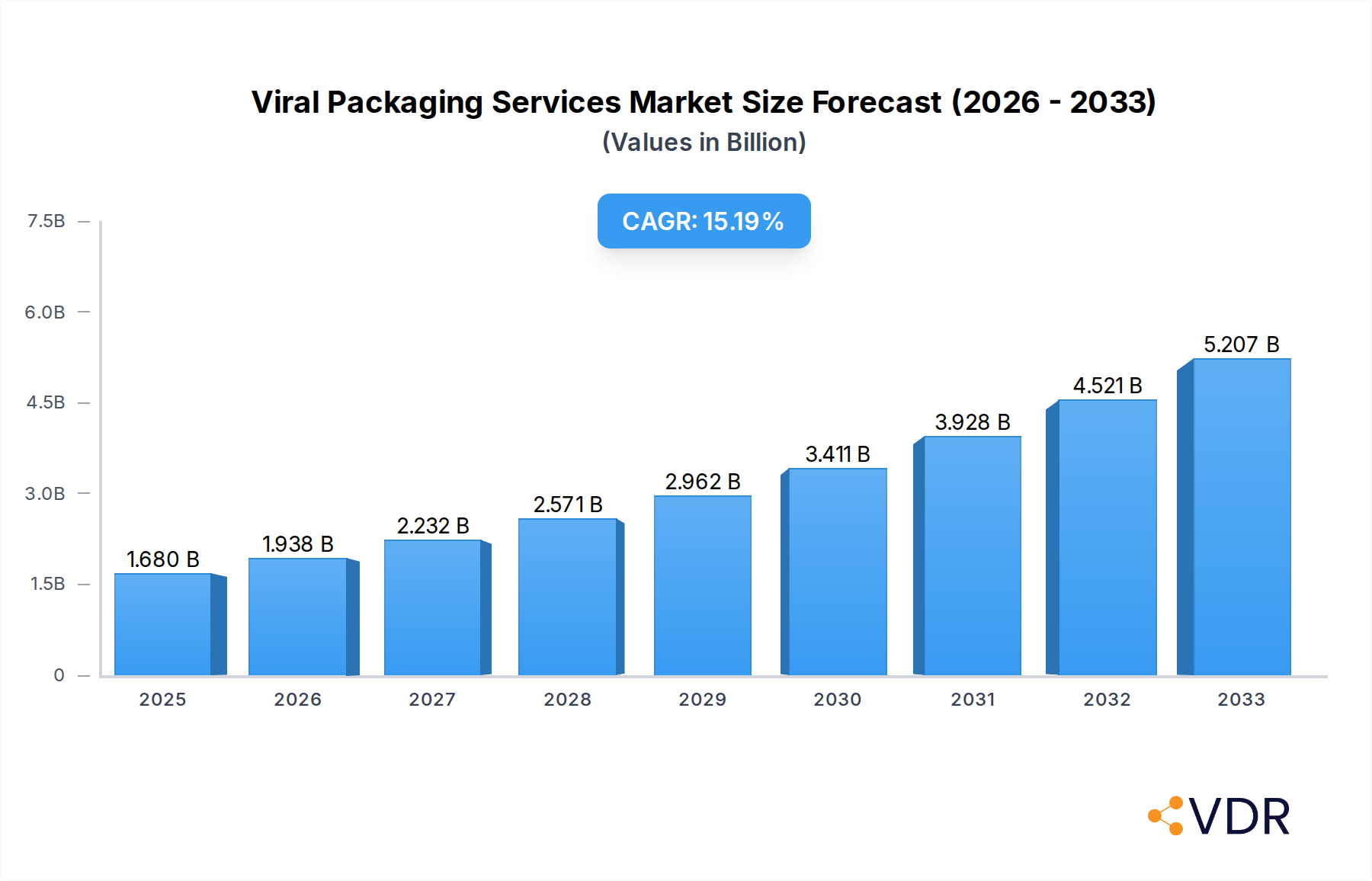
Viral Packaging Services Market Size (In Billion)

Emerging trends such as the rise of personalized medicine and the growing outsourcing of viral vector production to specialized Contract Development and Manufacturing Organizations (CDMOs) are further shaping the market landscape. Key players like Takara Bio, VectorBuilder, and Vigene Biosciences are actively investing in research and development to enhance their service offerings and expand their manufacturing capacities. While the market demonstrates immense potential, certain restraints, such as stringent regulatory hurdles and the high cost associated with manufacturing viral vectors, need to be addressed to ensure sustained and inclusive growth. Geographically, North America and Europe are expected to lead in market share due to established biopharmaceutical industries and substantial R&D investments. However, the Asia Pacific region is anticipated to exhibit the highest growth rate, driven by increasing government support for biotech research and a growing pool of skilled professionals.
Viral Packaging Services Company Market Share

Here's the SEO-optimized report description for Viral Packaging Services, meticulously crafted for maximum visibility and engagement without any placeholder modifications.
Viral Packaging Services Market Dynamics & Structure
The global viral packaging services market is characterized by dynamic forces shaping its trajectory. A moderate market concentration is observed, with key players like Takara Bio, Cell Biolabs, Applied Biological Materials, GeneCopoeia, Vigene Biosciences, VectorBuilder, HANBIO, OBiO Technology (Shanghai) Corp, and Yiming Cell holding significant, yet fragmented, market shares. Technological innovation is the primary driver, with continuous advancements in gene delivery vectors and purification techniques propelling the market forward. Regulatory frameworks, particularly those governing the use of viral vectors in gene therapies and clinical trials, play a crucial role in defining market entry and operational standards. Competitive product substitutes, while emerging, are yet to significantly challenge the dominance of established viral vector types such as AAV, Adenovirus, and Lentivirus packaging. End-user demographics are increasingly skewed towards academic and research institutions for fundamental research applications, alongside a growing demand from pharmaceutical and biotechnology companies for clinical trial phases. Mergers and acquisitions (M&A) are a notable trend, driven by the need for consolidation, expanded service offerings, and enhanced manufacturing capabilities, particularly in the parent market of biologics manufacturing, which is projected to reach $XXX billion units by 2033.
- Market Concentration: Moderate, with a mix of established large players and specialized niche providers.
- Technological Innovation Drivers: Enhanced vector design, improved production yields, downstream processing innovations, and quality control advancements.
- Regulatory Frameworks: Strict guidelines for preclinical and clinical applications, impacting service development and market access.
- Competitive Product Substitutes: Emerging non-viral delivery systems and alternative gene editing technologies present long-term competition.
- End-User Demographics: Growth driven by academic research, pharmaceutical R&D for gene therapy, and contract research organizations (CROs).
- M&A Trends: Strategic acquisitions aimed at vertical integration, expanding service portfolios, and increasing production capacity.
Viral Packaging Services Growth Trends & Insights
The global viral packaging services market is poised for substantial expansion, driven by the burgeoning field of gene and cell therapy. The market size, estimated at $X.XX billion units in 2025, is projected to witness a robust Compound Annual Growth Rate (CAGR) of XX.X% during the forecast period of 2025–2033. This impressive growth is underpinned by a rising number of gene therapy clinical trials and the increasing approval of gene therapy products, which directly translate into a higher demand for reliable and scalable viral vector production. Adoption rates for specialized viral packaging services are accelerating as research institutions and biopharmaceutical companies increasingly outsource this complex and resource-intensive process to specialized providers. Technological disruptions, including the development of novel viral vector serotypes with improved tropism and reduced immunogenicity, are further stimulating market growth. Consumer behavior shifts are evident, with a growing preference for end-to-end solutions, encompassing vector design, production, and quality control, from a single, trusted provider. The parent market for biologics, valued at approximately $XXX billion units in 2025, acts as a significant upstream indicator of growth for its specialized segments like viral packaging. The child market segments, focusing on specific therapeutic areas utilizing viral vectors, are experiencing rapid penetration as new indications are explored.
- Market Size Evolution: From an estimated $X.XX billion units in 2025, projected to reach $XX.XX billion units by 2033.
- CAGR: XX.X% during the forecast period (2025–2033).
- Adoption Rates: Increasing outsourcing of viral vector production by R&D entities due to complexity and specialization.
- Technological Disruptions: Development of novel viral vector platforms, including designer AAV capsids and advanced lentiviral systems.
- Consumer Behavior Shifts: Demand for integrated services, from plasmid to GMP-grade viral vectors, and a focus on regulatory compliance.
- Market Penetration: Deepening penetration into preclinical and clinical gene therapy development pipelines.
- Upstream Influence: The parent market of biologics manufacturing, valued at $XXX billion units in 2025, provides a strong foundation.
Dominant Regions, Countries, or Segments in Viral Packaging Services
North America, particularly the United States, currently dominates the global viral packaging services market. This regional supremacy is driven by a confluence of factors, including a robust and well-funded research ecosystem, a high concentration of leading biopharmaceutical companies, and a progressive regulatory environment that facilitates the advancement of gene therapies. The Application segment of Clinical Trial applications is a significant growth engine within this region, owing to the substantial number of gene therapy clinical trials being conducted, many of which are in advanced phases. Within the Types segment, AAV Packaging services are experiencing unparalleled demand, reflecting the widespread use of Adeno-Associated Viruses (AAV) as a preferred vector for gene delivery due to its safety profile and broad tropism. The market size in North America is estimated to be $X.XX billion units in 2025, capturing approximately XX% of the global market share. Key drivers include substantial government funding for biomedical research, significant venture capital investments in biotech startups, and the presence of academic institutions at the forefront of gene therapy innovation.
- Dominant Region: North America (primarily the United States).
- Leading Application Segment: Clinical Trial.
- Leading Type Segment: AAV Packaging.
- Market Share (North America): Approximately XX% of the global market in 2025.
- Market Size (North America): Estimated at $X.XX billion units in 2025.
- Key Drivers in North America:
- Extensive gene therapy clinical trial pipeline.
- Strong presence of leading gene therapy developers and Contract Development and Manufacturing Organizations (CDMOs).
- Government initiatives and funding for gene therapy research.
- Advanced technological infrastructure and skilled workforce.
- Growth Potential: Continued innovation in AAV vector engineering and expansion of therapeutic targets will sustain its dominance.
Viral Packaging Services Product Landscape
The product landscape of viral packaging services is characterized by a diverse array of vector types and customized solutions designed to meet the specific needs of gene therapy and research applications. Key offerings include AAV packaging, renowned for its versatility and broad application in treating genetic disorders; Adenovirus packaging, favored for its high transduction efficiency in certain cell types; and Lentivirus packaging, critical for stable gene integration into the host genome. Innovations are continuously emerging, focusing on developing novel capsid designs for improved tissue specificity and reduced immunogenicity, as well as optimizing production processes to achieve higher titers and purity. Performance metrics are rigorously assessed, encompassing vector titer, infectivity, purity, and batch-to-batch consistency, all crucial for downstream therapeutic efficacy and safety. Unique selling propositions often lie in the ability to provide GMP-grade viral vectors for clinical use, alongside specialized services for preclinical research.
Key Drivers, Barriers & Challenges in Viral Packaging Services
Key Drivers: The viral packaging services market is propelled by the exponential growth in gene and cell therapy research, the increasing number of clinical trials, and the rising demand for outsourced manufacturing solutions from biopharmaceutical companies. Technological advancements in vector design and production efficiency are also significant growth accelerators.
Barriers & Challenges: Key challenges include stringent and evolving regulatory requirements for viral vector production and use, the high cost associated with establishing and maintaining GMP-compliant manufacturing facilities, and the need for specialized expertise in viral vector engineering and production. Supply chain complexities for raw materials and the potential for immunogenicity of viral vectors also present significant hurdles. The competitive landscape, with established players and emerging CDMOs, intensifies pricing pressures.
Emerging Opportunities in Viral Packaging Services
Emerging opportunities in the viral packaging services sector are abundant, driven by the expanding therapeutic applications of gene therapies. The development of novel viral vector serotypes with enhanced targeting capabilities and reduced immunogenicity presents a significant avenue for innovation. Untapped markets include the application of viral vectors for non-coding RNA delivery and the expansion into rare disease indications with high unmet medical needs. Evolving consumer preferences for more personalized and precisely targeted gene therapies will also fuel demand for highly customized viral packaging solutions. The increasing use of viral vectors in areas beyond rare genetic disorders, such as oncology and infectious diseases, further broadens the market's potential.
Growth Accelerators in the Viral Packaging Services Industry
Several catalysts are accelerating the long-term growth of the viral packaging services industry. Technological breakthroughs in areas like CRISPR-based gene editing, which often rely on viral vectors for delivery, are expanding the utility of viral packaging. Strategic partnerships between academic institutions and biopharmaceutical companies, as well as collaborations between CDMOs and drug developers, are crucial for streamlining the development and commercialization of gene therapies. Market expansion strategies, including geographic diversification and the development of integrated service offerings from research-grade to commercial-grade production, are also key growth drivers. The increasing focus on rare diseases and the potential for one-time curative treatments are major forces driving sustained demand.
Key Players Shaping the Viral Packaging Services Market
- Takara Bio
- Cell Biolabs
- Applied Biological Materials
- GeneCopoeia
- Vigene Biosciences
- VectorBuilder
- HANBIO
- OBiO Technology (Shanghai) Corp
- Yiming Cell
Notable Milestones in Viral Packaging Services Sector
- 2019: Increased regulatory approvals for AAV-based gene therapies, driving demand for AAV packaging services.
- 2020: Expansion of GMP manufacturing capacity by several key players to meet rising clinical demand.
- 2021: Advancements in high-throughput screening of viral vector variants for improved efficiency.
- 2022: Growing interest in non-integrating lentiviral vectors for improved safety profiles.
- 2023: Strategic acquisitions and partnerships focused on expanding service portfolios to include end-to-end gene therapy manufacturing.
- 2024: Increased focus on developing cost-effective and scalable production methods for viral vectors.
In-Depth Viral Packaging Services Market Outlook
The future market outlook for viral packaging services is exceptionally promising, fueled by ongoing breakthroughs in gene therapy and a robust pipeline of clinical candidates. The continuous development of novel viral vector platforms, alongside advancements in manufacturing technologies, will be pivotal in unlocking new therapeutic possibilities and increasing accessibility. Strategic collaborations and the expansion of integrated service offerings are anticipated to play a crucial role in meeting the growing demand for both research-grade and GMP-grade viral vectors. The market is set to witness sustained growth, driven by the potential of gene therapies to address a wide range of debilitating diseases, solidifying viral packaging services as an indispensable component of modern medicine.
Viral Packaging Services Segmentation
-
1. Application
- 1.1. Fundamental Research
- 1.2. Clinical Trial
- 1.3. Others
-
2. Types
- 2.1. AAV Packaging
- 2.2. Adenovirus Packaging
- 2.3. Lentivirus Packaging
Viral Packaging Services Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
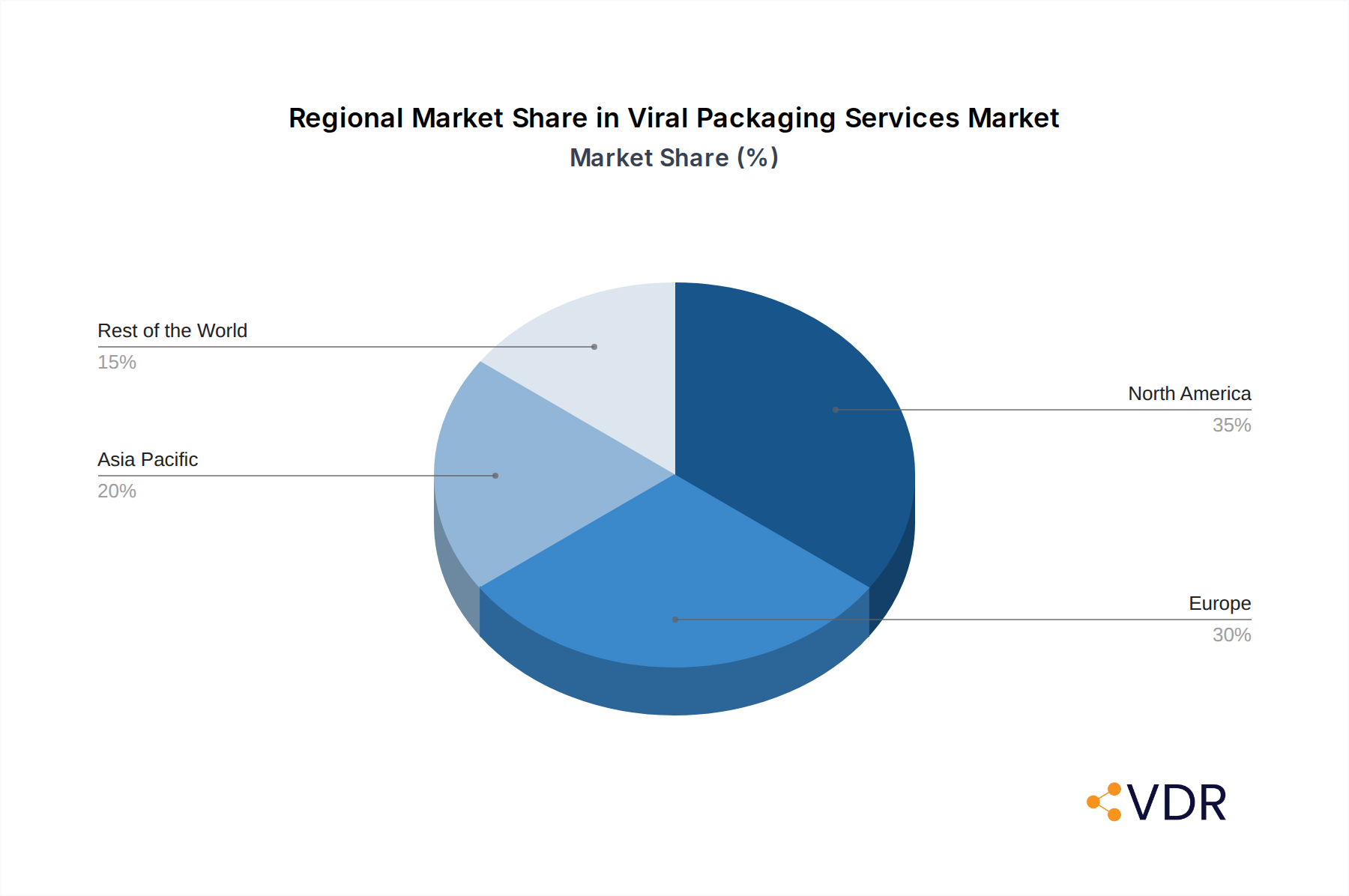
Viral Packaging Services Regional Market Share

Geographic Coverage of Viral Packaging Services
Viral Packaging Services REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 15.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Viral Packaging Services Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Fundamental Research
- 5.1.2. Clinical Trial
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. AAV Packaging
- 5.2.2. Adenovirus Packaging
- 5.2.3. Lentivirus Packaging
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Viral Packaging Services Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Fundamental Research
- 6.1.2. Clinical Trial
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. AAV Packaging
- 6.2.2. Adenovirus Packaging
- 6.2.3. Lentivirus Packaging
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Viral Packaging Services Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Fundamental Research
- 7.1.2. Clinical Trial
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. AAV Packaging
- 7.2.2. Adenovirus Packaging
- 7.2.3. Lentivirus Packaging
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Viral Packaging Services Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Fundamental Research
- 8.1.2. Clinical Trial
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. AAV Packaging
- 8.2.2. Adenovirus Packaging
- 8.2.3. Lentivirus Packaging
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Viral Packaging Services Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Fundamental Research
- 9.1.2. Clinical Trial
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. AAV Packaging
- 9.2.2. Adenovirus Packaging
- 9.2.3. Lentivirus Packaging
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Viral Packaging Services Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Fundamental Research
- 10.1.2. Clinical Trial
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. AAV Packaging
- 10.2.2. Adenovirus Packaging
- 10.2.3. Lentivirus Packaging
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Takara Bio
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Cell Biolabs
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Applied Biological Materials
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 GeneCopoeia
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Vigene Biosciences
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 VectorBuilder
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 HANBIO
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 OBiO Technology (Shanghai) Corp
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Yiming Cell
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.1 Takara Bio
List of Figures
- Figure 1: Global Viral Packaging Services Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Viral Packaging Services Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Viral Packaging Services Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Viral Packaging Services Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Viral Packaging Services Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Viral Packaging Services Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Viral Packaging Services Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Viral Packaging Services Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Viral Packaging Services Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Viral Packaging Services Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Viral Packaging Services Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Viral Packaging Services Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Viral Packaging Services Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Viral Packaging Services Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Viral Packaging Services Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Viral Packaging Services Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Viral Packaging Services Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Viral Packaging Services Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Viral Packaging Services Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Viral Packaging Services Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Viral Packaging Services Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Viral Packaging Services Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Viral Packaging Services Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Viral Packaging Services Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Viral Packaging Services Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Viral Packaging Services Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Viral Packaging Services Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Viral Packaging Services Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Viral Packaging Services Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Viral Packaging Services Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Viral Packaging Services Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Viral Packaging Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Viral Packaging Services Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Viral Packaging Services Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Viral Packaging Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Viral Packaging Services Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Viral Packaging Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Viral Packaging Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Viral Packaging Services Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Viral Packaging Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Viral Packaging Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Viral Packaging Services Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Viral Packaging Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Viral Packaging Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Viral Packaging Services Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Viral Packaging Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Viral Packaging Services Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Viral Packaging Services Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Viral Packaging Services Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Viral Packaging Services Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Viral Packaging Services?
The projected CAGR is approximately 15.3%.
2. Which companies are prominent players in the Viral Packaging Services?
Key companies in the market include Takara Bio, Cell Biolabs, Applied Biological Materials, GeneCopoeia, Vigene Biosciences, VectorBuilder, HANBIO, OBiO Technology (Shanghai) Corp, Yiming Cell.
3. What are the main segments of the Viral Packaging Services?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Viral Packaging Services," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Viral Packaging Services report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Viral Packaging Services?
To stay informed about further developments, trends, and reports in the Viral Packaging Services, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


