Key Insights
The global Interspinous Stabilization Device market is poised for robust expansion, projected to reach \$155 million in 2025 with a Compound Annual Growth Rate (CAGR) of 6.2% through 2033. This significant growth is primarily fueled by the escalating prevalence of degenerative spinal conditions, such as lumbar degenerative disease and spondylolisthesis, driven by an aging global population and increasingly sedentary lifestyles. The demand for minimally invasive surgical procedures is also a key catalyst, as interspinous stabilization devices offer a less invasive alternative to traditional fusion surgeries, leading to reduced patient recovery times and fewer complications. Furthermore, advancements in material science and device design are contributing to improved efficacy and patient outcomes, thereby driving market adoption. The "Others" application segment, encompassing a range of less common but emerging spinal pathologies, is also expected to contribute to market growth as research and development uncover new therapeutic applications.
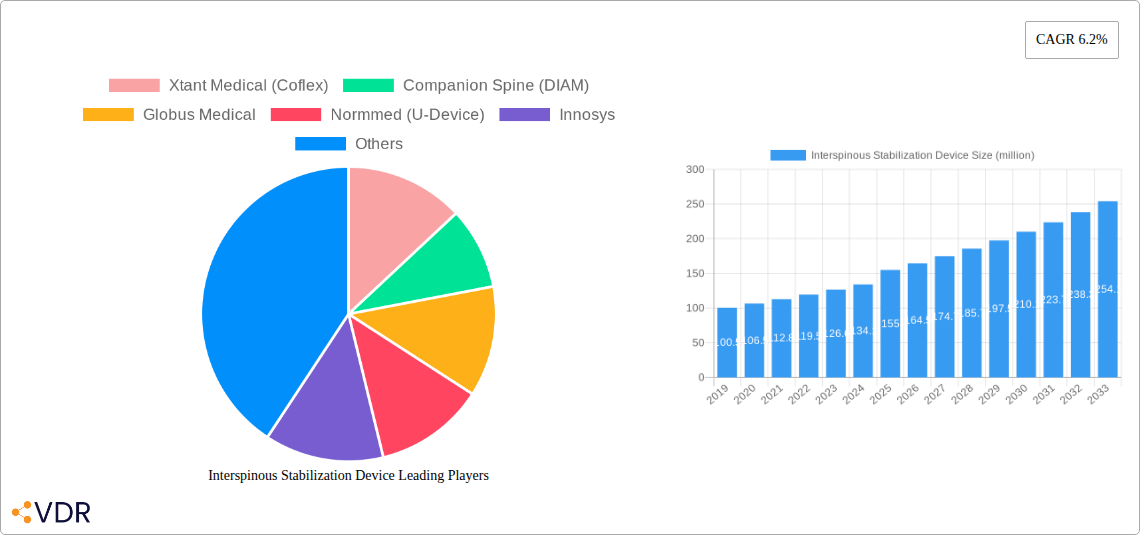
Interspinous Stabilization Device Market Size (In Million)

The market is characterized by intense competition, with key players like Xtant Medical, Companion Spine, and Globus Medical actively innovating and expanding their product portfolios. The distinction between Dynamic and Static Stabilization Devices caters to a spectrum of patient needs and surgical preferences, with dynamic devices offering more flexibility and potentially better preservation of spinal motion. Geographically, North America and Europe are expected to dominate the market, owing to advanced healthcare infrastructure, high healthcare spending, and a strong emphasis on adopting innovative medical technologies. However, the Asia Pacific region presents a substantial growth opportunity due to its large population, increasing disposable income, and a growing awareness of advanced spinal treatments, coupled with a rising incidence of spinal disorders. Despite the promising outlook, potential restraints such as the high cost of these devices and reimbursement challenges in certain regions could temper the market's full potential.
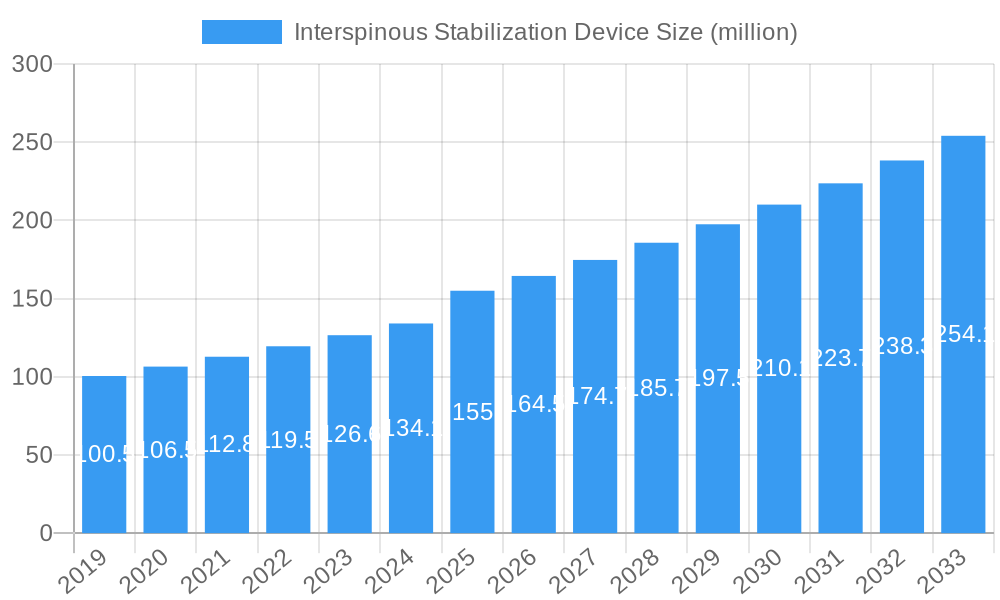
Interspinous Stabilization Device Company Market Share

This in-depth report provides a thorough examination of the global Interspinous Stabilization Device market, offering critical insights into its dynamics, growth trends, and future trajectory. Analyzing parent and child markets, the report leverages high-traffic keywords to maximize SEO visibility and engage orthopedic surgeons, neurosurgeons, spine specialists, medical device manufacturers, distributors, investors, and market research professionals. The study encompasses the historical period (2019-2024), base year (2025), estimated year (2025), and a comprehensive forecast period (2025-2033), presenting all quantitative data in million units.
Interspinous Stabilization Device Market Dynamics & Structure
The Interspinous Stabilization Device market is characterized by a moderate to high concentration, with several key players dominating the landscape. Technological innovation remains a primary driver, fueled by the continuous pursuit of minimally invasive surgical techniques and improved patient outcomes in lumbar degenerative disease and spondylolisthesis treatment. Regulatory frameworks, while stringent, are evolving to accommodate advancements in dynamic stabilisation device technology, fostering an environment conducive to novel product development. Competitive product substitutes, including traditional fusion procedures and other non-fusion technologies, present ongoing challenges, necessitating a focus on demonstrating superior efficacy and patient recovery profiles. End-user demographics are shifting towards an aging global population with a higher incidence of spinal pathologies, alongside a growing demand for less invasive surgical options. Mergers and acquisitions (M&A) are a significant trend, with larger entities acquiring smaller innovators to expand their product portfolios and market reach. The volume of M&A deals is projected to increase by 15% over the forecast period, reflecting the strategic importance of this market segment. Innovation barriers include the high cost of R&D, lengthy clinical trial processes, and the need for extensive surgeon training.
- Market Concentration: Dominated by a few key players, with increasing fragmentation in niche segments.
- Technological Innovation: Driven by advancements in biomaterials, implant design, and minimally invasive surgical approaches.
- Regulatory Frameworks: FDA and EMA approval pathways are critical, with a growing emphasis on real-world evidence.
- Competitive Substitutes: Traditional spinal fusion, interbody cages, and other motion-preserving technologies.
- End-User Demographics: Aging population, rising incidence of degenerative spinal conditions, preference for outpatient procedures.
- M&A Trends: Strategic acquisitions to gain market share and access innovative technologies.
Interspinous Stabilization Device Growth Trends & Insights
The Interspinous Stabilization Device market is poised for substantial growth, driven by an increasing global prevalence of spinal disorders and the growing preference for minimally invasive surgical (MIS) techniques. The market size is projected to expand from approximately $750 million in 2024 to an estimated $1,800 million by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of 9.5% during the forecast period. Adoption rates for interspinous devices are accelerating as surgeons gain more experience and patient outcomes demonstrate clear benefits, such as reduced pain, faster recovery times, and preservation of spinal motion. Technological disruptions are a constant, with advancements in implant materials, such as biocompatible polymers and titanium alloys, offering enhanced durability and reduced inflammatory responses. The development of "smart" interspinous devices with integrated sensors for real-time monitoring of spinal load and motion is also on the horizon, representing a significant shift in patient care. Consumer behavior is evolving; patients are becoming more informed and actively seek out less invasive treatment options with shorter hospital stays and quicker return to daily activities. This shift is a powerful catalyst for the adoption of interspinous stabilization devices, particularly for conditions like lumbar degenerative disease and spondylolisthesis. The market penetration of interspinous devices is expected to rise from 12% in 2024 to an estimated 25% by 2033 in the broader spinal stabilization market. The emphasis on value-based healthcare further supports the adoption of these devices, as they often lead to lower overall healthcare costs due to reduced complications and shorter rehabilitation periods. The child market of dynamic stabilization devices is anticipated to grow at a CAGR of 10.2%, significantly outpacing the static stabilization device segment.
Dominant Regions, Countries, or Segments in Interspinous Stabilization Device
North America currently leads the Interspinous Stabilization Device market, driven by a high prevalence of degenerative spinal conditions, advanced healthcare infrastructure, and strong adoption of innovative medical technologies. The United States, in particular, accounts for a significant portion of global market share, estimated at 45% in 2025. This dominance is underpinned by a well-established reimbursement system that supports advanced surgical procedures and a robust research and development ecosystem. The country's aging population, coupled with a proactive approach to adopting minimally invasive surgical techniques, further solidifies its leadership.
Among the application segments, Lumbar Degenerative Disease is the primary growth driver, accounting for an estimated 60% of the market revenue in 2025. This segment benefits from the high incidence of conditions like spinal stenosis and disc degeneration in the aging population. Spondylolisthesis represents the second-largest application, contributing approximately 25% to the market, with interspinous devices offering a compelling alternative to fusion for certain grades of slippage.
In terms of device type, Dynamic Stabilisation Devices are experiencing rapid growth and are projected to outpace static devices. This segment is expected to capture 70% of the market by 2033, driven by their ability to preserve spinal motion and offer a more physiological approach to stabilization. The market share for dynamic stabilization devices is estimated at 65% in 2025, while static stabilization devices hold the remaining 35%.
Key drivers for regional dominance include:
- Economic Policies: Favorable reimbursement policies and investment in healthcare infrastructure.
- Technological Adoption: Early and widespread acceptance of advanced medical devices.
- Surgeon Expertise: A high concentration of skilled orthopedic and neurosurgeons specializing in spine surgery.
- Patient Awareness: Educated patient populations actively seeking less invasive treatment options.
- Regulatory Support: Efficient and supportive regulatory pathways for new device approvals.
Asia Pacific is emerging as a significant growth region, driven by a rapidly expanding middle class, increasing healthcare expenditure, and a growing awareness of advanced medical treatments. Countries like China and India are witnessing substantial growth due to their large populations and improving access to healthcare services.
Interspinous Stabilization Device Product Landscape
The product landscape for interspinous stabilization devices is characterized by ongoing innovation focused on enhancing biomechanical performance, patient comfort, and ease of surgical implantation. Companies are developing devices with improved fixation mechanisms, offering superior stability while minimizing disruption to surrounding tissues. Advancements in materials science have led to the introduction of radiolucent implants, which do not interfere with post-operative imaging, and bio-integrated materials that promote better tissue healing. Unique selling propositions often revolve around the device's ability to preserve segmental motion, reduce adjacent segment degeneration, and provide a less invasive alternative to fusion. Technological advancements are also extending to the development of patient-specific implants and navigation-assisted surgical techniques, further personalizing treatment and optimizing outcomes.
Key Drivers, Barriers & Challenges in Interspinous Stabilization Device
The Interspinous Stabilization Device market is propelled by several key drivers: a rising global geriatric population experiencing increased spinal degeneration, a growing demand for minimally invasive surgical procedures, advancements in implant design and materials, and favorable reimbursement policies for non-fusion alternatives. The increasing focus on motion-preserving spinal surgery further fuels market expansion, as interspinous devices are a prime example of this trend.
- Technological Advancement: Development of novel materials, improved implant fixation, and compatibility with MIS techniques.
- Aging Population: Higher incidence of degenerative spinal conditions driving demand.
- Minimally Invasive Surgery: Patient and surgeon preference for less invasive procedures.
- Reimbursement: Favorable policies for non-fusion alternatives.
Key challenges and restraints include the high cost of research and development and manufacturing, the need for extensive surgeon training and education, and the presence of established competitors offering alternative surgical solutions like spinal fusion. Regulatory hurdles and the lengthy approval processes can also slow down market entry for new products. Supply chain disruptions, particularly for specialized materials and components, pose a continuous threat. The perceived risk associated with novel technologies compared to traditional fusion methods can also be a significant barrier. The market is estimated to lose $50 million in potential revenue annually due to these restraints.
- High R&D and Manufacturing Costs: Significant investment required for innovation and production.
- Surgeon Training and Education: Need for comprehensive training to ensure proper utilization.
- Competition from Fusion: Established preference and reimbursement for traditional fusion procedures.
- Regulatory Hurdles: Lengthy and complex approval processes.
- Supply Chain Disruptions: Potential for material and component shortages.
Emerging Opportunities in Interspinous Stabilization Device
Emerging opportunities in the Interspinous Stabilization Device market lie in the development of next-generation dynamic stabilization systems with enhanced biocompatibility and load-sharing capabilities. The expanding use of these devices in treating adjacent segment disease post-fusion also presents a significant untapped market. Furthermore, the integration of AI and machine learning in surgical planning and device selection for interspinous implants holds immense potential. The increasing adoption of telehealth and remote patient monitoring can also pave the way for new service models around interspinous device therapy, focusing on post-operative care and rehabilitation.
Growth Accelerators in the Interspinous Stabilization Device Industry
Long-term growth in the Interspinous Stabilization Device industry will be significantly accelerated by continued technological breakthroughs in biomaterials and implant mechanics, leading to devices that more closely mimic natural spinal function. Strategic partnerships between device manufacturers and research institutions will drive innovation, while collaborations with hospitals and surgical centers will facilitate increased adoption and training. Market expansion strategies targeting emerging economies with growing healthcare needs and increasing disposable incomes will also serve as key growth accelerators. The development of user-friendly, navigated surgical systems specifically designed for interspinous device implantation will further enhance surgeon confidence and procedural efficiency.
Key Players Shaping the Interspinous Stabilization Device Market
- Xtant Medical
- Companion Spine
- Globus Medical
- Normmed
- Innosys
- Spinal Elements
- ARTFX Medical
- SeohanCare
- Wenzel Spine
- Changzhou Geasure Medical
Notable Milestones in Interspinous Stabilization Device Sector
- 2019: Launch of advanced biomaterial-enhanced interspinous devices with improved osseointegration.
- 2020: Significant increase in clinical studies demonstrating superior patient outcomes for MIS interspinous stabilization in lumbar degenerative disease.
- 2021: Major acquisition of a key innovator in dynamic interspinous technology by a leading spinal implant company.
- 2022: FDA approval for a novel interspinous device featuring a modular design for enhanced surgical flexibility.
- 2023: Growing adoption of interspinous devices for treating adjacent segment disease, showing promising results.
- 2024: Introduction of next-generation static interspinous implants offering improved bone purchase and revision potential.
In-Depth Interspinous Stabilization Device Market Outlook
The Interspinous Stabilization Device market is on an upward trajectory, driven by an aging global population, a relentless pursuit of less invasive surgical solutions, and continuous technological advancements. Future market potential is substantial, with a strong emphasis on dynamic stabilization and motion preservation. Strategic opportunities include further penetration into emerging markets, development of AI-integrated surgical planning tools, and expansion of product offerings for complex spinal pathologies. The market is expected to witness sustained growth, offering significant returns for stakeholders invested in spinal innovation.
Interspinous Stabilization Device Segmentation
-
1. Application
- 1.1. Lumbar Degenerative Disease
- 1.2. Spondylolisthesis
- 1.3. Others
-
2. Type
- 2.1. Dynamic Stabilisation Device
- 2.2. Static Stabilisation Device
Interspinous Stabilization Device Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
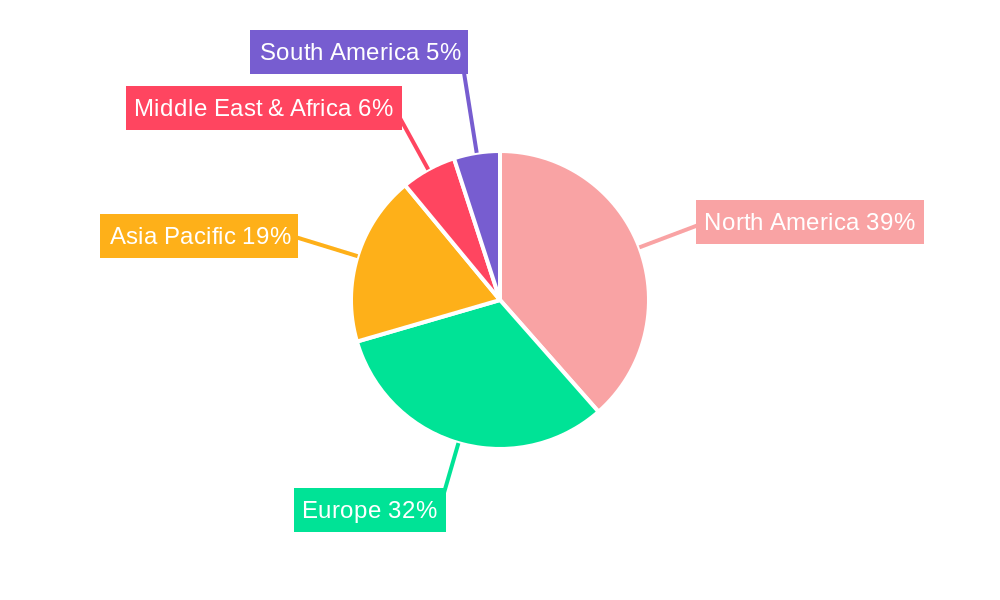
Interspinous Stabilization Device Regional Market Share

Geographic Coverage of Interspinous Stabilization Device
Interspinous Stabilization Device REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.2% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Lumbar Degenerative Disease
- 5.1.2. Spondylolisthesis
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Dynamic Stabilisation Device
- 5.2.2. Static Stabilisation Device
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Interspinous Stabilization Device Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Lumbar Degenerative Disease
- 6.1.2. Spondylolisthesis
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Dynamic Stabilisation Device
- 6.2.2. Static Stabilisation Device
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Interspinous Stabilization Device Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Lumbar Degenerative Disease
- 7.1.2. Spondylolisthesis
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Dynamic Stabilisation Device
- 7.2.2. Static Stabilisation Device
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Interspinous Stabilization Device Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Lumbar Degenerative Disease
- 8.1.2. Spondylolisthesis
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Dynamic Stabilisation Device
- 8.2.2. Static Stabilisation Device
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Interspinous Stabilization Device Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Lumbar Degenerative Disease
- 9.1.2. Spondylolisthesis
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Dynamic Stabilisation Device
- 9.2.2. Static Stabilisation Device
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Interspinous Stabilization Device Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Lumbar Degenerative Disease
- 10.1.2. Spondylolisthesis
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Dynamic Stabilisation Device
- 10.2.2. Static Stabilisation Device
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Interspinous Stabilization Device Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Lumbar Degenerative Disease
- 11.1.2. Spondylolisthesis
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Type
- 11.2.1. Dynamic Stabilisation Device
- 11.2.2. Static Stabilisation Device
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Xtant Medical (Coflex)
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Companion Spine (DIAM)
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Globus Medical
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Normmed (U-Device)
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Innosys
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Spinal Elements
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 ARTFX Medical
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 SeohanCare
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Wenzel Spine
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Changzhou Geasure Medical
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.1 Xtant Medical (Coflex)
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Interspinous Stabilization Device Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Interspinous Stabilization Device Revenue (million), by Application 2025 & 2033
- Figure 3: North America Interspinous Stabilization Device Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Interspinous Stabilization Device Revenue (million), by Type 2025 & 2033
- Figure 5: North America Interspinous Stabilization Device Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Interspinous Stabilization Device Revenue (million), by Country 2025 & 2033
- Figure 7: North America Interspinous Stabilization Device Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Interspinous Stabilization Device Revenue (million), by Application 2025 & 2033
- Figure 9: South America Interspinous Stabilization Device Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Interspinous Stabilization Device Revenue (million), by Type 2025 & 2033
- Figure 11: South America Interspinous Stabilization Device Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Interspinous Stabilization Device Revenue (million), by Country 2025 & 2033
- Figure 13: South America Interspinous Stabilization Device Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Interspinous Stabilization Device Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Interspinous Stabilization Device Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Interspinous Stabilization Device Revenue (million), by Type 2025 & 2033
- Figure 17: Europe Interspinous Stabilization Device Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Interspinous Stabilization Device Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Interspinous Stabilization Device Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Interspinous Stabilization Device Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Interspinous Stabilization Device Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Interspinous Stabilization Device Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa Interspinous Stabilization Device Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Interspinous Stabilization Device Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Interspinous Stabilization Device Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Interspinous Stabilization Device Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Interspinous Stabilization Device Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Interspinous Stabilization Device Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific Interspinous Stabilization Device Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Interspinous Stabilization Device Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Interspinous Stabilization Device Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Interspinous Stabilization Device Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Interspinous Stabilization Device Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global Interspinous Stabilization Device Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Interspinous Stabilization Device Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Interspinous Stabilization Device Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global Interspinous Stabilization Device Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Interspinous Stabilization Device Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Interspinous Stabilization Device Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global Interspinous Stabilization Device Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Interspinous Stabilization Device Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Interspinous Stabilization Device Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global Interspinous Stabilization Device Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Interspinous Stabilization Device Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Interspinous Stabilization Device Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global Interspinous Stabilization Device Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Interspinous Stabilization Device Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Interspinous Stabilization Device Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global Interspinous Stabilization Device Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Interspinous Stabilization Device Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Interspinous Stabilization Device?
The projected CAGR is approximately 6.2%.
2. Which companies are prominent players in the Interspinous Stabilization Device?
Key companies in the market include Xtant Medical (Coflex), Companion Spine (DIAM), Globus Medical, Normmed (U-Device), Innosys, Spinal Elements, ARTFX Medical, SeohanCare, Wenzel Spine, Changzhou Geasure Medical.
3. What are the main segments of the Interspinous Stabilization Device?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 155 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Interspinous Stabilization Device," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Interspinous Stabilization Device report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Interspinous Stabilization Device?
To stay informed about further developments, trends, and reports in the Interspinous Stabilization Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
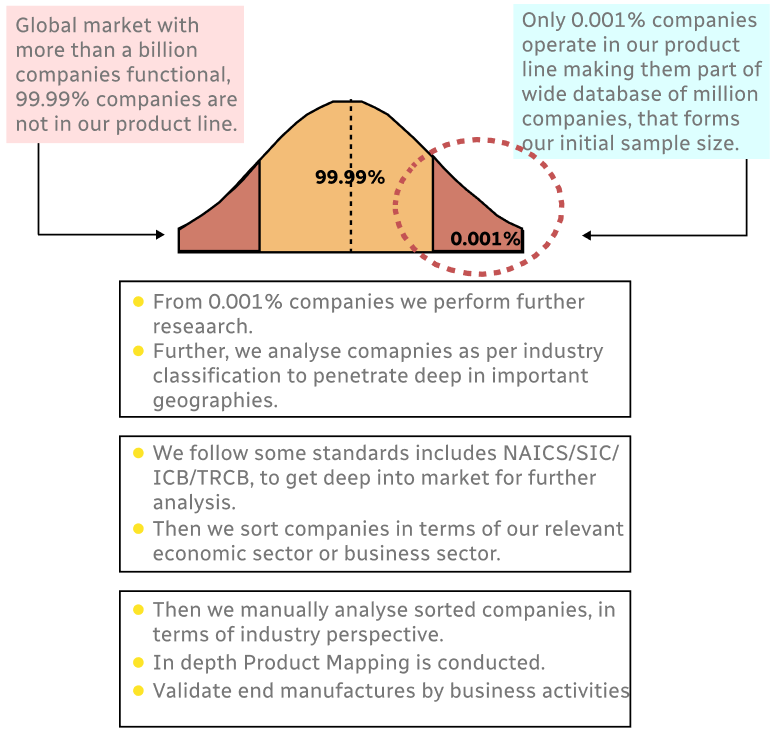
Step 1 - Identification of Relevant Samples Size from Population Database
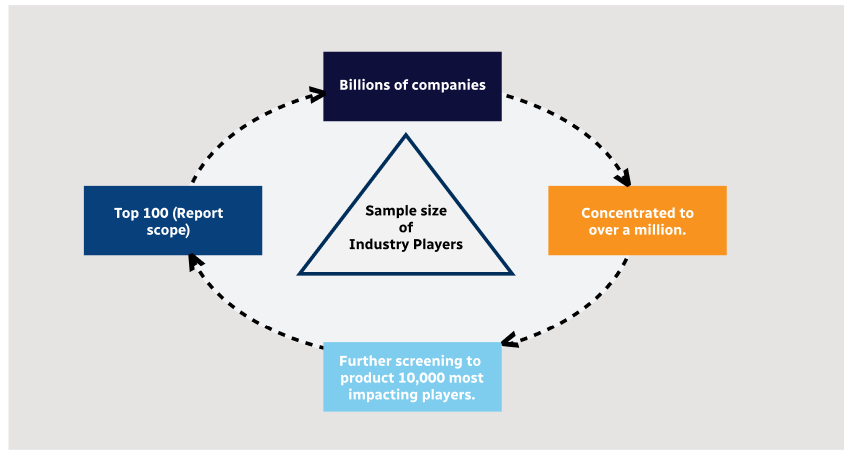
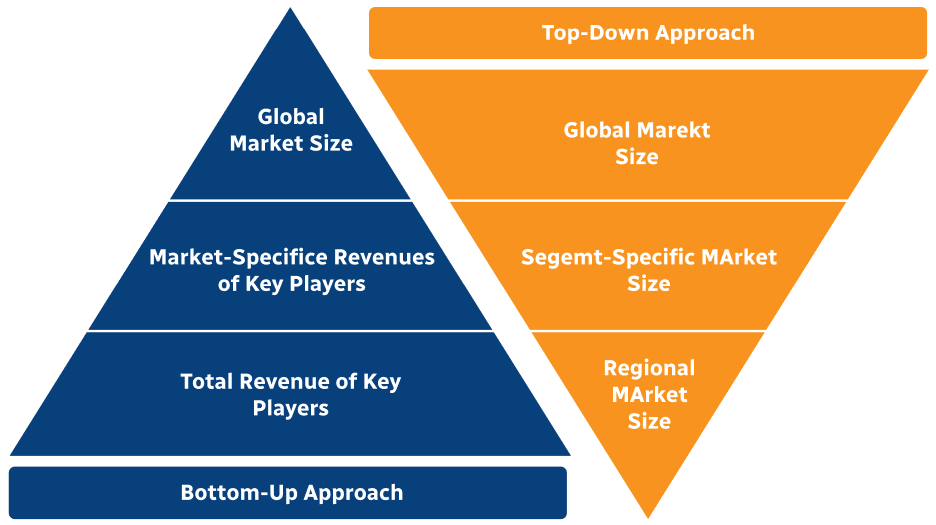
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
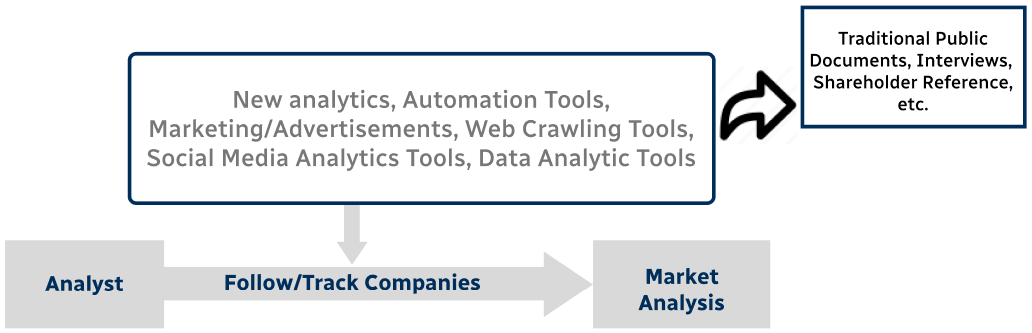
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


