Key Insights
The global market for sterile cell culture inserts is poised for significant expansion, projected to reach a market size of USD 154 million in 2025 and grow at a robust Compound Annual Growth Rate (CAGR) of 6.4% through 2033. This sustained growth is propelled by an increasing reliance on advanced cell-based assays for drug discovery and development, alongside a growing demand for more accurate and reproducible preclinical research. Key drivers include the burgeoning biopharmaceutical sector, advancements in cell culture technologies enabling more sophisticated research, and the rising prevalence of chronic diseases requiring novel therapeutic interventions. Furthermore, academic and research institutions are increasingly adopting sterile cell culture inserts for fundamental biological studies, contributing to market expansion. The market is characterized by a diverse range of applications, from diagnostic companies and laboratories to pharmaceutical factories and academic settings, highlighting its broad utility across the life sciences spectrum.
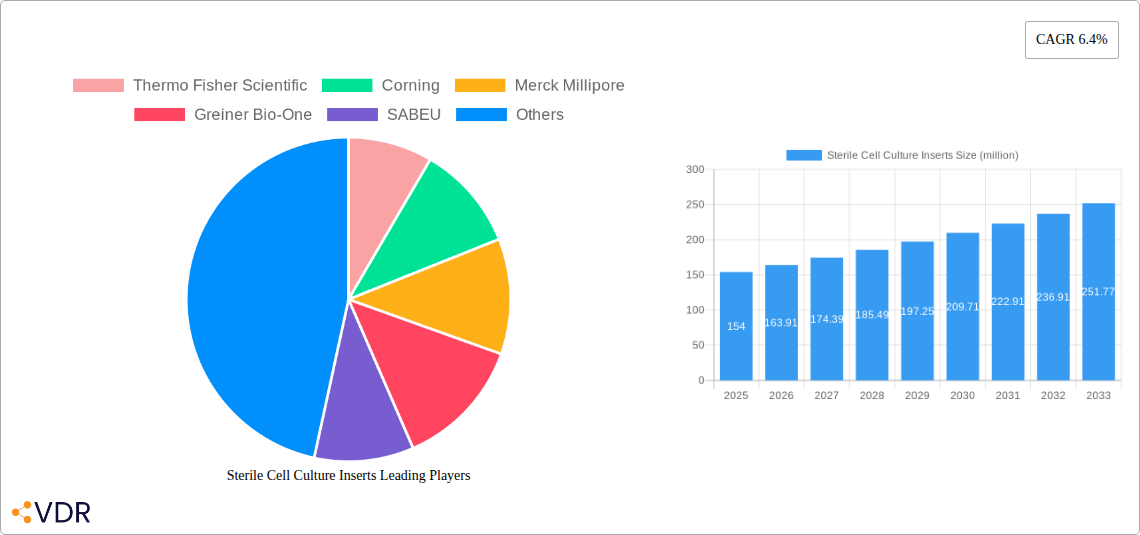
Sterile Cell Culture Inserts Market Size (In Million)

The market segments for sterile cell culture inserts are primarily defined by their material composition and application. Dominant types include PTFE Membrane, Mixed Cellulose Esters Membrane, and Polycarbonate Membrane, each offering unique properties suited to different cell culture protocols. Diagnostic companies and laboratories represent a substantial application segment due to the critical need for reliable cell culture in diagnostic test development and quality control. Pharmaceutical factories are leveraging these inserts for in-vitro drug efficacy and toxicity testing, streamlining the drug development pipeline. The competitive landscape features prominent players such as Thermo Fisher Scientific, Corning, and Merck Millipore, alongside specialized manufacturers like Ibidi GmbH and MatTek Corporation, fostering innovation and a broad product portfolio. Regional analysis indicates that North America and Europe are currently leading the market, driven by established biopharmaceutical industries and significant R&D investments. However, the Asia Pacific region is expected to witness the fastest growth, fueled by increasing government support for biotechnology and a growing number of research facilities. Restraints such as the high cost of advanced materials and the need for stringent quality control measures are being navigated by continuous technological advancements and increasing awareness of the long-term benefits of precision cell culture.
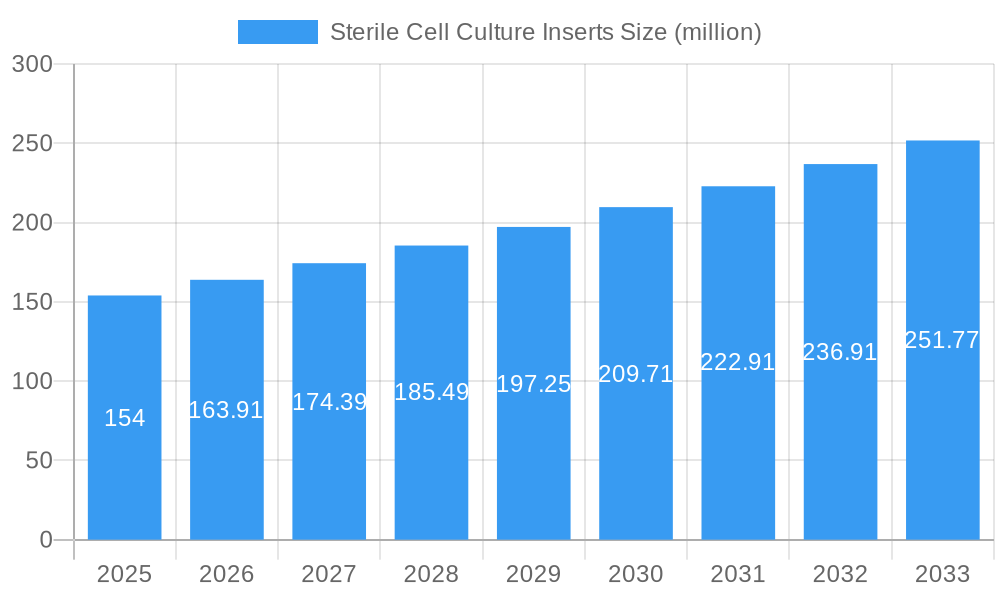
Sterile Cell Culture Inserts Company Market Share

Sterile Cell Culture Inserts Market: Comprehensive Report 2019-2033
This in-depth report provides a thorough analysis of the global Sterile Cell Culture Inserts market, offering unparalleled insights into its dynamics, growth trajectory, regional dominance, product landscape, and key players. Covering the historical period from 2019 to 2024, the base year of 2025, and a forecast period extending to 2033, this report is your definitive guide to understanding the current state and future potential of this vital segment within the life sciences industry.
Sterile Cell Culture Inserts Market Dynamics & Structure
The Sterile Cell Culture Inserts market exhibits a moderately concentrated structure, with leading companies such as Thermo Fisher Scientific, Corning, and Merck Millipore holding significant market shares. Technological innovation is a primary driver, with continuous advancements in membrane materials and pore sizes enhancing cell viability and functionality. Regulatory frameworks, particularly those governing pharmaceutical and diagnostic applications, play a crucial role in shaping market access and product development. Competitive product substitutes include traditional petri dishes and flask-based cultures, though inserts offer distinct advantages in specialized applications. End-user demographics span Diagnostic Companies and Laboratories, Pharmaceutical Factories, and Academic and Research Institutes, each with unique demands. Mergers and acquisitions (M&A) activity, averaging approximately 2-3 significant deals annually between 2019-2024, reflects a trend towards consolidation and market expansion. Key innovation barriers include the high cost of advanced material development and the stringent validation processes required for clinical applications.
- Market Concentration: Dominated by a few key players, with Thermo Fisher Scientific and Corning leading.
- Technological Innovation Drivers: Advancements in porous materials and sterile manufacturing processes.
- Regulatory Frameworks: FDA and EMA guidelines significantly impact product approvals.
- Competitive Product Substitutes: Flasks, multi-well plates, and spheroid culture technologies.
- End-User Demographics: Growing demand from pharmaceutical R&D and diagnostics.
- M&A Trends: Consistent activity for market share expansion and technology acquisition.
- Innovation Barriers: High R&D costs and lengthy regulatory approval pathways.
Sterile Cell Culture Inserts Growth Trends & Insights
The global Sterile Cell Culture Inserts market has witnessed robust growth, projected to expand at a Compound Annual Growth Rate (CAGR) of approximately 7.5% from 2025 to 2033. This expansion is underpinned by escalating demand from the pharmaceutical and biotechnology sectors for advanced cell culture solutions that mimic in vivo environments. Increased investment in drug discovery and development, particularly in areas like regenerative medicine, immunotherapy, and cancer research, directly fuels the adoption of sterile cell culture inserts. Furthermore, the growing prevalence of chronic diseases and infectious outbreaks necessitates faster and more reliable diagnostic testing, driving demand in diagnostic laboratories. Technological disruptions, such as the development of novel biocompatible membrane materials and the integration of inserts into automated high-throughput screening systems, are further accelerating market penetration. Consumer behavior shifts are also evident, with researchers increasingly prioritizing cell culture systems that offer superior cell-to-cell interaction and physiological relevance, capabilities readily provided by advanced insert technologies. The market size, estimated at USD 450 million in 2025, is anticipated to reach USD 800 million by 2033. This growth is attributed to the increasing complexity of biological research, the need for precise control over cellular microenvironments, and the drive for more accurate in vitro models that reduce reliance on animal testing. The adoption rates are expected to rise as awareness of the benefits of using sterile cell culture inserts for complex cell assays, organoid development, and drug screening becomes more widespread across research institutions and pharmaceutical companies.
Dominant Regions, Countries, or Segments in Sterile Cell Culture Inserts
North America currently dominates the global Sterile Cell Culture Inserts market, driven by its well-established pharmaceutical and biotechnology industries, extensive academic research infrastructure, and substantial government funding for life sciences research. The United States, in particular, accounts for a significant portion of regional market share due to the presence of numerous leading pharmaceutical companies, contract research organizations (CROs), and a high concentration of academic and research institutions actively engaged in cell-based research and development.
Within the Application segment, Academic and Research Institutes represent the largest and fastest-growing segment. This dominance is fueled by continuous advancements in scientific understanding, the exploration of novel therapeutic targets, and the fundamental need for precise cell culture tools to support groundbreaking discoveries. The relentless pursuit of knowledge in fields like oncology, neuroscience, immunology, and regenerative medicine necessitates the use of sterile cell culture inserts for complex assays, co-culture studies, and the development of advanced cell-based models.
Regarding Type, PTFE Membrane inserts are leading the market. Their superior biocompatibility, chemical inertness, and controlled porosity make them ideal for a wide range of cell culture applications, including cell migration assays, barrier function studies, and the creation of complex 3D cell cultures. The ability of PTFE membranes to facilitate nutrient exchange while preventing cell passage is critical for mimicking physiological environments and achieving reproducible research outcomes.
- Dominant Region: North America, led by the United States.
- Key Drivers: Strong pharmaceutical R&D, robust academic funding, advanced healthcare infrastructure.
- Market Share: Estimated at 40% of the global market in 2025.
- Growth Potential: Continual innovation in biotechnology and increasing demand for cell-based therapies.
- Dominant Application Segment: Academic and Research Institutes.
- Key Drivers: Fundamental research needs, drug discovery efforts, focus on complex cell models.
- Market Share: Accounts for approximately 35% of the market in 2025.
- Growth Potential: Expanding fields like immunotherapy and gene editing.
- Dominant Type: PTFE Membrane.
- Key Drivers: Superior biocompatibility and controlled porosity for advanced cell assays.
- Market Share: Estimated at 45% of the total insert types.
- Growth Potential: Increasing demand for precise cell microenvironment control.
Sterile Cell Culture Inserts Product Landscape
The Sterile Cell Culture Inserts product landscape is characterized by continuous innovation aimed at enhancing cell viability, facilitating complex co-culture models, and enabling sophisticated assay development. Manufacturers are focusing on optimizing membrane pore sizes, surface treatments, and material compositions to achieve better cell adhesion, growth, and differentiation. Key applications include cell migration and invasion assays, barrier function studies, drug screening, spheroid formation, and the creation of 3D cellular models that better mimic in vivo tissue architecture. Performance metrics emphasize high cell recovery rates, minimal non-specific binding, and consistent pore uniformity, crucial for reproducible experimental results. Unique selling propositions often revolve around specific membrane technologies, such as ultra-thin membranes for enhanced diffusion or specialized coatings that promote specific cell behaviors.
Key Drivers, Barriers & Challenges in Sterile Cell Culture Inserts
Key Drivers: The primary forces propelling the Sterile Cell Culture Inserts market include the escalating demand for advanced cell-based assays in drug discovery and development, the increasing prevalence of complex diseases requiring sophisticated research models, and the growing emphasis on in vitro methodologies to reduce animal testing. Technological advancements in membrane materials and manufacturing processes, leading to enhanced cell functionality and experimental accuracy, are also significant drivers.
Key Barriers & Challenges: Key challenges facing the market include the high cost of advanced sterile cell culture inserts, which can limit adoption in resource-constrained research settings. Stringent regulatory requirements for use in pharmaceutical development and diagnostics can also pose hurdles. Supply chain disruptions for specialized raw materials, and competitive pressures from alternative cell culture technologies, such as organ-on-a-chip systems, present ongoing challenges. The need for rigorous validation and optimization for specific cell types and experimental protocols also adds complexity.
Emerging Opportunities in Sterile Cell Culture Inserts
Emerging opportunities in the Sterile Cell Culture Inserts market lie in the development of custom-designed inserts for niche applications, such as organoid engineering and tissue regeneration research. The increasing adoption of high-throughput screening platforms presents an opportunity for inserts compatible with automation. Furthermore, the growing global focus on personalized medicine and the development of cell-specific therapies will drive demand for inserts that can precisely control cellular microenvironments and facilitate patient-derived cell culture. Untapped markets in emerging economies with expanding research capacities also represent significant growth potential.
Growth Accelerators in the Sterile Cell Culture Inserts Industry
Technological breakthroughs in biomaterials science, leading to novel membrane properties and biocompatible coatings, are significant growth accelerators. Strategic partnerships between insert manufacturers and reagent suppliers, or between insert providers and automation companies, foster market expansion and wider adoption. Market expansion strategies, including targeting emerging research areas like synthetic biology and the increasing demand for in vitro toxicology testing, will also accelerate growth. The continuous refinement of sterile manufacturing processes to reduce costs and improve scalability will further enhance market penetration across diverse research segments.
Key Players Shaping the Sterile Cell Culture Inserts Market
- Thermo Fisher Scientific
- Corning
- Merck Millipore
- Greiner Bio-One
- SABEU
- Ibidi GmbH
- Eppendorf
- Sarstedt
- Oxyphen (Filtration Group)
- Celltreat Scientific Products
- HiMedia Laboratories
- MatTek Corporation
- BRAND GMBH + CO KG
- Wuxi NEST BIOTECHNOLOGY
- SAINING
Notable Milestones in Sterile Cell Culture Inserts Sector
- 2019: Launch of new porous membrane materials with enhanced hydrophilicity for improved cell attachment.
- 2020: Increased demand for inserts in COVID-19 research, particularly for viral propagation and vaccine development assays.
- 2021: Introduction of customizable insert formats for advanced organoid culture.
- 2022: Significant M&A activity as larger players sought to expand their cell culture portfolio.
- 2023: Development of ultra-thin membrane inserts for faster nutrient diffusion and drug penetration studies.
- 2024: Focus on sustainable manufacturing practices and biodegradable membrane options.
In-Depth Sterile Cell Culture Inserts Market Outlook
The future outlook for the Sterile Cell Culture Inserts market is exceptionally positive, driven by the sustained growth in life sciences research and the expanding applications of cell-based technologies. Key growth accelerators include ongoing technological innovations in membrane science, the increasing demand for sophisticated in vitro models in drug discovery and toxicology, and the burgeoning field of regenerative medicine. Strategic opportunities for market players involve expanding their product portfolios to cater to specialized research needs, forming collaborations to integrate inserts into automated workflows, and targeting growth in emerging geographical markets. The market is poised for continued expansion as researchers increasingly rely on these versatile tools to advance scientific understanding and develop next-generation therapies.
Sterile Cell Culture Inserts Segmentation
-
1. Application
- 1.1. Diagnostic Companies and Laboratories
- 1.2. Pharmaceutical Factory
- 1.3. Academic and Research Institutes
- 1.4. Others
-
2. Types
- 2.1. PTFE Membrane
- 2.2. Mixed Cellulose Esters Membrane
- 2.3. Polycarbonate Membrane
- 2.4. PET Membrane
Sterile Cell Culture Inserts Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
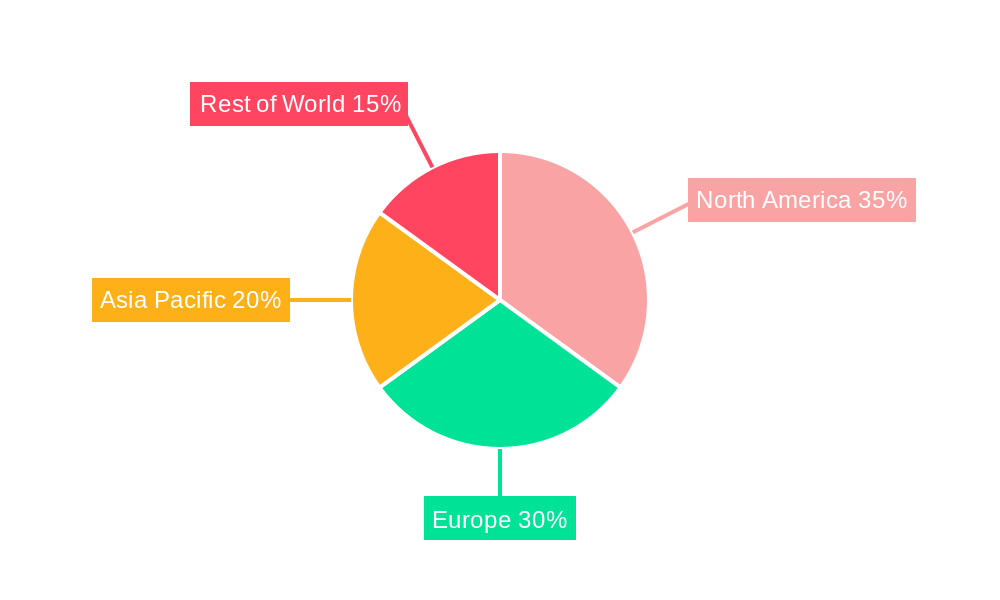
Sterile Cell Culture Inserts Regional Market Share

Geographic Coverage of Sterile Cell Culture Inserts
Sterile Cell Culture Inserts REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Diagnostic Companies and Laboratories
- 5.1.2. Pharmaceutical Factory
- 5.1.3. Academic and Research Institutes
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. PTFE Membrane
- 5.2.2. Mixed Cellulose Esters Membrane
- 5.2.3. Polycarbonate Membrane
- 5.2.4. PET Membrane
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Sterile Cell Culture Inserts Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Diagnostic Companies and Laboratories
- 6.1.2. Pharmaceutical Factory
- 6.1.3. Academic and Research Institutes
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. PTFE Membrane
- 6.2.2. Mixed Cellulose Esters Membrane
- 6.2.3. Polycarbonate Membrane
- 6.2.4. PET Membrane
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Sterile Cell Culture Inserts Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Diagnostic Companies and Laboratories
- 7.1.2. Pharmaceutical Factory
- 7.1.3. Academic and Research Institutes
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. PTFE Membrane
- 7.2.2. Mixed Cellulose Esters Membrane
- 7.2.3. Polycarbonate Membrane
- 7.2.4. PET Membrane
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Sterile Cell Culture Inserts Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Diagnostic Companies and Laboratories
- 8.1.2. Pharmaceutical Factory
- 8.1.3. Academic and Research Institutes
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. PTFE Membrane
- 8.2.2. Mixed Cellulose Esters Membrane
- 8.2.3. Polycarbonate Membrane
- 8.2.4. PET Membrane
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Sterile Cell Culture Inserts Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Diagnostic Companies and Laboratories
- 9.1.2. Pharmaceutical Factory
- 9.1.3. Academic and Research Institutes
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. PTFE Membrane
- 9.2.2. Mixed Cellulose Esters Membrane
- 9.2.3. Polycarbonate Membrane
- 9.2.4. PET Membrane
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Sterile Cell Culture Inserts Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Diagnostic Companies and Laboratories
- 10.1.2. Pharmaceutical Factory
- 10.1.3. Academic and Research Institutes
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. PTFE Membrane
- 10.2.2. Mixed Cellulose Esters Membrane
- 10.2.3. Polycarbonate Membrane
- 10.2.4. PET Membrane
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Sterile Cell Culture Inserts Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Diagnostic Companies and Laboratories
- 11.1.2. Pharmaceutical Factory
- 11.1.3. Academic and Research Institutes
- 11.1.4. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. PTFE Membrane
- 11.2.2. Mixed Cellulose Esters Membrane
- 11.2.3. Polycarbonate Membrane
- 11.2.4. PET Membrane
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Thermo Fisher Scientific
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Corning
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Merck Millipore
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Greiner Bio-One
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 SABEU
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Ibidi GmbH
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Eppendorf
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Sarstedt
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Oxyphen (Filtration Group)
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Celltreat Scientific Products
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 HiMedia Laboratories
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 MatTek Corporation
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 BRAND GMBH + CO KG
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Wuxi NEST BIOTECHNOLOGY
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 SAINING
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.1 Thermo Fisher Scientific
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Sterile Cell Culture Inserts Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Sterile Cell Culture Inserts Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Sterile Cell Culture Inserts Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Sterile Cell Culture Inserts Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Sterile Cell Culture Inserts Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Sterile Cell Culture Inserts Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Sterile Cell Culture Inserts Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Sterile Cell Culture Inserts Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Sterile Cell Culture Inserts Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Sterile Cell Culture Inserts Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Sterile Cell Culture Inserts Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Sterile Cell Culture Inserts Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Sterile Cell Culture Inserts Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Sterile Cell Culture Inserts Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Sterile Cell Culture Inserts Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Sterile Cell Culture Inserts Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Sterile Cell Culture Inserts Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Sterile Cell Culture Inserts Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Sterile Cell Culture Inserts Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Sterile Cell Culture Inserts Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Sterile Cell Culture Inserts Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Sterile Cell Culture Inserts Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Sterile Cell Culture Inserts Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Sterile Cell Culture Inserts Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Sterile Cell Culture Inserts Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Sterile Cell Culture Inserts Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Sterile Cell Culture Inserts Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Sterile Cell Culture Inserts Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Sterile Cell Culture Inserts Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Sterile Cell Culture Inserts Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Sterile Cell Culture Inserts Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Sterile Cell Culture Inserts Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Sterile Cell Culture Inserts Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Sterile Cell Culture Inserts?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the Sterile Cell Culture Inserts?
Key companies in the market include Thermo Fisher Scientific, Corning, Merck Millipore, Greiner Bio-One, SABEU, Ibidi GmbH, Eppendorf, Sarstedt, Oxyphen (Filtration Group), Celltreat Scientific Products, HiMedia Laboratories, MatTek Corporation, BRAND GMBH + CO KG, Wuxi NEST BIOTECHNOLOGY, SAINING.
3. What are the main segments of the Sterile Cell Culture Inserts?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Sterile Cell Culture Inserts," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Sterile Cell Culture Inserts report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Sterile Cell Culture Inserts?
To stay informed about further developments, trends, and reports in the Sterile Cell Culture Inserts, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


