Key Insights
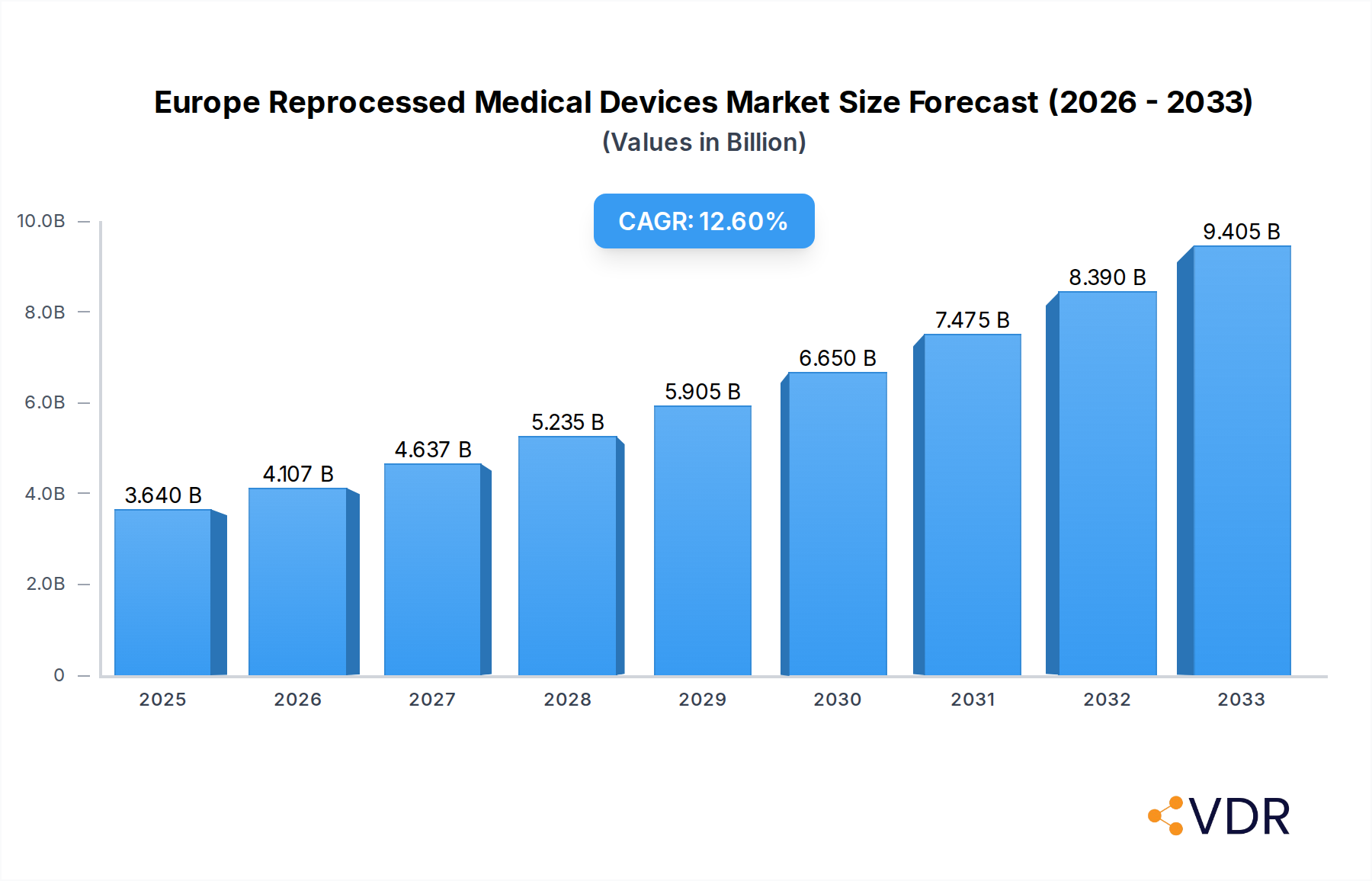
The Europe Reprocessed Medical Devices Market is poised for significant expansion, with a current market size estimated at USD 3.64 billion in 2025. This robust growth is driven by a projected Compound Annual Growth Rate (CAGR) of 12.7% throughout the forecast period of 2025-2033. This upward trajectory is primarily fueled by increasing cost-containment pressures within healthcare systems across Europe, compelling hospitals and healthcare providers to seek more economical alternatives to single-use devices. Furthermore, a growing environmental consciousness and a desire to reduce medical waste are contributing to the adoption of reprocessed medical devices, aligning with European sustainability goals. The expanding range of compatible devices, coupled with advancements in reprocessing technologies that ensure safety and efficacy, also plays a crucial role in market expansion. Regulatory bodies are also increasingly providing clear guidelines for reprocessing, fostering greater trust and acceptance among healthcare professionals and institutions.
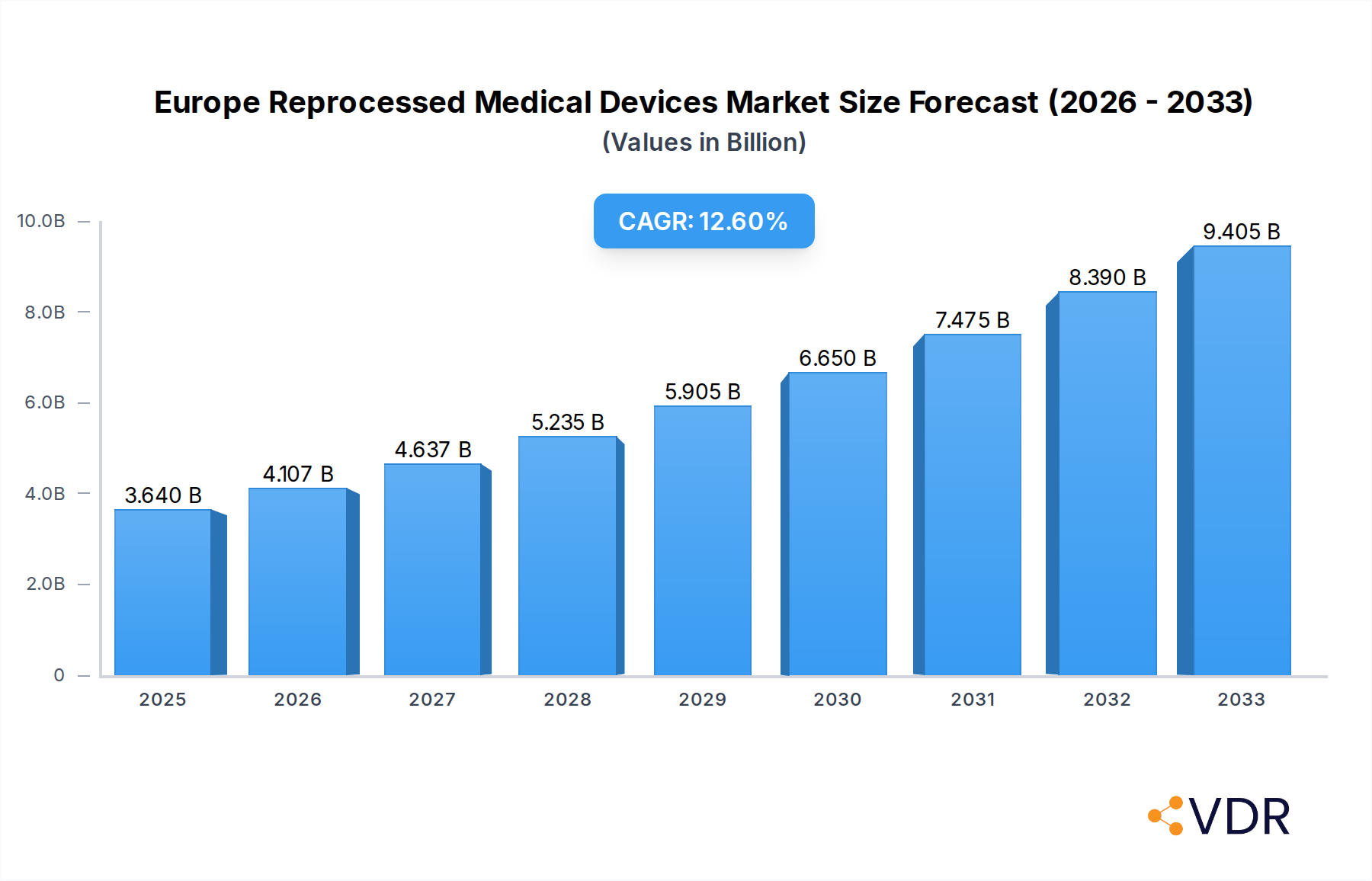
Europe Reprocessed Medical Devices Market Market Size (In Billion)

The market segmentation reveals a strong performance across both Class I and Class II reprocessed medical devices. Class II devices, which include critical items like pulse oximeter sensors, sequential compression sleeves, and catheters and guidewires, are expected to witness substantial demand due to their widespread use in various medical procedures and the significant cost savings achievable through reprocessing. Key players such as Stryker Corporation, Cardinal Health (Sustainable Technologies), and STERIS are actively investing in R&D and expanding their reprocessing capabilities, further intensifying market competition and innovation. Geographically, Western European nations like Germany, the United Kingdom, and France are anticipated to lead the market, owing to their well-established healthcare infrastructure and a proactive approach to adopting cost-effective and sustainable medical solutions. The rest of Europe is also showing promising growth potential as awareness and acceptance of reprocessed medical devices increase.
Europe Reprocessed Medical Devices Market Company Market Share

This report offers an in-depth analysis of the Europe Reprocessed Medical Devices Market, encompassing a detailed examination of market dynamics, growth trends, regional dominance, product landscape, key players, and future outlook. Leveraging extensive research and predictive modeling, this report provides actionable insights for stakeholders seeking to navigate and capitalize on the evolving landscape of sustainable medical device solutions. The study period spans from 2019 to 2033, with a base year of 2025 and a forecast period from 2025 to 2033, building upon historical data from 2019 to 2024. All values are presented in billions of units where applicable.
Europe Reprocessed Medical Devices Market Market Dynamics & Structure
The Europe Reprocessed Medical Devices Market is characterized by a moderately concentrated structure, with a few dominant players and a growing number of specialized firms. Technological innovation is a significant driver, focusing on advanced sterilization techniques, quality control, and traceability systems that enhance the safety and efficacy of reprocessed devices. The regulatory framework, while evolving, presents a crucial element influencing market entry and operational standards. The European Union's Medical Device Regulation (MDR) significantly impacts the reprocessing sector, demanding stringent adherence to quality management systems and post-market surveillance. Competitive product substitutes, primarily new single-use devices, pose a constant challenge, although the cost-effectiveness and environmental benefits of reprocessing are gaining traction. End-user demographics, including hospitals, clinics, and surgical centers, are increasingly driven by budget constraints and a growing awareness of sustainability. Mergers and acquisitions (M&A) are a key trend, with larger entities acquiring specialized reprocessing companies to expand their service offerings and market reach.
- Market Concentration: Moderately concentrated, with key players like STERIS, Cardinal Health, and Medline Industries Inc. holding significant market share.
- Technological Innovation Drivers: Advancements in cleaning, disinfection, sterilization technologies, and digital tracking systems for enhanced safety and compliance.
- Regulatory Frameworks: EU MDR and national regulations impose strict quality and safety standards, influencing operational processes and market access.
- Competitive Product Substitutes: New single-use medical devices remain a primary substitute, necessitating a strong value proposition for reprocessed alternatives.
- End-User Demographics: Hospitals and healthcare systems are the primary end-users, driven by cost savings and increasing corporate social responsibility mandates.
- M&A Trends: Strategic acquisitions by larger medical device companies and dedicated reprocessing firms to consolidate market share and expand capabilities.
Europe Reprocessed Medical Devices Market Growth Trends & Insights
The Europe Reprocessed Medical Devices Market is poised for robust growth, driven by a confluence of economic, environmental, and regulatory factors. The estimated market size for reprocessed medical devices in Europe is projected to reach $8.5 billion by 2025, with a projected Compound Annual Growth Rate (CAGR) of 12.5% from 2025 to 2033. This upward trajectory is fueled by increasing healthcare expenditure across the continent, coupled with a strong imperative to reduce healthcare costs. Reprocessed medical devices offer a significant cost advantage over their single-use counterparts, making them an attractive option for budget-conscious healthcare providers. Furthermore, a heightened global focus on environmental sustainability is compelling healthcare institutions to adopt more circular economy practices, with medical device reprocessing emerging as a key strategy for waste reduction.
The adoption rates of reprocessed medical devices are steadily increasing as regulatory bodies and healthcare professionals gain greater confidence in the safety and efficacy of third-party reprocessing services. Technological disruptions in sterilization and validation methods are continuously enhancing the quality and reliability of reprocessed devices, addressing historical concerns about contamination and performance. Consumer behavior shifts are also playing a crucial role, with an increasing number of healthcare organizations actively seeking out sustainable procurement options. The market penetration of reprocessed medical devices, while still lower than in North America, is expanding as awareness grows and regulatory hurdles are better understood and navigated.
Specific metrics highlight this growth: The market for reprocessed Class II devices, which include high-volume items like pulse oximeter sensors and sequential compression sleeves, is expected to witness particularly strong demand, driven by their frequent use and substantial cost savings achievable through reprocessing. Innovations in diagnostic and monitoring devices reprocessing are also contributing to market expansion.
The integration of digital technologies for tracking and quality assurance further bolsters confidence in the reprocessed medical device supply chain, making it more transparent and accountable. This growing acceptance, combined with the inherent economic and environmental benefits, underpins the positive growth outlook for the Europe Reprocessed Medical Devices Market. The market's capacity to deliver substantial cost savings without compromising patient safety is a cornerstone of its expanding adoption.
Dominant Regions, Countries, or Segments in Europe Reprocessed Medical Devices Market
Within the Europe Reprocessed Medical Devices Market, Germany emerges as the dominant country, showcasing the highest market share and sustained growth potential. This leadership is attributed to a robust healthcare infrastructure, a strong emphasis on cost containment in public healthcare systems, and a proactive regulatory environment that, while stringent, facilitates clear pathways for approved reprocessing operations. Germany's economic strength and high volume of medical procedures contribute to a substantial demand for medical devices, both new and reprocessed.
The dominance of Germany is further amplified by its extensive network of hospitals and specialized healthcare facilities that are increasingly adopting sustainable procurement strategies. The country’s commitment to environmental policies also plays a crucial role in driving the adoption of reprocessed medical devices as a means of waste reduction. Furthermore, the presence of leading reprocessing companies and a skilled workforce adept at handling complex sterilization and validation processes solidify Germany’s position.
Among the device types, Class II Devices are currently driving the market, representing a larger share due to their higher volume of usage in various medical procedures and their significant cost-saving potential when reprocessed. This segment includes critical items such as:
- Pulse Oximeter Sensors: Frequently used across various care settings, reprocessing offers substantial cost efficiencies.
- Sequential Compression Sleeves: Essential for preventing deep vein thrombosis, their widespread use makes them prime candidates for reprocessing.
- Catheters and Guidewires: While requiring highly specialized reprocessing, the high cost of new devices makes reprocessing a financially attractive option for healthcare providers.
- Other Class II Devices: This broad category encompasses a multitude of instruments and devices where reprocessing can yield considerable savings.
The United Kingdom follows closely, with its National Health Service (NHS) actively exploring cost-saving measures. However, regulatory advisories, such as the one issued by the Medicines and Healthcare products Regulatory Agency (MHRA) in June 2022, advising against reprocessing, have introduced some market uncertainty, although the underlying economic drivers for reprocessing remain strong.
France and the Nordic countries also represent significant markets, driven by similar economic pressures and a growing environmental consciousness. The recent regulatory shift in Poland, allowing the reprocessing of single-use medical devices, signifies a positive development for market expansion in Eastern Europe, potentially unlocking new growth opportunities.
The growth in the Class II segment is further propelled by technological advancements in reprocessing techniques that enhance the safety and efficacy of these devices, making them more palatable for wider adoption. The combined economic benefits, environmental considerations, and expanding regulatory acceptance are key factors propelling the dominance of Germany and the Class II device segment in the Europe Reprocessed Medical Devices Market.
Europe Reprocessed Medical Devices Market Product Landscape
The product landscape of the Europe Reprocessed Medical Devices Market is characterized by a growing diversity of device types that can be safely and effectively reprocessed. Leading reprocessors focus on optimizing their processes for common instruments such as surgical graspers, scalpels, tourniquet cuffs, pulse oximeter sensors, sequential compression sleeves, and various types of catheters and guidewires. The primary emphasis is on ensuring that reprocessed devices meet or exceed the original manufacturer's specifications for performance, safety, and sterility. Unique selling propositions often revolve around advanced sterilization technologies, rigorous quality control protocols, and comprehensive traceability systems that provide end-users with absolute confidence in the reprocessed products. Technological advancements continually expand the scope of reprocessable devices, making the market more dynamic and offering greater cost-saving opportunities across a wider array of medical equipment.
Key Drivers, Barriers & Challenges in Europe Reprocessed Medical Devices Market
Key Drivers:
- Cost Savings: Reprocessing offers significant cost reductions for healthcare facilities compared to purchasing new single-use devices, a crucial factor in budget-constrained healthcare systems.
- Environmental Sustainability: Growing awareness of healthcare's environmental footprint drives demand for reprocessing as a means to reduce medical waste and conserve resources.
- Regulatory Support (Evolving): While challenging, the ongoing clarification and potential adaptation of regulations, as seen in Poland, can foster market growth.
- Technological Advancements: Innovations in sterilization, cleaning, and validation technologies enhance the safety and reliability of reprocessed devices.
Barriers & Challenges:
- Stringent Regulatory Landscape: The complexity and evolving nature of regulations, such as the EU MDR, create significant compliance hurdles and investment requirements for reprocessors.
- Perception and Trust: Lingering concerns among some healthcare professionals and patients regarding the safety and efficacy of reprocessed devices can impede adoption.
- Competition from New Devices: The continuous innovation and availability of new single-use devices present a constant competitive pressure.
- Supply Chain Disruptions: Global supply chain issues can affect the availability of necessary chemicals, equipment, and skilled labor required for reprocessing.
- Initial Investment Costs: Setting up and maintaining a certified reprocessing facility requires substantial capital investment.
- Limited Scope of Reprocessable Devices: Not all single-use medical devices are suitable for reprocessing, limiting the overall market potential.
Emerging Opportunities in Europe Reprocessed Medical Devices Market
Emerging opportunities in the Europe Reprocessed Medical Devices Market lie in the expansion of reprocessing capabilities for more complex and specialized medical devices, such as certain types of endoscopic instruments and advanced surgical tools. Untapped markets in Eastern and Southern Europe, where awareness and infrastructure for reprocessing are still developing, present significant growth potential. Evolving consumer preferences for sustainable healthcare solutions are creating demand for transparent and certified reprocessing services. Furthermore, partnerships between reprocessing companies and original equipment manufacturers (OEMs) could unlock new avenues for device take-back programs and streamlined reprocessing workflows, driving innovation and market penetration.
Growth Accelerators in the Europe Reprocessed Medical Devices Market Industry
Key growth accelerators for the Europe Reprocessed Medical Devices Market include the increasing emphasis on circular economy principles within healthcare, driven by both governmental policies and institutional sustainability goals. Technological breakthroughs in automated cleaning and advanced sterilization techniques, such as low-temperature sterilization methods, are expanding the range of reprocessable devices and improving efficiency. Strategic partnerships between reprocessing firms and large hospital networks, as well as the consolidation of smaller players by established market leaders, are creating economies of scale and enhancing market reach. Furthermore, proactive engagement with regulatory bodies to clarify and potentially streamline approval processes for reprocessed devices will be critical in unlocking further market potential.
Key Players Shaping the Europe Reprocessed Medical Devices Market Market
- Arjo
- Sterilmed Inc (Johnson & Johnson)
- Wassenburg Medical BV
- NEScientific
- Stryker Corporation
- MATACHANA
- Cardinal Health (Sustainable Technologies)
- Medline Industries Inc
- Vanguard
- STERIS
Notable Milestones in Europe Reprocessed Medical Devices Market Sector
- June 2022: The Medicines and Healthcare products Regulatory Agency of the United Kingdom advised against the reprocessing of single-use medical devices in its guidance, creating market uncertainty in the UK.
- May 2022: The President of Poland signed the Act on Medical Devices. Under the new law, reprocessing of single-use medical devices has been allowed in Poland, marking a significant positive regulatory shift for the Eastern European market.
In-Depth Europe Reprocessed Medical Devices Market Market Outlook
The future outlook for the Europe Reprocessed Medical Devices Market is exceptionally positive, driven by a persistent demand for cost-effective healthcare solutions and an escalating commitment to environmental sustainability. Growth accelerators such as advanced sterilization technologies, stringent quality controls, and increasing regulatory clarity are poised to fuel market expansion. Strategic initiatives by key players, including capacity expansions and the development of new reprocessing services, will further solidify market growth. The inherent economic advantages and environmental benefits of reprocessing medical devices position it as a vital component of a more sustainable and accessible European healthcare system, indicating significant untapped potential and robust future growth prospects.
Europe Reprocessed Medical Devices Market Segmentation
-
1. Device Type
-
1.1. Class I Devices
- 1.1.1. Laparoscopic Graspers
- 1.1.2. Scalpels
- 1.1.3. Tourniquet Cuffs
- 1.1.4. Other Class I Devices
-
1.2. Class II Devices
- 1.2.1. Pulse Oximeter Sensors
- 1.2.2. Sequential Compression Sleeves
- 1.2.3. Catheters and Guidewires
- 1.2.4. Other Class II Devices
-
1.1. Class I Devices
Europe Reprocessed Medical Devices Market Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe
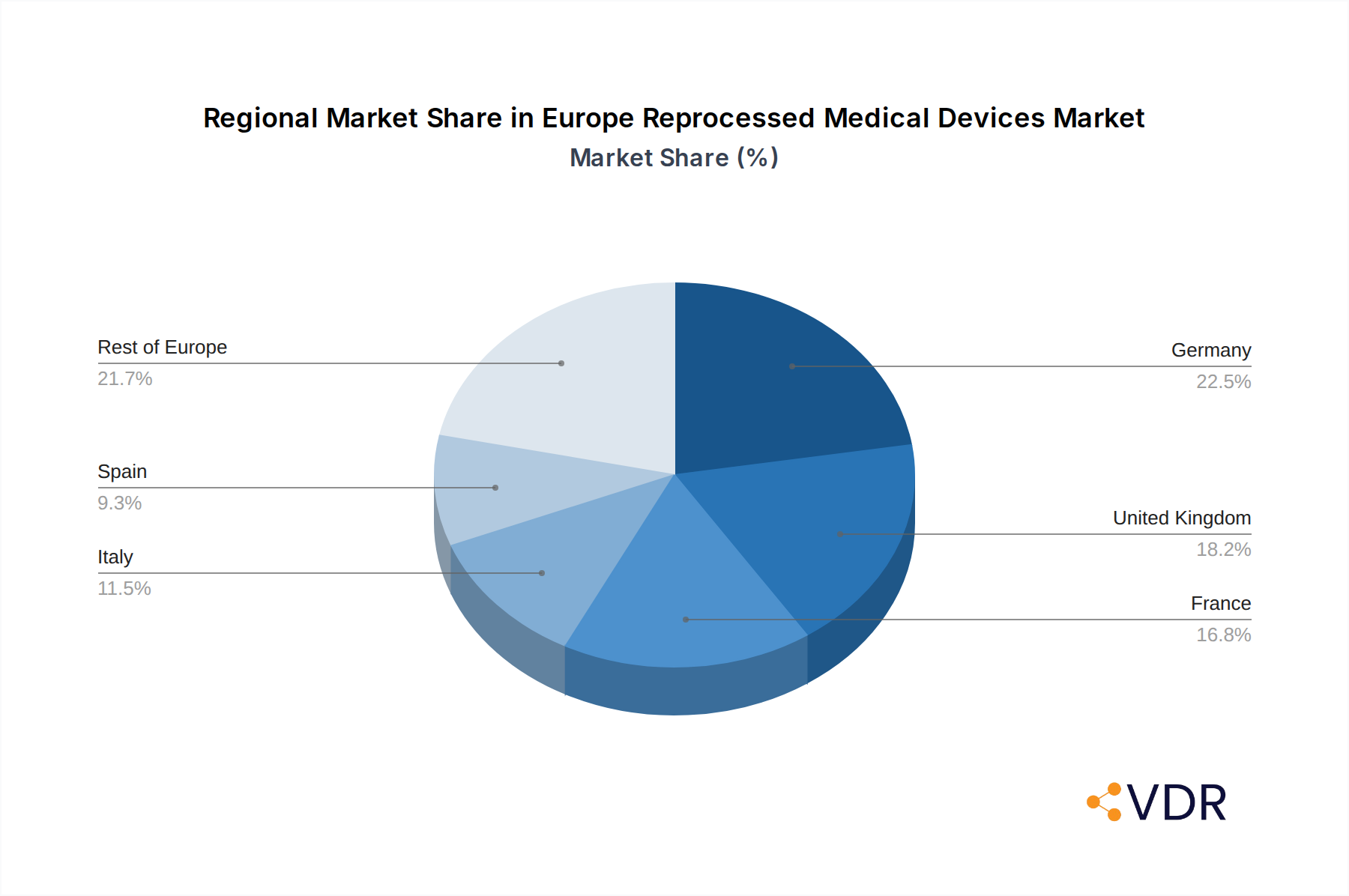
Europe Reprocessed Medical Devices Market Regional Market Share

Geographic Coverage of Europe Reprocessed Medical Devices Market
Europe Reprocessed Medical Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.7% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 5.1.1. Class I Devices
- 5.1.1.1. Laparoscopic Graspers
- 5.1.1.2. Scalpels
- 5.1.1.3. Tourniquet Cuffs
- 5.1.1.4. Other Class I Devices
- 5.1.2. Class II Devices
- 5.1.2.1. Pulse Oximeter Sensors
- 5.1.2.2. Sequential Compression Sleeves
- 5.1.2.3. Catheters and Guidewires
- 5.1.2.4. Other Class II Devices
- 5.1.1. Class I Devices
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Germany
- 5.2.2. United Kingdom
- 5.2.3. France
- 5.2.4. Italy
- 5.2.5. Spain
- 5.2.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 6. Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 6.1.1. Class I Devices
- 6.1.1.1. Laparoscopic Graspers
- 6.1.1.2. Scalpels
- 6.1.1.3. Tourniquet Cuffs
- 6.1.1.4. Other Class I Devices
- 6.1.2. Class II Devices
- 6.1.2.1. Pulse Oximeter Sensors
- 6.1.2.2. Sequential Compression Sleeves
- 6.1.2.3. Catheters and Guidewires
- 6.1.2.4. Other Class II Devices
- 6.1.1. Class I Devices
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 7. Germany Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Device Type
- 7.1.1. Class I Devices
- 7.1.1.1. Laparoscopic Graspers
- 7.1.1.2. Scalpels
- 7.1.1.3. Tourniquet Cuffs
- 7.1.1.4. Other Class I Devices
- 7.1.2. Class II Devices
- 7.1.2.1. Pulse Oximeter Sensors
- 7.1.2.2. Sequential Compression Sleeves
- 7.1.2.3. Catheters and Guidewires
- 7.1.2.4. Other Class II Devices
- 7.1.1. Class I Devices
- 7.1. Market Analysis, Insights and Forecast - by Device Type
- 8. United Kingdom Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Device Type
- 8.1.1. Class I Devices
- 8.1.1.1. Laparoscopic Graspers
- 8.1.1.2. Scalpels
- 8.1.1.3. Tourniquet Cuffs
- 8.1.1.4. Other Class I Devices
- 8.1.2. Class II Devices
- 8.1.2.1. Pulse Oximeter Sensors
- 8.1.2.2. Sequential Compression Sleeves
- 8.1.2.3. Catheters and Guidewires
- 8.1.2.4. Other Class II Devices
- 8.1.1. Class I Devices
- 8.1. Market Analysis, Insights and Forecast - by Device Type
- 9. France Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Device Type
- 9.1.1. Class I Devices
- 9.1.1.1. Laparoscopic Graspers
- 9.1.1.2. Scalpels
- 9.1.1.3. Tourniquet Cuffs
- 9.1.1.4. Other Class I Devices
- 9.1.2. Class II Devices
- 9.1.2.1. Pulse Oximeter Sensors
- 9.1.2.2. Sequential Compression Sleeves
- 9.1.2.3. Catheters and Guidewires
- 9.1.2.4. Other Class II Devices
- 9.1.1. Class I Devices
- 9.1. Market Analysis, Insights and Forecast - by Device Type
- 10. Italy Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Device Type
- 10.1.1. Class I Devices
- 10.1.1.1. Laparoscopic Graspers
- 10.1.1.2. Scalpels
- 10.1.1.3. Tourniquet Cuffs
- 10.1.1.4. Other Class I Devices
- 10.1.2. Class II Devices
- 10.1.2.1. Pulse Oximeter Sensors
- 10.1.2.2. Sequential Compression Sleeves
- 10.1.2.3. Catheters and Guidewires
- 10.1.2.4. Other Class II Devices
- 10.1.1. Class I Devices
- 10.1. Market Analysis, Insights and Forecast - by Device Type
- 11. Spain Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Device Type
- 11.1.1. Class I Devices
- 11.1.1.1. Laparoscopic Graspers
- 11.1.1.2. Scalpels
- 11.1.1.3. Tourniquet Cuffs
- 11.1.1.4. Other Class I Devices
- 11.1.2. Class II Devices
- 11.1.2.1. Pulse Oximeter Sensors
- 11.1.2.2. Sequential Compression Sleeves
- 11.1.2.3. Catheters and Guidewires
- 11.1.2.4. Other Class II Devices
- 11.1.1. Class I Devices
- 11.1. Market Analysis, Insights and Forecast - by Device Type
- 12. Rest of Europe Europe Reprocessed Medical Devices Market Analysis, Insights and Forecast, 2021-2033
- 12.1. Market Analysis, Insights and Forecast - by Device Type
- 12.1.1. Class I Devices
- 12.1.1.1. Laparoscopic Graspers
- 12.1.1.2. Scalpels
- 12.1.1.3. Tourniquet Cuffs
- 12.1.1.4. Other Class I Devices
- 12.1.2. Class II Devices
- 12.1.2.1. Pulse Oximeter Sensors
- 12.1.2.2. Sequential Compression Sleeves
- 12.1.2.3. Catheters and Guidewires
- 12.1.2.4. Other Class II Devices
- 12.1.1. Class I Devices
- 12.1. Market Analysis, Insights and Forecast - by Device Type
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Arjo
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Sterilmed Inc (Johnson & Johnson)
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Wassenburg Medical BV
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 NEScientific
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Stryker Corporation
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 MATACHANA
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Cardinal Health (Sustainable Technologies)
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Medline Industries Inc
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Vanguard
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 STERIS
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.1 Arjo
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Europe Reprocessed Medical Devices Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Europe Reprocessed Medical Devices Market Share (%) by Company 2025
List of Tables
- Table 1: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 2: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 3: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 5: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 6: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 7: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 8: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 9: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 10: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 11: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 13: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 14: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 15: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 16: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 18: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 19: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 20: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 21: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 22: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 23: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 26: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Device Type 2020 & 2033
- Table 27: Europe Reprocessed Medical Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 28: Europe Reprocessed Medical Devices Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Europe Reprocessed Medical Devices Market?
The projected CAGR is approximately 12.7%.
2. Which companies are prominent players in the Europe Reprocessed Medical Devices Market?
Key companies in the market include Arjo, Sterilmed Inc (Johnson & Johnson), Wassenburg Medical BV, NEScientific, Stryker Corporation, MATACHANA, Cardinal Health (Sustainable Technologies), Medline Industries Inc, Vanguard, STERIS.
3. What are the main segments of the Europe Reprocessed Medical Devices Market?
The market segments include Device Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 3.64 billion as of 2022.
5. What are some drivers contributing to market growth?
Cost Savings Through Reprocessing Single-use Devices; Regulatory Pressure to Reduce Volume of Medical Waste.
6. What are the notable trends driving market growth?
Scalpels Segment is Poised to Register Significant Growth During the Forecast Period.
7. Are there any restraints impacting market growth?
Potential of Material Alteration and Cross Infection with Reprocessed Device; Preconceived Notions Regarding the Quality of Reprocessed Single-use Medical Devices (SUDs).
8. Can you provide examples of recent developments in the market?
June 2022: The Medicines and Healthcare products Regulatory Agency of the United Kingdom advised against the reprocessing of single-use medical devices in its guidance.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Europe Reprocessed Medical Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Europe Reprocessed Medical Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Europe Reprocessed Medical Devices Market?
To stay informed about further developments, trends, and reports in the Europe Reprocessed Medical Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


