Key Insights
The France Spinal Surgery Devices Market is poised for significant expansion, projected to reach an estimated €14.7 billion in 2025, driven by an anticipated Compound Annual Growth Rate (CAGR) of 6% throughout the forecast period (2025-2033). This robust growth is primarily fueled by an aging population experiencing a higher incidence of spinal disorders such as degenerative disc disease, scoliosis, and spinal stenosis. Advancements in minimally invasive surgical techniques and the development of innovative spinal implants and instruments are also major catalysts, offering patients improved outcomes and faster recovery times. Furthermore, increasing healthcare expenditure and a greater emphasis on specialized spine care within the French healthcare system are contributing to the market's upward trajectory.
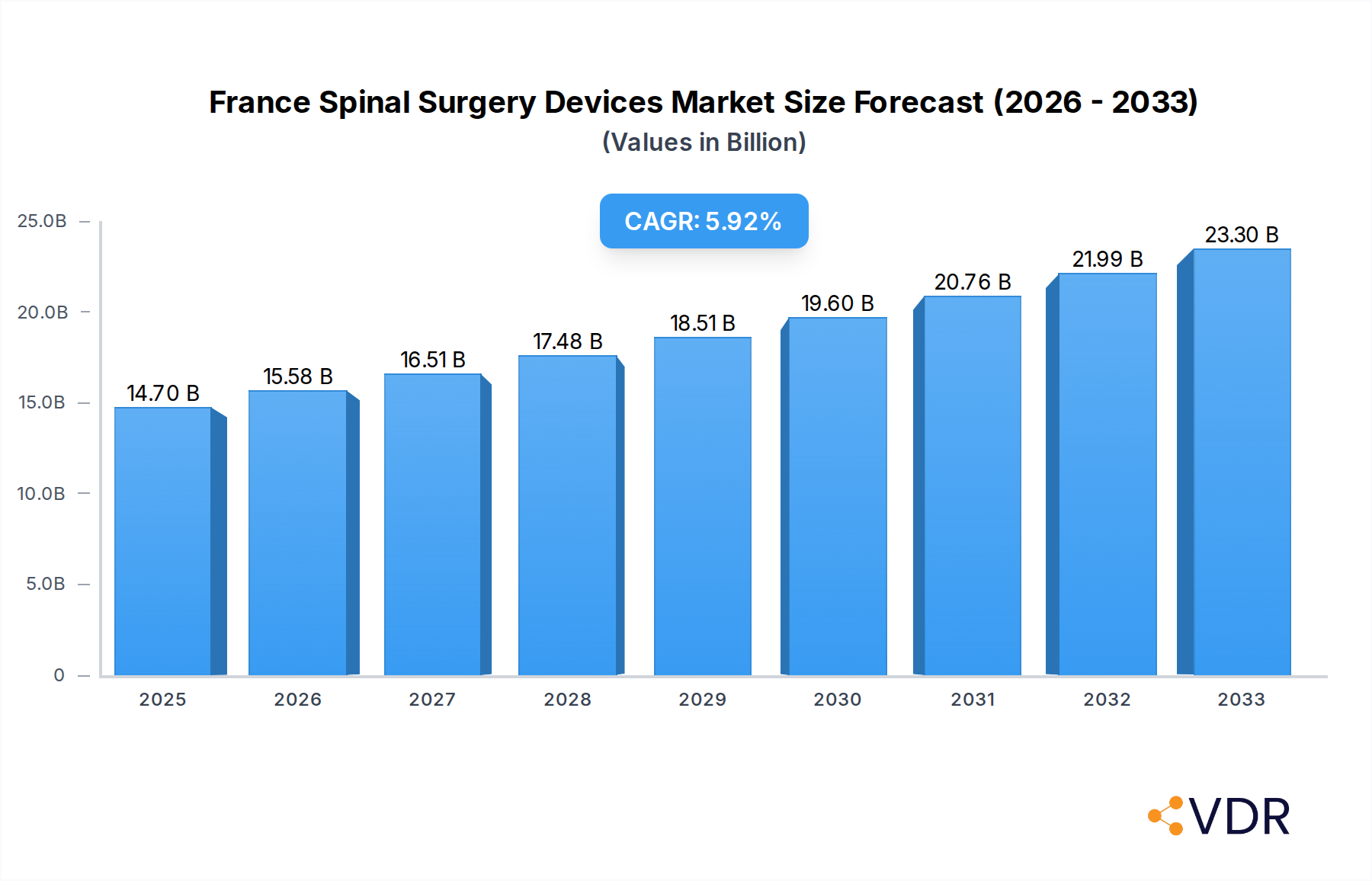
France Spinal Surgery Devices Market Market Size (In Billion)

The market segmentation for spinal surgery devices in France is diverse, encompassing critical categories like Spinal Decompression (including Corpectomy, Discectomy, Facetectomy, Foraminotomy, and Laminotomy), Spinal Fusion (covering Cervical Fusion, Interbody Fusion, and ThoracoLumbar Fusion), Fracture Repair Devices, Arthroplasty Devices, and Non-fusion Devices. Key players such as Medtronic PLC, Johnson & Johnson, Stryker Corporation, Globus Medical, and NuVasive Inc. are actively investing in research and development to introduce next-generation spinal solutions. While the market demonstrates strong growth potential, it faces certain restraints, including the high cost of advanced spinal surgery devices and the need for specialized surgeon training, which could potentially moderate the pace of adoption in certain segments. However, the overall outlook remains exceptionally positive, with a strong demand for effective treatments for spinal conditions.
France Spinal Surgery Devices Market Company Market Share

This in-depth report provides a definitive analysis of the France Spinal Surgery Devices Market, offering critical insights for stakeholders navigating this dynamic sector. Leveraging high-traffic keywords such as "spinal surgery devices France," "orthopedic implants France," "minimally invasive spine surgery," "spinal fusion devices," "spinal decompression devices," and "vertebral fracture repair," this report is SEO-optimized to maximize visibility and engagement within the industry. Our comprehensive study, spanning from 2019 to 2033 with a base year of 2025, delves into market dynamics, growth trends, regional dominance, product landscape, key drivers, challenges, emerging opportunities, and the competitive strategies of leading players.
France Spinal Surgery Devices Market Market Dynamics & Structure
The France Spinal Surgery Devices Market is characterized by a moderately concentrated landscape, with leading global players like Medtronic PLC, Johnson & Johnson, and Stryker Corporation holding significant market share. Technological innovation is a paramount driver, with continuous advancements in minimally invasive surgical techniques, robotic-assisted surgery, and biomaterials significantly influencing product development and adoption. The stringent regulatory framework in France, overseen by bodies like the ANSM, ensures patient safety and product efficacy, albeit presenting a barrier to entry for novel technologies. Competitive product substitutes, including conservative treatments and alternative surgical approaches, also shape market dynamics. End-user demographics, particularly the aging population and the increasing prevalence of degenerative spine conditions, fuel demand. Mergers and acquisitions (M&A) activity, while present, is strategic, aiming to consolidate market presence or acquire innovative technologies. For instance, historical M&A activity in the broader European spine market suggests a trend towards acquiring specialized technology providers, contributing to market consolidation. The market's structure is further influenced by the increasing demand for patient-specific solutions and the growing integration of digital health technologies in surgical planning and execution.
- Market Concentration: Moderately concentrated with key global players.
- Technological Innovation: Driven by minimally invasive techniques, robotics, and advanced biomaterials.
- Regulatory Framework: Robust, ensuring safety and efficacy.
- Competitive Landscape: Includes conservative treatments and alternative surgical methods.
- End-User Demographics: Aging population and rising incidence of spinal disorders.
- M&A Trends: Strategic acquisitions of technology specialists.
France Spinal Surgery Devices Market Growth Trends & Insights
The France Spinal Surgery Devices Market is poised for robust growth, projected to witness a Compound Annual Growth Rate (CAGR) of approximately 6.5% between 2025 and 2033. This expansion is underpinned by several pivotal factors. The escalating prevalence of age-related spinal conditions, such as degenerative disc disease and spinal stenosis, directly translates into increased demand for surgical interventions. Furthermore, a growing awareness among both patients and healthcare professionals regarding the benefits of minimally invasive spine surgery (MISS) – including reduced hospital stays, faster recovery times, and decreased post-operative pain – is a significant adoption driver. This shift towards less invasive procedures necessitates specialized spinal surgery devices, thereby stimulating market growth.
Technological advancements are continuously reshaping the market. The integration of artificial intelligence (AI) in surgical planning and navigation, the development of advanced implant materials with enhanced biocompatibility and longevity, and the refinement of robotic-assisted surgical systems are all contributing to improved patient outcomes and procedural efficiency. For instance, the adoption of 3D-printed implants for complex reconstructions is gaining traction, offering customized solutions for intricate anatomical challenges.
Consumer behavior is also evolving. Patients are increasingly seeking proactive solutions for spinal pain and are more informed about treatment options, often actively participating in their healthcare decisions. This heightened patient engagement, coupled with favorable reimbursement policies for certain advanced procedures, further propels market expansion. The market penetration for advanced spinal fusion devices, for example, is expected to rise significantly as surgeons become more proficient in their use and as evidence of their superior clinical outcomes continues to accumulate. The overall market size is estimated to reach approximately €1.8 billion by 2033, up from an estimated €1.1 billion in 2025. This growth trajectory is indicative of a market that is not only expanding in volume but also in the sophistication and value of the solutions it offers.
- Market Size Evolution: Projected to reach €1.8 billion by 2033.
- CAGR (2025–2033): Approximately 6.5%.
- Adoption Rates: Driven by increasing awareness and benefits of MISS.
- Technological Disruptions: AI in surgical planning, advanced biomaterials, robotics.
- Consumer Behavior Shifts: Increased patient awareness and proactive healthcare seeking.
- Market Penetration: Rising for advanced spinal fusion and MISS devices.
Dominant Regions, Countries, or Segments in France Spinal Surgery Devices Market
Within the France Spinal Surgery Devices Market, the Spinal Fusion segment is a dominant force, consistently driving market growth and innovation. This segment, encompassing Cervical Fusion, Interbody Fusion, ThoracoLumbar Fusion, and Other Spinal Fusions, represents the largest share of the market due to the high incidence of degenerative conditions, spinal deformities, and trauma requiring stabilization and fusion procedures. The increasing demand for advanced interbody fusion devices, particularly those utilizing porous structures for bone ingrowth and biologic augmentation, is a key growth driver within this segment.
The ThoracoLumbar Fusion sub-segment, specifically, is experiencing significant expansion. This is attributable to the high prevalence of degenerative disc disease and spinal stenosis in the lower back, which are common conditions in the aging French population. Furthermore, the increasing adoption of minimally invasive surgical techniques for thoracolumbar fusion, such as lateral access and anterior approaches, contributes to its growth by offering improved patient recovery and reduced complications. The availability of innovative implants like expandable cages and biomechanically superior screw-rod systems further bolsters this sub-segment.
In terms of geographical dominance within France, the Île-de-France region, encompassing Paris, stands out as the leading market. This is primarily due to the concentration of highly specialized spine surgery centers, leading orthopedic hospitals, and a dense network of experienced spine surgeons. The region also benefits from a higher disposable income, allowing for greater adoption of advanced and premium spinal surgery devices. Economic policies that support medical innovation and research, coupled with robust healthcare infrastructure, further cement Île-de-France's position. The market share within this region is estimated to be over 30% of the total French market.
The Spinal Decompression segment, including procedures like Corpectomy, Discectomy, Facetectomy, Foraminotomy, and Laminotomy, also holds a significant market share, driven by the need to alleviate neurological compression caused by herniated discs, bone spurs, and thickened ligaments. While not as large as spinal fusion, its consistent demand, particularly for discectomy and laminectomy, makes it a crucial segment. The development of advanced tools for minimally invasive decompression, such as endoscopic instruments, is a key factor in its continued growth.
- Dominant Segment: Spinal Fusion (including Cervical Fusion, Interbody Fusion, ThoracoLumbar Fusion, Other Spinal Fusions).
- Key Sub-Segment Driver: ThoracoLumbar Fusion, driven by degenerative conditions and MISS adoption.
- Dominant Region in France: Île-de-France, due to concentration of expertise and advanced facilities.
- Leading Segment Growth Factors: High prevalence of degenerative conditions, adoption of MISS, advanced implant technologies.
- Market Share in Île-de-France: Estimated to be over 30% of the total French market.
- Significant Segment: Spinal Decompression, fueled by neurological compression issues.
France Spinal Surgery Devices Market Product Landscape
The France Spinal Surgery Devices Market is defined by a diverse and rapidly evolving product landscape. Innovations are consistently focused on enhancing patient outcomes through minimally invasive techniques and advanced implant materials. Key product categories include spinal fusion devices, such as interbody cages made from PEEK (polyether ether ketone) and titanium, as well as advanced pedicle screw and rod systems designed for improved biomechanical stability. Spinal decompression devices are seeing advancements in endoscopic and microscopic surgical instruments, enabling more precise nerve root decompression. Fracture repair devices are incorporating innovative fixation technologies for vertebral compression fractures, including kyphoplasty balloons and minimally invasive screw systems. Arthroplasty devices, though a smaller segment, are exploring motion-preserving technologies to mitigate adjacent segment disease. The unique selling proposition for many of these products lies in their biocompatibility, radiolucency, and ability to promote faster bone healing. Technological advancements are also evident in navigation and robotic-assisted surgery systems, which are increasingly being integrated with spinal surgery devices to improve accuracy and reduce procedure times. The market is also witnessing the introduction of absorbable or bioresorbable implants designed to degrade over time, reducing the need for hardware removal and minimizing long-term complications.
Key Drivers, Barriers & Challenges in France Spinal Surgery Devices Market
Key Drivers:
The France Spinal Surgery Devices Market is propelled by several significant drivers. The aging demographic of the French population, leading to a higher incidence of degenerative spinal conditions like osteoarthritis and spinal stenosis, is a primary catalyst. The increasing adoption of minimally invasive spine surgery (MISS) techniques, driven by their benefits such as reduced patient trauma and faster recovery, fuels demand for specialized devices. Technological advancements, including robotic surgery, AI-assisted navigation, and novel biomaterials, are creating a demand for sophisticated implants and instruments. Furthermore, growing patient awareness and demand for effective pain management solutions are encouraging more individuals to seek surgical interventions. Favorable reimbursement policies for certain advanced spine procedures also contribute to market growth.
Barriers & Challenges:
Despite the positive outlook, the market faces several barriers and challenges. The high cost of advanced spinal surgery devices and technologies can be a significant restraint, particularly in healthcare systems facing budget constraints. Stringent regulatory approval processes in France and the European Union can prolong the time-to-market for new products, acting as a barrier to innovation adoption. The availability of skilled surgeons trained in complex MISS techniques and robotic surgery is also a limiting factor. Intense competition among global and local manufacturers leads to price pressures. Additionally, the potential for surgical complications and the need for extensive post-operative rehabilitation can influence patient and surgeon decision-making. Supply chain disruptions, as experienced globally in recent years, can also pose challenges for manufacturers in ensuring timely availability of critical components and finished products, potentially impacting delivery timelines and increasing costs.
Emerging Opportunities in France Spinal Surgery Devices Market
Emerging opportunities in the France Spinal Surgery Devices Market are largely centered around technological integration and an increasing focus on patient-specific solutions. The expansion of AI and machine learning in surgical planning and real-time intraoperative guidance presents a significant avenue for growth, promising enhanced precision and improved outcomes. The development of biologics and regenerative medicine solutions designed to augment spinal fusion and accelerate bone healing offers a promising area for innovation, potentially reducing the reliance on hardware alone. Furthermore, the growing interest in motion-preserving technologies, such as advanced artificial disc replacements for cervical and lumbar spine, represents a significant opportunity to address the issue of adjacent segment degeneration, a known complication of traditional fusion. The untapped potential in developing countries within Europe, and niche applications for spinal surgery devices in treating conditions beyond the common degenerative ones, also presents avenues for market expansion.
Growth Accelerators in the France Spinal Surgery Devices Market Industry
Several key catalysts are accelerating long-term growth within the France Spinal Surgery Devices Market. The continuous evolution of robotic-assisted surgical systems and their increasing integration into spinal procedures is a major accelerator, offering enhanced dexterity, precision, and visualization for surgeons. Strategic partnerships between device manufacturers and technology companies, such as those focusing on AI and data analytics, are fostering innovation and the development of smarter, more integrated surgical solutions. Furthermore, advancements in materials science, leading to the development of stronger, lighter, and more biocompatible implant materials, are expanding the possibilities for complex spinal reconstructions and improving implant longevity. The growing trend towards value-based healthcare models, which emphasize patient outcomes and cost-effectiveness, encourages the adoption of technologies that demonstrate superior clinical efficacy and a reduced total cost of care, thereby accelerating market growth.
Key Players Shaping the France Spinal Surgery Devices Market Market
- Joimax GmbH
- Orthofix Holdings Inc
- Medtronic PLC
- Globus Medical
- Nuvasive Inc
- Johnson & Johnson
- Stryker Corporation
- Zimmer Biomet
- SeaSpine Holdings Corporation
- RTI Surgical Holdings Inc
Notable Milestones in France Spinal Surgery Devices Market Sector
- November 2022: Implanet signed an exclusive distribution contract for SMTP Technology Co.'s ultrasonic surgical scalpel in France, enhancing its offerings in spine surgery within French medical centers.
- December 2022: Spineart and eCential Robotics entered a long-term collaboration agreement to expand their offerings in France and Switzerland, integrating robotics, surgical navigation, and 2D/3D robotic imaging for spine surgery solutions.
In-Depth France Spinal Surgery Devices Market Market Outlook
The future outlook for the France Spinal Surgery Devices Market is exceptionally promising, driven by sustained technological advancements and an expanding patient base. Growth accelerators, including the pervasive integration of robotics and AI in surgical interventions, will further refine procedural accuracy and patient outcomes. The increasing emphasis on value-based healthcare will favor innovative solutions that demonstrate superior efficacy and cost-effectiveness, thereby fostering the adoption of advanced spinal fusion and motion-preserving technologies. Strategic alliances and collaborations will continue to be pivotal in bringing novel solutions to market, addressing unmet clinical needs. The market is set to witness a paradigm shift towards personalized medicine, with custom-designed implants and data-driven treatment pathways becoming more prevalent. This forward-looking approach positions the France Spinal Surgery Devices Market for continued expansion and innovation, catering to the evolving demands of both healthcare providers and patients seeking effective solutions for spinal health.
France Spinal Surgery Devices Market Segmentation
-
1. Device Type
-
1.1. Spinal Decompression
- 1.1.1. Corpectomy
- 1.1.2. Discectomy
- 1.1.3. Facetectomy
- 1.1.4. Foraminotomy
- 1.1.5. Laminotomy
-
1.2. Spinal Fusion
- 1.2.1. Cervical Fusion
- 1.2.2. Interbody Fusion
- 1.2.3. ThoracoLumbar Fusion
- 1.2.4. Other Spinal Fusions
- 1.3. Fracture Repair Devices
- 1.4. Arthroplasty Devices
- 1.5. Non-fusion Devices
- 1.6. Other Fracture Repair Devices
-
1.1. Spinal Decompression
France Spinal Surgery Devices Market Segmentation By Geography
- 1. France
France Spinal Surgery Devices Market Regional Market Share

Geographic Coverage of France Spinal Surgery Devices Market
France Spinal Surgery Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 5.1.1. Spinal Decompression
- 5.1.1.1. Corpectomy
- 5.1.1.2. Discectomy
- 5.1.1.3. Facetectomy
- 5.1.1.4. Foraminotomy
- 5.1.1.5. Laminotomy
- 5.1.2. Spinal Fusion
- 5.1.2.1. Cervical Fusion
- 5.1.2.2. Interbody Fusion
- 5.1.2.3. ThoracoLumbar Fusion
- 5.1.2.4. Other Spinal Fusions
- 5.1.3. Fracture Repair Devices
- 5.1.4. Arthroplasty Devices
- 5.1.5. Non-fusion Devices
- 5.1.6. Other Fracture Repair Devices
- 5.1.1. Spinal Decompression
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. France
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 6. France Spinal Surgery Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 6.1.1. Spinal Decompression
- 6.1.1.1. Corpectomy
- 6.1.1.2. Discectomy
- 6.1.1.3. Facetectomy
- 6.1.1.4. Foraminotomy
- 6.1.1.5. Laminotomy
- 6.1.2. Spinal Fusion
- 6.1.2.1. Cervical Fusion
- 6.1.2.2. Interbody Fusion
- 6.1.2.3. ThoracoLumbar Fusion
- 6.1.2.4. Other Spinal Fusions
- 6.1.3. Fracture Repair Devices
- 6.1.4. Arthroplasty Devices
- 6.1.5. Non-fusion Devices
- 6.1.6. Other Fracture Repair Devices
- 6.1.1. Spinal Decompression
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Joimax GmbH
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Orthofix Holdings Inc
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Medtronic PLC
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Globus Medical
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Nuvasive Inc
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Johnson & Johnson
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 Stryker Corporation
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Zimmer Biomet
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 SeaSpine Holdings Corporation
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 RTI Surgical Holdings Inc
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.1 Joimax GmbH
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: France Spinal Surgery Devices Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: France Spinal Surgery Devices Market Share (%) by Company 2025
List of Tables
- Table 1: France Spinal Surgery Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 2: France Spinal Surgery Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 3: France Spinal Surgery Devices Market Revenue billion Forecast, by Region 2020 & 2033
- Table 4: France Spinal Surgery Devices Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 5: France Spinal Surgery Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 6: France Spinal Surgery Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 7: France Spinal Surgery Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 8: France Spinal Surgery Devices Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the France Spinal Surgery Devices Market?
The projected CAGR is approximately 6%.
2. Which companies are prominent players in the France Spinal Surgery Devices Market?
Key companies in the market include Joimax GmbH, Orthofix Holdings Inc, Medtronic PLC, Globus Medical, Nuvasive Inc, Johnson & Johnson, Stryker Corporation, Zimmer Biomet, SeaSpine Holdings Corporation, RTI Surgical Holdings Inc .
3. What are the main segments of the France Spinal Surgery Devices Market?
The market segments include Device Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 14.7 billion as of 2022.
5. What are some drivers contributing to market growth?
Rise in Technological Advances in Spinal Surgery and Increasing Demand for Minimally Invasive Surgical Procedures; Increasing Incidence of Obesity. Aging Population. and Associated Spine Disorders.
6. What are the notable trends driving market growth?
Arthroplasty Devices Segment is Expected to Hold Significant Share in the French Spinal Surgery Devices Market.
7. Are there any restraints impacting market growth?
Expensive Treatment Procedures and Stringent Reimbursement Concerns.
8. Can you provide examples of recent developments in the market?
November 2022: Implanet signed an exclusive distribution contract for SMTP Technology Co.'s ultrasonic surgical scalpel in France. Implanet is a company specializing in spine surgery, and its foothold is in French medical centers.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "France Spinal Surgery Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the France Spinal Surgery Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the France Spinal Surgery Devices Market?
To stay informed about further developments, trends, and reports in the France Spinal Surgery Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
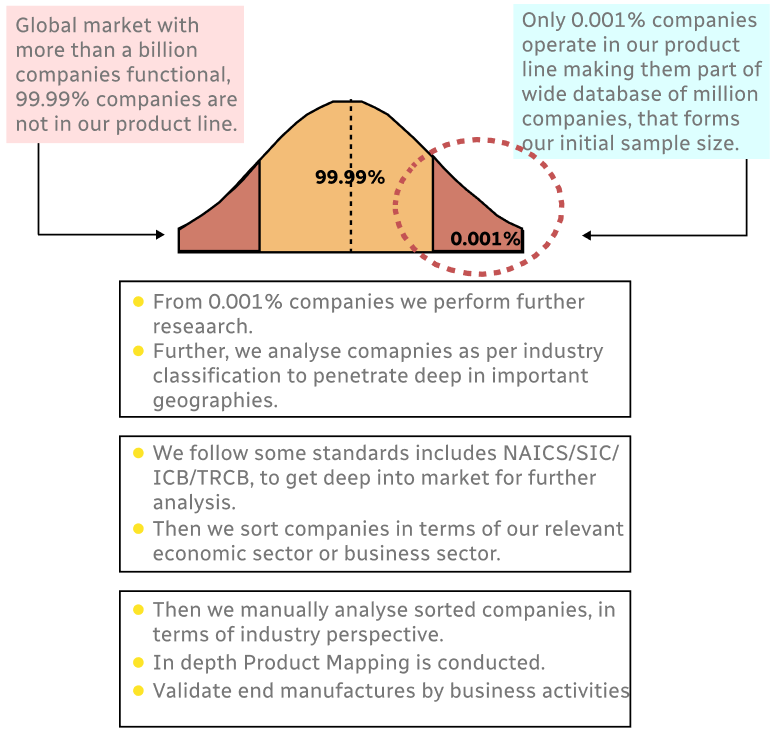
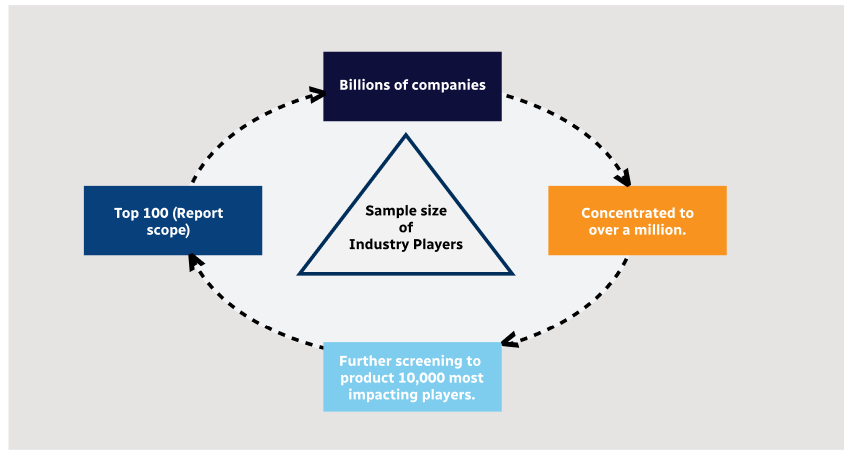
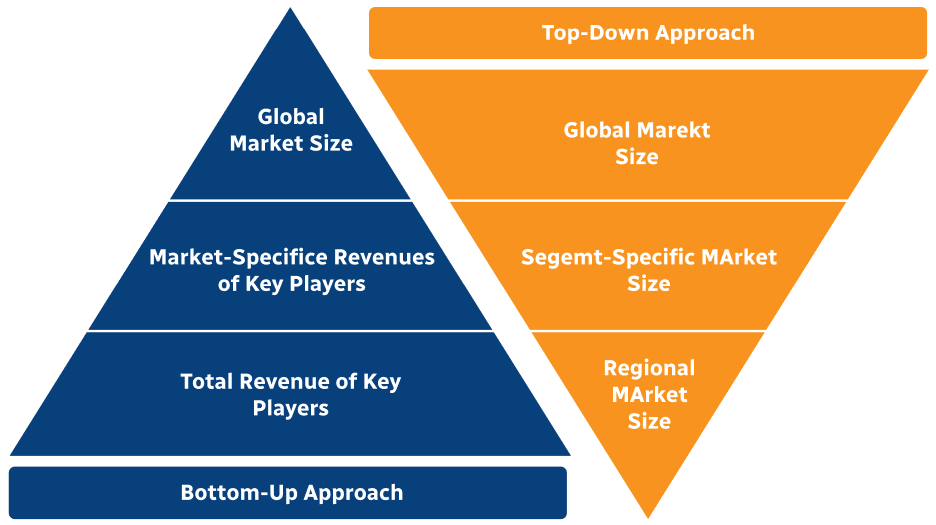
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


