Key Insights
The Clinical Grade Wearable Device market is poised for remarkable expansion, projected to reach a significant USD 28.92 billion in 2025, exhibiting a robust Compound Annual Growth Rate (CAGR) of 17.5% through 2033. This impressive growth is fueled by an increasing global focus on proactive health management, the escalating prevalence of chronic diseases, and the continuous advancements in sensor technology and miniaturization. The growing acceptance of remote patient monitoring by healthcare providers, driven by its ability to improve patient outcomes and reduce healthcare costs, further accentuates the market's upward trajectory. Furthermore, the burgeoning demand for personalized health insights and early disease detection among consumers is a pivotal driver, pushing the adoption of sophisticated wearable devices capable of delivering medical-grade accuracy.
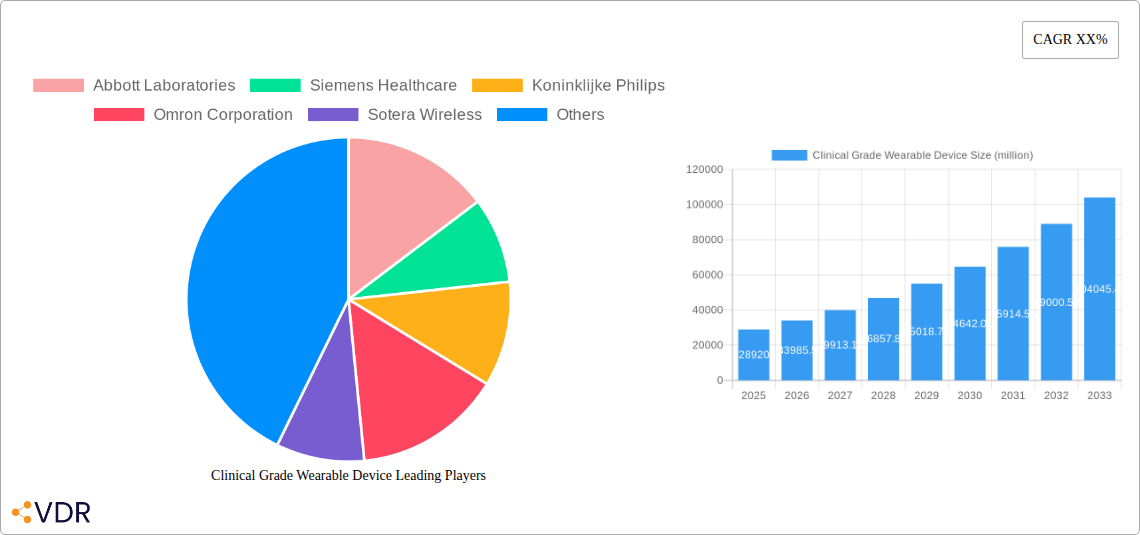
Clinical Grade Wearable Device Market Size (In Billion)

The market is segmented into key applications including advanced detection and precise treatment solutions, alongside other critical uses. Within types, diagnostic devices are expected to lead, supported by a strong pipeline of therapeutic devices and emerging innovative solutions. Leading companies such as Abbott Laboratories, Siemens Healthcare, Koninklijke Philips, and Medtronic are at the forefront, investing heavily in research and development to introduce next-generation clinical-grade wearables. Geographically, North America is anticipated to maintain a dominant market share, owing to its well-established healthcare infrastructure, high disposable incomes, and early adoption of innovative medical technologies. However, the Asia Pacific region is expected to witness the fastest growth, propelled by rising healthcare expenditures, increasing awareness of chronic disease management, and a burgeoning patient population. The primary drivers for this expansion include the integration of AI and machine learning for enhanced data analysis, the development of non-invasive monitoring techniques, and the increasing regulatory support for digital health solutions, all contributing to a more proactive and personalized healthcare landscape.
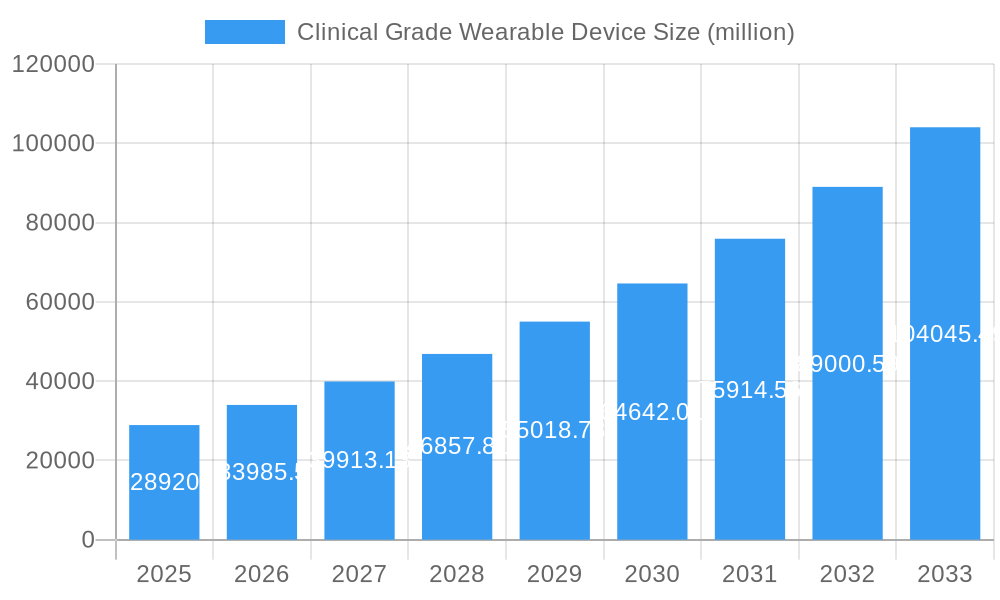
Clinical Grade Wearable Device Company Market Share

This comprehensive report delves into the dynamic clinical grade wearable device market, a rapidly expanding sector poised for significant growth. With an estimated market size of $XX billion in 2025, this industry is revolutionizing healthcare delivery by enabling continuous, accurate, and actionable health data collection outside traditional clinical settings. This report provides an in-depth analysis of market trends, growth drivers, challenges, and key players, offering invaluable insights for stakeholders across the parent market of Digital Health and the child market of Remote Patient Monitoring devices.
Clinical Grade Wearable Device Market Dynamics & Structure
The clinical grade wearable device market is characterized by a moderate level of concentration, with a few prominent players like Abbott Laboratories, Siemens Healthcare, and Koninklijke Philips holding significant market share. However, the landscape is increasingly populated by innovative startups such as Sotera Wireless, Everist Health, and Vital Connect, driving intense competition and rapid technological advancement. Key drivers for market growth include the increasing prevalence of chronic diseases, the growing demand for personalized healthcare solutions, and the supportive regulatory environment for medical devices.
- Technological Innovation Drivers: Advancements in sensor technology, miniaturization, AI-powered data analytics, and secure data transmission are continuously enhancing the capabilities of clinical grade wearables. Features like real-time anomaly detection and predictive analytics are becoming standard.
- Regulatory Frameworks: Stringent regulatory approvals from bodies like the FDA and EMA are crucial for market entry and product credibility, ensuring patient safety and data integrity.
- Competitive Product Substitutes: While direct substitutes are limited, traditional diagnostic methods and infrequent in-clinic monitoring represent indirect competition. However, the continuous monitoring capabilities of wearables offer a clear advantage.
- End-User Demographics: The primary end-users include healthcare providers, research institutions, and increasingly, direct-to-consumer markets for chronic condition management and preventative health. Aging populations and tech-savvy younger demographics are key target segments.
- M&A Trends: The market has witnessed strategic acquisitions as larger companies seek to expand their digital health portfolios and incorporate cutting-edge wearable technologies. For instance, XX significant M&A deals were recorded in the historical period (2019-2024), with an estimated value of $XX billion.
Clinical Grade Wearable Device Growth Trends & Insights
The clinical grade wearable device market is experiencing robust growth, projected to expand at a Compound Annual Growth Rate (CAGR) of XX% from 2025 to 2033. The estimated market size is expected to reach $XX billion by 2033, a significant leap from the $XX billion in 2025. This growth is fueled by a confluence of factors, including the accelerating adoption of telehealth and remote patient monitoring (RPM) programs, a growing emphasis on preventative care, and the increasing sophistication of wearable technology itself. Consumer behavior is shifting towards proactive health management, with individuals becoming more willing to embrace wearable devices for continuous health tracking and early disease detection.
Technological disruptions are playing a pivotal role, with the integration of advanced sensors capable of monitoring a wider range of physiological parameters with greater accuracy. This includes continuous glucose monitoring (CGM) devices from companies like Dexcom, advanced ECG and heart rate monitoring, and even non-invasive blood pressure estimation. The development of AI and machine learning algorithms is further enhancing the value proposition by enabling sophisticated data analysis, providing actionable insights to both patients and clinicians, and predicting potential health events before they become critical.
The market penetration of clinical grade wearables is steadily increasing, particularly within specialized applications like cardiology, diabetes management, and post-operative care. Healthcare providers are increasingly recognizing the benefits of these devices in improving patient outcomes, reducing hospital readmissions, and optimizing resource allocation. The ability to collect real-time data allows for more timely interventions, personalized treatment plans, and a deeper understanding of individual patient health trajectories. Furthermore, the growing comfort level of consumers with connected health devices, driven by the mainstream adoption of consumer wearables, is paving the way for wider acceptance of clinical-grade solutions. This paradigm shift towards continuous health monitoring is not merely a trend but a fundamental transformation in how healthcare is delivered and experienced.
Dominant Regions, Countries, or Segments in Clinical Grade Wearable Device
The North America region currently stands as a dominant force in the clinical grade wearable device market, driven by a robust healthcare infrastructure, high patient awareness regarding chronic disease management, and significant investments in digital health technologies. The United States, in particular, leads the charge due to favorable reimbursement policies for remote patient monitoring services and a strong presence of leading medical device manufacturers and innovative startups. The Application: Detect segment is also a significant growth driver, with an estimated market share of XX% in 2025.
North America's Dominance:
- Economic Policies: Favorable reimbursement structures for telehealth and remote patient monitoring (RPM) services encourage widespread adoption by healthcare providers.
- Infrastructure: Advanced healthcare systems and widespread internet connectivity support the seamless integration of wearable data into clinical workflows.
- Technological Adoption: High consumer adoption rates for health-tracking devices and a strong research and development ecosystem foster innovation.
- Market Share: North America is projected to hold approximately XX% of the global clinical grade wearable device market share in 2025.
Application: Detect Segment Growth:
- Early Disease Identification: Wearables are increasingly utilized for early detection of conditions such as atrial fibrillation (AFib), sleep apnea, and changes in vital signs indicative of potential health deterioration.
- Proactive Health Management: Devices that offer continuous monitoring for conditions like diabetes (e.g., Dexcom's CGM) empower patients to manage their health proactively.
- Research and Clinical Trials: The precision and continuous data provided by these devices are invaluable for medical research and clinical trials.
- Growth Potential: This segment is expected to witness a CAGR of XX% during the forecast period, outpacing other applications.
Types: Diagnostic Devices Dominance:
- Diagnostic Capabilities: Within the "Types" segment, Diagnostic Devices are leading the market, comprising an estimated XX% of the market share in 2025. These devices are crucial for continuous monitoring and early identification of physiological anomalies.
- Versatility: Diagnostic wearables can track a wide array of parameters, including heart rate, ECG, blood oxygen saturation (SpO2), respiration rate, and body temperature, providing comprehensive health insights.
- Integration with Healthcare Systems: Their ability to integrate with electronic health records (EHRs) makes them invaluable for healthcare providers.
The strong emphasis on preventive healthcare and the increasing burden of chronic diseases globally continue to propel the growth of clinical grade wearable devices, with regions like Europe and Asia-Pacific showing substantial potential for future expansion.
Clinical Grade Wearable Device Product Landscape
The clinical grade wearable device product landscape is defined by a rapid evolution of sophisticated sensor technologies, enhanced data analytics capabilities, and user-centric designs. Innovations focus on improving accuracy, miniaturization, and the ability to monitor a wider spectrum of physiological parameters continuously. For instance, the development of non-invasive sensors for blood pressure and continuous glucose monitoring are transforming chronic disease management. These devices are not just tracking data but are increasingly providing actionable insights through AI-powered platforms, offering predictive analytics and personalized health recommendations. The integration with cloud-based platforms ensures secure data storage and seamless sharing with healthcare providers, further solidifying their role in remote patient monitoring and telehealth.
Key Drivers, Barriers & Challenges in Clinical Grade Wearable Device
Key Drivers: The clinical grade wearable device market is propelled by several critical factors. The burgeoning global prevalence of chronic diseases, such as cardiovascular conditions and diabetes, necessitates continuous monitoring and proactive management, for which wearables are ideally suited. Supportive government initiatives and increasing healthcare expenditure aimed at improving patient outcomes and reducing healthcare costs are also significant catalysts. Furthermore, rapid technological advancements in sensor technology, artificial intelligence, and miniaturization are enhancing device functionality and accuracy. The growing consumer demand for personalized healthcare solutions and the increasing acceptance of telehealth services are also major drivers.
Barriers & Challenges: Despite the strong growth trajectory, the market faces certain challenges. High upfront costs for advanced clinical grade devices can be a barrier to widespread adoption, particularly in cost-sensitive markets. Stringent regulatory approvals, while essential for patient safety, can be time-consuming and complex, potentially delaying market entry. Data privacy and security concerns remain paramount, requiring robust measures to protect sensitive patient information. Interoperability issues between different devices and healthcare systems can also hinder seamless data integration. Finally, physician and patient education regarding the accurate interpretation and utilization of wearable data is crucial for maximizing their clinical utility and overcoming potential skepticism. The supply chain disruptions witnessed in recent years can also impact production and availability.
Emerging Opportunities in Clinical Grade Wearable Device
Emerging opportunities in the clinical grade wearable device sector are vast and multifaceted. The integration of these devices into comprehensive telehealth platforms presents a significant avenue for growth, enabling seamless remote patient monitoring and virtual consultations. Untapped markets in emerging economies, where the demand for accessible healthcare solutions is high, offer substantial expansion potential. The development of wearables for specialized applications, such as mental health monitoring, early detection of infectious diseases, and personalized rehabilitation programs, represents innovative frontiers. Furthermore, the increasing focus on preventative healthcare and wellness is driving demand for consumer-grade wearables with advanced health-tracking capabilities, blurring the lines between consumer and clinical devices and opening avenues for hybrid solutions. The use of AI for predictive analytics and personalized intervention strategies is another key opportunity.
Growth Accelerators in the Clinical Grade Wearable Device Industry
Several key accelerators are driving the long-term growth of the clinical grade wearable device industry. Technological breakthroughs in areas like bio-sensing, materials science for enhanced comfort and durability, and advanced AI algorithms for real-time data interpretation are continuously pushing the boundaries of what these devices can achieve. Strategic partnerships between wearable technology manufacturers, healthcare providers, pharmaceutical companies, and insurance providers are crucial for developing integrated care pathways and expanding market reach. Government incentives, favorable reimbursement policies for remote patient monitoring, and increasing investment in digital health infrastructure are also significant growth boosters. Moreover, the growing body of clinical evidence demonstrating the efficacy of wearables in improving patient outcomes and reducing healthcare costs is further accelerating adoption and investment.
Key Players Shaping the Clinical Grade Wearable Device Market
- Abbott Laboratories
- Siemens Healthcare
- Koninklijke Philips
- Omron Corporation
- Sotera Wireless
- Everist Health
- Polar Electro
- Zephyr Technology
- Intelesens
- Fitbit
- LifeWatch
- Garmin
- Contec Medical Systems
- Vital Connect
- Dexcom
- Medtronic
- Ge Healthcare
Notable Milestones in Clinical Grade Wearable Device Sector
- 2019: FDA clearance for expanded indications for atrial fibrillation detection in wearable devices, increasing clinical utility.
- 2020: Significant surge in remote patient monitoring (RPM) adoption driven by the COVID-19 pandemic, accelerating investment in wearable solutions.
- 2021: Launch of next-generation continuous glucose monitors (CGMs) with improved accuracy and user experience, impacting diabetes management.
- 2022: Increased investment in AI and machine learning integration for predictive analytics in wearable health data.
- 2023: Growing focus on non-invasive blood pressure monitoring wearables entering clinical trials and seeking regulatory approvals.
- 2023: Expansion of wearable device use in post-operative care and rehabilitation to reduce hospital readmissions.
- 2024: Introduction of advanced sleep tracking and analysis features in clinical grade wearables for diagnosing sleep disorders.
In-Depth Clinical Grade Wearable Device Market Outlook
The outlook for the clinical grade wearable device market is exceptionally promising, driven by persistent technological innovation and a global imperative for more efficient and personalized healthcare. Growth accelerators include the deepening integration of AI for predictive diagnostics and personalized treatment recommendations, the expansion of these devices into new therapeutic areas such as mental health and infectious disease monitoring, and the strategic collaborations that are creating comprehensive digital health ecosystems. The market is poised for substantial expansion as reimbursement models continue to evolve and as both healthcare providers and consumers recognize the immense value of continuous, actionable health data. The projected market size by 2033 signifies a transformative shift towards proactive, data-driven healthcare facilitated by these advanced wearable technologies.
Clinical Grade Wearable Device Segmentation
-
1. Application
- 1.1. Detect
- 1.2. Treatment
- 1.3. Others
-
2. Types
- 2.1. Diagnostic Devices
- 2.2. Therapeutic Devices
- 2.3. Others
Clinical Grade Wearable Device Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
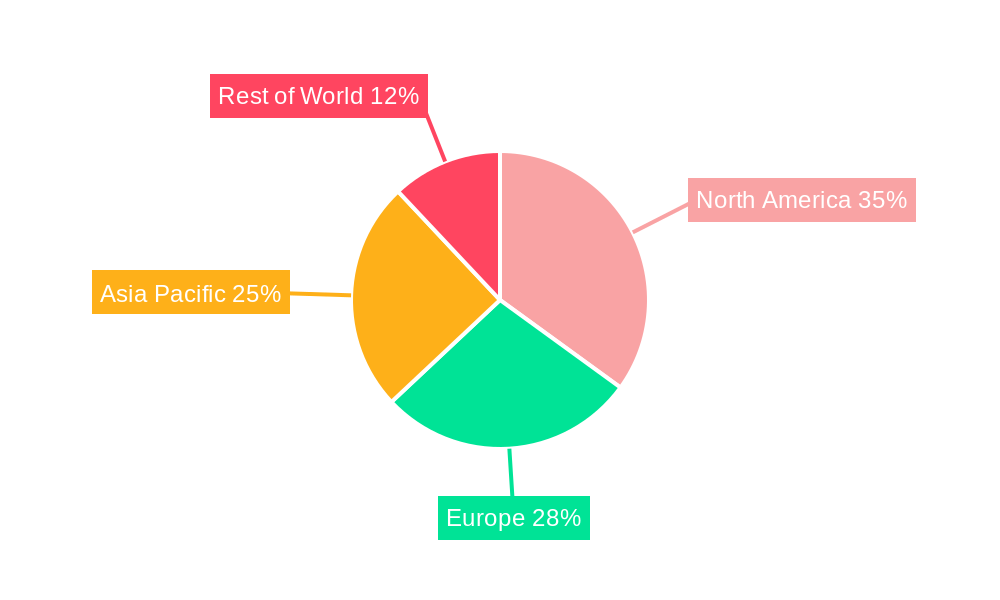
Clinical Grade Wearable Device Regional Market Share

Geographic Coverage of Clinical Grade Wearable Device
Clinical Grade Wearable Device REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Clinical Grade Wearable Device Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Detect
- 5.1.2. Treatment
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Diagnostic Devices
- 5.2.2. Therapeutic Devices
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Clinical Grade Wearable Device Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Detect
- 6.1.2. Treatment
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Diagnostic Devices
- 6.2.2. Therapeutic Devices
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Clinical Grade Wearable Device Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Detect
- 7.1.2. Treatment
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Diagnostic Devices
- 7.2.2. Therapeutic Devices
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Clinical Grade Wearable Device Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Detect
- 8.1.2. Treatment
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Diagnostic Devices
- 8.2.2. Therapeutic Devices
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Clinical Grade Wearable Device Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Detect
- 9.1.2. Treatment
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Diagnostic Devices
- 9.2.2. Therapeutic Devices
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Clinical Grade Wearable Device Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Detect
- 10.1.2. Treatment
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Diagnostic Devices
- 10.2.2. Therapeutic Devices
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Abbott Laboratories
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Siemens Healthcare
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Koninklijke Philips
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Omron Corporation
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Sotera Wireless
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Everist Health
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Polar Electro
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Zephyr Technology
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Intelesens
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Fitbit
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 LifeWatch
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Garmin
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Contec Medical Systems
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Vital Connect
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Dexcom
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Medtronic
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Ge Healthcare
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.1 Abbott Laboratories
List of Figures
- Figure 1: Global Clinical Grade Wearable Device Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Clinical Grade Wearable Device Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Clinical Grade Wearable Device Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Clinical Grade Wearable Device Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Clinical Grade Wearable Device Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Clinical Grade Wearable Device Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Clinical Grade Wearable Device Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Clinical Grade Wearable Device Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Clinical Grade Wearable Device Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Clinical Grade Wearable Device Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Clinical Grade Wearable Device Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Clinical Grade Wearable Device Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Clinical Grade Wearable Device Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Clinical Grade Wearable Device Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Clinical Grade Wearable Device Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Clinical Grade Wearable Device Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Clinical Grade Wearable Device Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Clinical Grade Wearable Device Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Clinical Grade Wearable Device Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Clinical Grade Wearable Device Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Clinical Grade Wearable Device Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Clinical Grade Wearable Device Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Clinical Grade Wearable Device Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Clinical Grade Wearable Device Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Clinical Grade Wearable Device Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Clinical Grade Wearable Device Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Clinical Grade Wearable Device Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Clinical Grade Wearable Device Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Clinical Grade Wearable Device Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Clinical Grade Wearable Device Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Clinical Grade Wearable Device Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Clinical Grade Wearable Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Clinical Grade Wearable Device Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Clinical Grade Wearable Device?
The projected CAGR is approximately 10.9%.
2. Which companies are prominent players in the Clinical Grade Wearable Device?
Key companies in the market include Abbott Laboratories, Siemens Healthcare, Koninklijke Philips, Omron Corporation, Sotera Wireless, Everist Health, Polar Electro, Zephyr Technology, Intelesens, Fitbit, LifeWatch, Garmin, Contec Medical Systems, Vital Connect, Dexcom, Medtronic, Ge Healthcare.
3. What are the main segments of the Clinical Grade Wearable Device?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Clinical Grade Wearable Device," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Clinical Grade Wearable Device report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Clinical Grade Wearable Device?
To stay informed about further developments, trends, and reports in the Clinical Grade Wearable Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
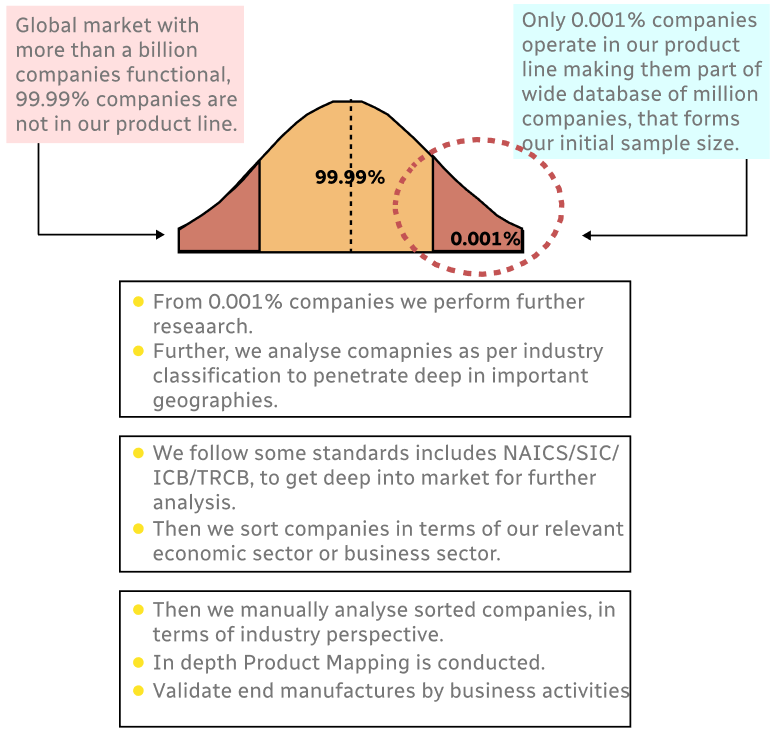
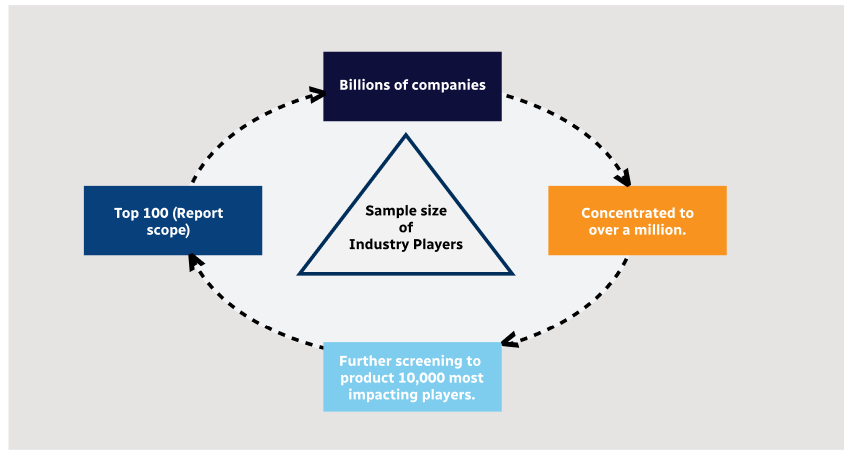
Step 1 - Identification of Relevant Samples Size from Population Database
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
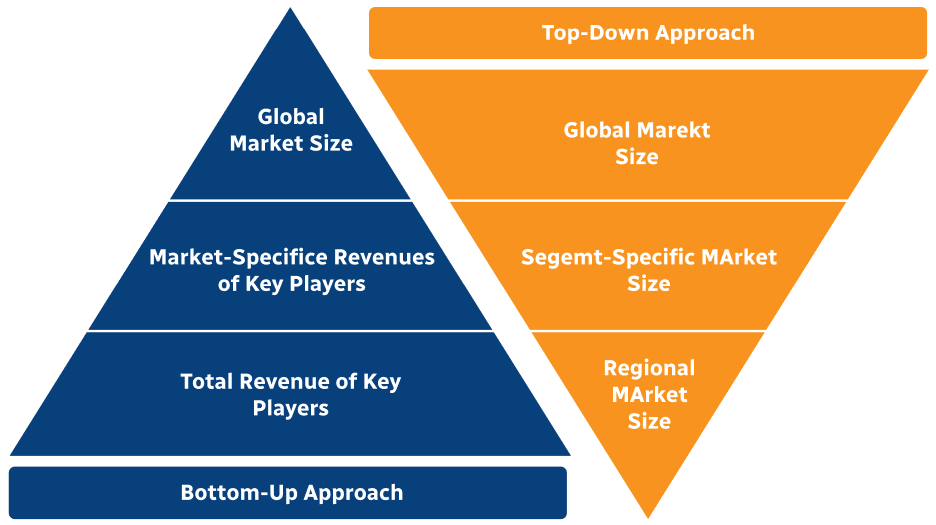
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
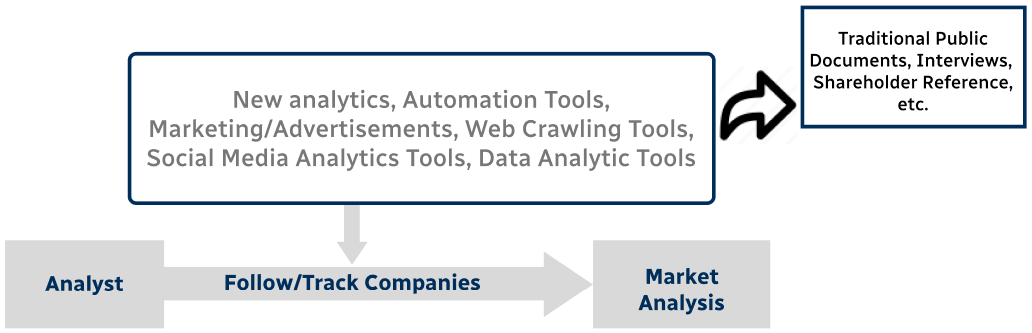
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


