Key Insights
The Cell and Gene Therapy Manufacturing Services Market is poised for remarkable expansion, projected to reach a market size of $88.84 billion by 2025. This robust growth is fueled by a compelling CAGR of 28.1%, indicating a dynamic and rapidly evolving sector. The escalating prevalence of chronic and rare diseases, coupled with significant advancements in genetic engineering and cellular biology, are primary drivers. These innovative therapies offer novel treatment paradigms for previously intractable conditions, generating substantial investment and accelerating clinical development. The market's trajectory is further bolstered by increasing outsourcing by pharmaceutical and biotechnology companies seeking specialized expertise and manufacturing capacity, as well as a growing emphasis on personalized medicine. The demand for both allogeneic and autologous cell therapies, alongside a surge in viral vector-based gene therapies, underscores the technological diversity and broad applicability of these transformative treatments.
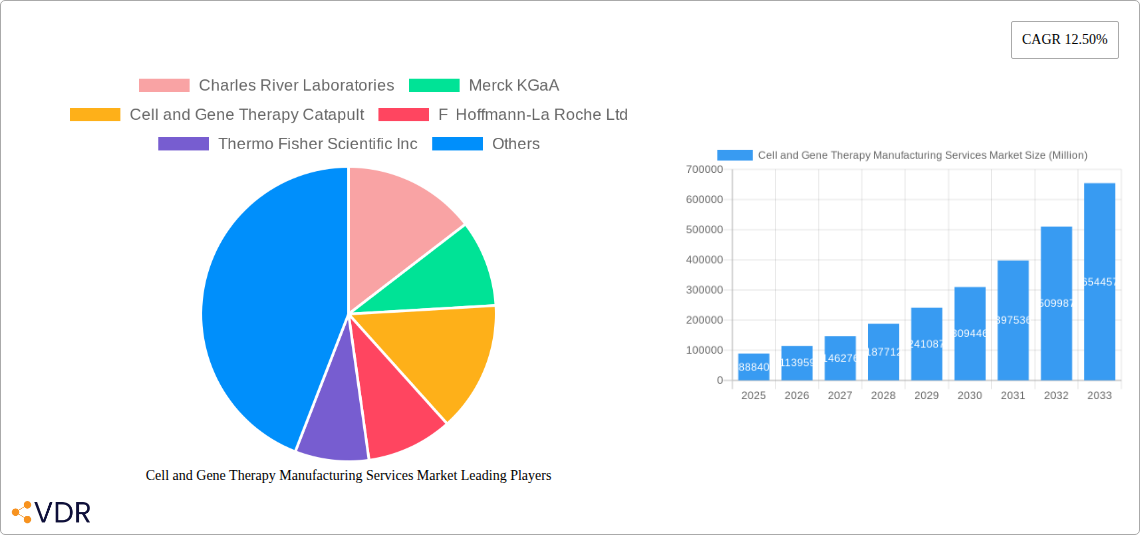
Cell and Gene Therapy Manufacturing Services Market Market Size (In Billion)

The market's impressive growth is further propelled by a burgeoning pipeline of cell and gene therapies entering clinical trials and gaining regulatory approvals. The clinical manufacturing segment is experiencing a substantial uplift as more candidates progress through development stages, while the commercial manufacturing segment is anticipated to accelerate as approved therapies gain market traction. Key indications like oncology are leading the charge, benefiting from the targeted efficacy of these advanced modalities. Cardiovascular and orthopedic diseases are emerging as significant growth areas, with ongoing research exploring the therapeutic potential of cell and gene-based interventions. The participation of major pharmaceutical giants, dedicated cell and gene therapy specialists, and leading academic research institutions highlights the collaborative ecosystem driving innovation and market penetration. Despite challenges such as complex regulatory pathways and high manufacturing costs, the overwhelming therapeutic promise and expanding patient access are expected to sustain this high growth momentum, making the cell and gene therapy manufacturing services market a critical and expanding frontier in healthcare.
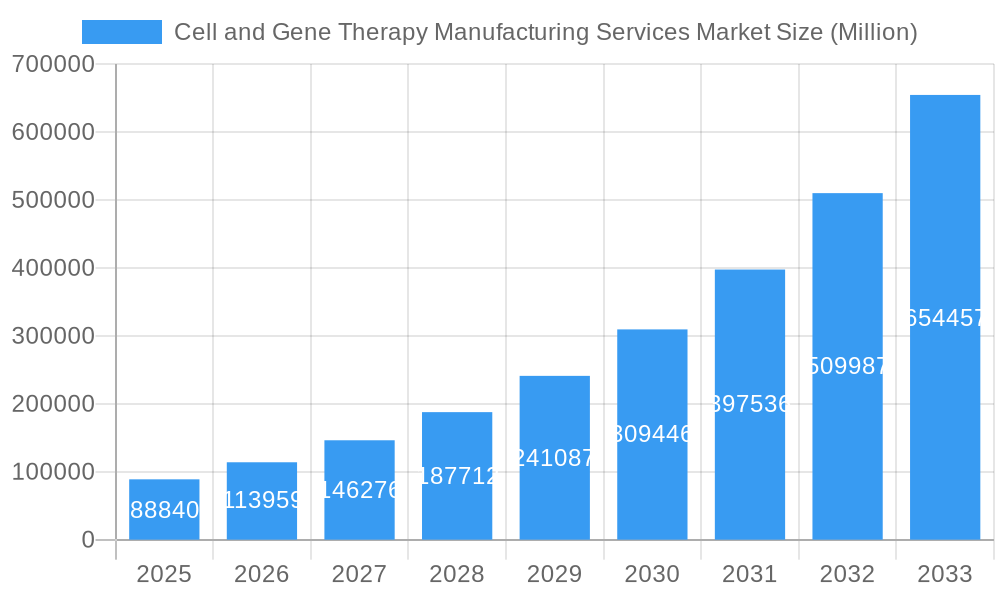
Cell and Gene Therapy Manufacturing Services Market Company Market Share

This comprehensive report offers an in-depth analysis of the global Cell and Gene Therapy Manufacturing Services Market, a rapidly expanding sector critical to the future of medicine. With an estimated market size of $12.8 billion in 2025, this report provides a detailed forecast through 2033, highlighting key growth drivers, emerging opportunities, and the competitive landscape. We dissect parent and child markets, examining Cell Therapy manufacturing (including allogeneic and autologous options, and viral vector production) and Gene Therapy manufacturing (encompassing viral vectors and non-viral vectors). The analysis covers applications in clinical manufacturing and commercial manufacturing, and indications such as oncology, cardiovascular diseases, orthopedic diseases, and infectious diseases. End-users including pharmaceutical and biotechnology companies, academic and research institutes, and others are thoroughly examined.
Cell and Gene Therapy Manufacturing Services Market Dynamics & Structure
The Cell and Gene Therapy Manufacturing Services Market is characterized by a dynamic interplay of technological innovation, evolving regulatory frameworks, and strategic investments. Market concentration is moderately fragmented, with key players actively engaging in mergers, acquisitions, and collaborations to expand their capabilities and market reach. Technological innovation is a paramount driver, fueled by advancements in viral vector production, cell therapy platforms, and gene editing technologies. The demand for efficient and scalable manufacturing solutions for complex autologous and allogeneic cell therapies, as well as gene therapies, is pushing the boundaries of current manufacturing processes. Regulatory bodies worldwide are continuously adapting to oversee the safety and efficacy of these novel treatments, influencing development timelines and manufacturing standards. Competitive product substitutes are limited due to the highly specialized nature of these therapies, but advancements in alternative delivery mechanisms and manufacturing approaches present potential long-term competition. End-user demographics are primarily driven by the needs of pharmaceutical and biotechnology companies seeking to outsource their complex manufacturing needs to specialized contract development and manufacturing organizations (CDMOs).
- Market Concentration: Moderately fragmented, with a mix of established CDMOs and emerging specialized players.
- Technological Innovation Drivers: Advancements in viral vector technology, CRISPR, mRNA platforms, and automated manufacturing processes.
- Regulatory Frameworks: Evolving guidelines from agencies like the FDA and EMA are crucial for market entry and expansion.
- Competitive Product Substitutes: Currently limited for advanced therapies, but focus on improved delivery and efficiency.
- End-User Demographics: Dominated by pharmaceutical and biotechnology companies; growing involvement from academic institutions.
- M&A Trends: Active consolidation and strategic partnerships to enhance capacity and expertise.
Cell and Gene Therapy Manufacturing Services Market Growth Trends & Insights
The global Cell and Gene Therapy Manufacturing Services Market is experiencing exponential growth, projected to reach $40.2 billion by 2033, with a robust CAGR of 15.2% during the forecast period (2025–2033). This surge is directly attributable to the increasing number of cell and gene therapy candidates progressing through clinical trials and gaining regulatory approvals. The oncology segment continues to be the primary therapeutic area driving demand, with a significant pipeline of CAR T-cell therapies and oncolytic virus treatments. Furthermore, the growing pipeline for rare diseases, cardiovascular conditions, and neurological disorders is broadening the application landscape for manufacturing services.
Adoption rates for these advanced therapies are steadily increasing as clinical evidence accumulates and manufacturing scalability improves. The transition from clinical manufacturing to commercial manufacturing presents a significant opportunity for service providers capable of delivering high-quality, cost-effective, and compliant large-scale production. Technological disruptions, such as advancements in viral vector manufacturing, including lentiviral vectors and adenoviral vectors, are enhancing efficiency and reducing production timelines. Innovations in non-viral vector delivery methods are also gaining traction, offering alternative manufacturing pathways.
Consumer behavior shifts are evident as healthcare providers and patients become more receptive to personalized medicine and innovative treatment modalities. The increasing recognition of the therapeutic potential of allogeneic cell therapies over autologous therapies, due to their off-the-shelf availability and potentially lower cost of goods, is also influencing market dynamics. The demand for specialized gene therapy manufacturing services, encompassing both viral vector and non-viral vector approaches, is escalating as more gene therapies receive market authorization. The outsourcing trend among pharmaceutical and biotechnology companies, driven by the complexity and capital intensity of cell and gene therapy manufacturing, is a key growth accelerator, allowing developers to focus on research and clinical development. The market penetration of specialized manufacturing services is expected to deepen as capacity expands and costs become more competitive, making these life-saving therapies accessible to a wider patient population.
Dominant Regions, Countries, or Segments in Cell and Gene Therapy Manufacturing Services Market
North America currently dominates the Cell and Gene Therapy Manufacturing Services Market, driven by a robust ecosystem of leading pharmaceutical and biotechnology companies, extensive research and development activities, and a favorable regulatory environment. The United States, in particular, plays a pivotal role due to its high concentration of innovative biotech firms and academic research institutions actively involved in cell and gene therapy development. The region's substantial investment in clinical manufacturing and its pioneering role in commercializing groundbreaking therapies like CAR T-cell therapies for oncology have solidified its leadership. Government initiatives and funding for biomedical research further bolster this dominance.
In terms of Service Type, Cell Therapy manufacturing, specifically for allogeneic and autologous therapies utilizing viral vectors, represents a significant market segment. The escalating number of CAR T-cell therapy approvals and the expanding pipeline for other cell therapies like stem cell therapies are key drivers. Within Gene Therapy, viral vector manufacturing remains the most prevalent, with lentiviral vectors and adenoviral vectors being widely adopted for a range of indications. However, advancements in non-viral vectors are creating new avenues for growth.
Geographically, Europe follows North America, with countries like Germany, the UK, and Switzerland exhibiting strong growth due to established biopharmaceutical industries and supportive government policies for innovation. Asia-Pacific is emerging as a rapidly growing region, propelled by increasing investments, a growing number of clinical trials, and a burgeoning domestic biopharmaceutical sector, particularly in China and Japan.
Looking at Application, Clinical Manufacturing is currently a larger segment due to the ongoing development of numerous cell and gene therapies. However, Commercial Manufacturing is projected to witness the highest growth rate as more therapies gain regulatory approval and move towards large-scale production. The increasing demand for therapies targeting oncology remains the dominant Indication, accounting for a substantial share of the market. However, significant growth potential is also observed in cardiovascular diseases, orthopedic diseases, and infectious diseases as research progresses and new therapeutic approaches emerge.
Among End Users, Pharmaceutical and Biotechnology Companies are the primary consumers of cell and gene therapy manufacturing services, driving the majority of market revenue. Academic and Research Institutes also contribute significantly, particularly in the early stages of therapy development and clinical trials. The increasing complexity and cost of manufacturing are compelling more organizations to outsource their manufacturing needs to specialized CDMOs.
- Dominant Region: North America (particularly the United States).
- Key Service Type Drivers: Allogeneic and Autologous Cell Therapy using Viral Vectors, Gene Therapy using Viral Vectors.
- Application Trend: Rapid growth in Commercial Manufacturing from Clinical Manufacturing.
- Indication Focus: Oncology leads, with significant potential in Cardiovascular and Rare Diseases.
- Primary End User: Pharmaceutical and Biotechnology Companies.
Cell and Gene Therapy Manufacturing Services Market Product Landscape
The product landscape within the Cell and Gene Therapy Manufacturing Services Market is defined by innovative solutions aimed at addressing the complexities of producing advanced therapies. Key offerings include specialized viral vector manufacturing platforms, robust cell therapy expansion systems, and advanced gene editing tools. These services are crucial for enabling the production of autologous and allogeneic cell therapies, as well as various gene therapies utilizing viral and non-viral vectors. Performance metrics are evaluated based on yield, purity, scalability, and regulatory compliance. Unique selling propositions often lie in proprietary technologies that enhance efficiency, reduce costs, and accelerate time-to-market for novel treatments targeting indications like oncology and rare diseases. Technological advancements are continually shaping the product offerings, leading to more integrated and automated manufacturing solutions.
Key Drivers, Barriers & Challenges in Cell and Gene Therapy Manufacturing Services Market
The Cell and Gene Therapy Manufacturing Services Market is propelled by several key drivers. The burgeoning pipeline of advanced therapies, particularly in oncology, is a primary catalyst. Increased investment in R&D by pharmaceutical and biotechnology companies, coupled with significant venture capital funding, fuels demand for specialized manufacturing expertise. Advances in gene editing technologies like CRISPR and improved viral vector production methods are enabling the development of more complex and effective therapies. The growing adoption of allogeneic approaches, offering scalability and faster patient access, further stimulates market growth.
However, significant barriers and challenges exist. The high cost of manufacturing remains a critical impediment, limiting accessibility for some patients and payers. Regulatory hurdles associated with complex biologics, requiring rigorous validation and quality control, add to development timelines and costs. Supply chain complexities for specialized raw materials and the need for highly skilled personnel create bottlenecks. Furthermore, the limited availability of manufacturing capacity, particularly for commercial-scale production, can constrain the market. Competition, while present among CDMOs, is often focused on specialized capabilities rather than direct product substitution for the therapies themselves.
Emerging Opportunities in Cell and Gene Therapy Manufacturing Services Market
Emerging opportunities in the Cell and Gene Therapy Manufacturing Services Market are abundant and diverse. The expansion of cell and gene therapies into new therapeutic areas beyond oncology, such as cardiovascular diseases, neurological disorders, and infectious diseases, presents significant untapped markets. The development and adoption of non-viral vector delivery systems offer innovative manufacturing pathways and potentially lower costs. Furthermore, the increasing focus on developing allogeneic therapies, which require scalable manufacturing solutions, represents a substantial growth area. The demand for end-to-end solutions, from process development to commercialization, is also on the rise. There is a growing opportunity for specialized services in areas like vector engineering, cell engineering, and analytics, supporting the maturation of the industry.
Growth Accelerators in the Cell and Gene Therapy Manufacturing Services Market Industry
Several catalysts are accelerating long-term growth in the Cell and Gene Therapy Manufacturing Services Market. Technological breakthroughs in areas such as continuous manufacturing, automation, and artificial intelligence are poised to enhance efficiency, reduce costs, and improve the scalability of production. Strategic partnerships between CDMOs, pharmaceutical companies, and academic institutions are fostering innovation and accelerating the translation of research into commercial products. Market expansion strategies, including the establishment of new manufacturing facilities in emerging regions, are crucial for meeting the growing global demand. The increasing number of regulatory approvals for novel cell and gene therapies is a strong indicator of future market expansion and a testament to the success of these advanced treatments.
Key Players Shaping the Cell and Gene Therapy Manufacturing Services Market Market
- Charles River Laboratories
- Merck KGaA
- Cell and Gene Therapy Catapult
- F Hoffmann-La Roche Ltd
- Thermo Fisher Scientific Inc
- Takara Bio Inc
- WuXi AppTec
- Lonza
- Oxford Biomedica PLC
- Catalent Inc
- Nikon CeLL innovation Co Ltd
- Fujifilm Holdings Corporation
Notable Milestones in Cell and Gene Therapy Manufacturing Services Market Sector
- March 2023: Thermo Fisher Scientific opened a cell therapy facility at the University of California, San Francisco (UCSF) to accelerate the development of breakthrough therapies for cancer, rare diseases, and other illnesses.
- January 2022: AllogeneTherapeutics Inc. and AntionBiosciences Inc. entered into an exclusive collaboration and global license agreement for Antion's miRNA technology (miCAR) to advance multiplex gene silencing as an additional tool to develop next-generation allogeneic CAR T products.
In-Depth Cell and Gene Therapy Manufacturing Services Market Market Outlook
The future outlook for the Cell and Gene Therapy Manufacturing Services Market is exceptionally promising, driven by a confluence of accelerating factors. Continued investment in novel therapeutic development, particularly for unmet medical needs in oncology and rare diseases, will sustain robust demand. Advancements in manufacturing technologies, such as automation and single-use systems, will further enhance efficiency and scalability, making therapies more accessible. Strategic collaborations and partnerships will remain critical for navigating the complex regulatory landscape and optimizing production processes. The growing pipeline of allogeneic cell therapies and the increasing development of non-viral vector gene therapies present substantial growth opportunities for specialized service providers. The market is poised for significant expansion as more therapies move through clinical trials and achieve commercialization, ultimately benefiting a wider patient population seeking life-changing treatments.
Cell and Gene Therapy Manufacturing Services Market Segmentation
-
1. Service Type
-
1.1. Cell Therapy
- 1.1.1. Allogeneic
- 1.1.2. Autologous
- 1.1.3. Viral Vector
-
1.2. Gene Therapy
- 1.2.1. Non-viral Vectors
- 1.2.2. Viral Vectors
-
1.1. Cell Therapy
-
2. Application
- 2.1. Clinical Manufacturing
- 2.2. Commercial Manufacturing
-
3. Indication
- 3.1. Oncology
- 3.2. Cardiovascular Diseases
- 3.3. Orthopedic Diseases
- 3.4. Infectious Diseases
- 3.5. Other Indications
-
4. End User
- 4.1. Pharmaceutical and Biotechnology Companies
- 4.2. Academic and Research Institutes
- 4.3. Other End Users
Cell and Gene Therapy Manufacturing Services Market Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
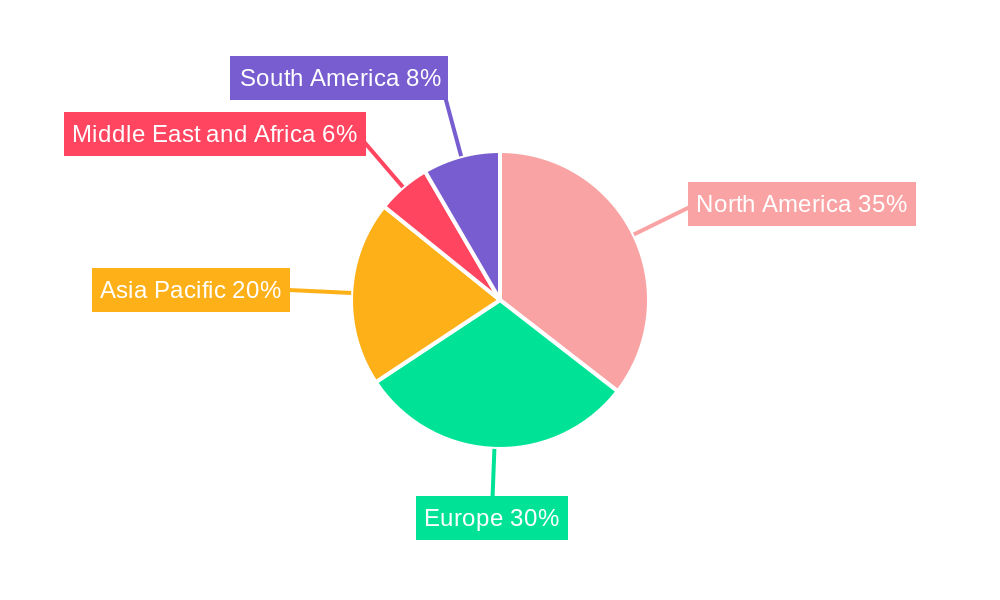
Cell and Gene Therapy Manufacturing Services Market Regional Market Share

Geographic Coverage of Cell and Gene Therapy Manufacturing Services Market
Cell and Gene Therapy Manufacturing Services Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 28.1% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Service Type
- 5.1.1. Cell Therapy
- 5.1.1.1. Allogeneic
- 5.1.1.2. Autologous
- 5.1.1.3. Viral Vector
- 5.1.2. Gene Therapy
- 5.1.2.1. Non-viral Vectors
- 5.1.2.2. Viral Vectors
- 5.1.1. Cell Therapy
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Clinical Manufacturing
- 5.2.2. Commercial Manufacturing
- 5.3. Market Analysis, Insights and Forecast - by Indication
- 5.3.1. Oncology
- 5.3.2. Cardiovascular Diseases
- 5.3.3. Orthopedic Diseases
- 5.3.4. Infectious Diseases
- 5.3.5. Other Indications
- 5.4. Market Analysis, Insights and Forecast - by End User
- 5.4.1. Pharmaceutical and Biotechnology Companies
- 5.4.2. Academic and Research Institutes
- 5.4.3. Other End Users
- 5.5. Market Analysis, Insights and Forecast - by Region
- 5.5.1. North America
- 5.5.2. Europe
- 5.5.3. Asia Pacific
- 5.5.4. Middle East and Africa
- 5.5.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Service Type
- 6. Global Cell and Gene Therapy Manufacturing Services Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Service Type
- 6.1.1. Cell Therapy
- 6.1.1.1. Allogeneic
- 6.1.1.2. Autologous
- 6.1.1.3. Viral Vector
- 6.1.2. Gene Therapy
- 6.1.2.1. Non-viral Vectors
- 6.1.2.2. Viral Vectors
- 6.1.1. Cell Therapy
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Clinical Manufacturing
- 6.2.2. Commercial Manufacturing
- 6.3. Market Analysis, Insights and Forecast - by Indication
- 6.3.1. Oncology
- 6.3.2. Cardiovascular Diseases
- 6.3.3. Orthopedic Diseases
- 6.3.4. Infectious Diseases
- 6.3.5. Other Indications
- 6.4. Market Analysis, Insights and Forecast - by End User
- 6.4.1. Pharmaceutical and Biotechnology Companies
- 6.4.2. Academic and Research Institutes
- 6.4.3. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Service Type
- 7. North America Cell and Gene Therapy Manufacturing Services Market Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Service Type
- 7.1.1. Cell Therapy
- 7.1.1.1. Allogeneic
- 7.1.1.2. Autologous
- 7.1.1.3. Viral Vector
- 7.1.2. Gene Therapy
- 7.1.2.1. Non-viral Vectors
- 7.1.2.2. Viral Vectors
- 7.1.1. Cell Therapy
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Clinical Manufacturing
- 7.2.2. Commercial Manufacturing
- 7.3. Market Analysis, Insights and Forecast - by Indication
- 7.3.1. Oncology
- 7.3.2. Cardiovascular Diseases
- 7.3.3. Orthopedic Diseases
- 7.3.4. Infectious Diseases
- 7.3.5. Other Indications
- 7.4. Market Analysis, Insights and Forecast - by End User
- 7.4.1. Pharmaceutical and Biotechnology Companies
- 7.4.2. Academic and Research Institutes
- 7.4.3. Other End Users
- 7.1. Market Analysis, Insights and Forecast - by Service Type
- 8. Europe Cell and Gene Therapy Manufacturing Services Market Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Service Type
- 8.1.1. Cell Therapy
- 8.1.1.1. Allogeneic
- 8.1.1.2. Autologous
- 8.1.1.3. Viral Vector
- 8.1.2. Gene Therapy
- 8.1.2.1. Non-viral Vectors
- 8.1.2.2. Viral Vectors
- 8.1.1. Cell Therapy
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Clinical Manufacturing
- 8.2.2. Commercial Manufacturing
- 8.3. Market Analysis, Insights and Forecast - by Indication
- 8.3.1. Oncology
- 8.3.2. Cardiovascular Diseases
- 8.3.3. Orthopedic Diseases
- 8.3.4. Infectious Diseases
- 8.3.5. Other Indications
- 8.4. Market Analysis, Insights and Forecast - by End User
- 8.4.1. Pharmaceutical and Biotechnology Companies
- 8.4.2. Academic and Research Institutes
- 8.4.3. Other End Users
- 8.1. Market Analysis, Insights and Forecast - by Service Type
- 9. Asia Pacific Cell and Gene Therapy Manufacturing Services Market Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Service Type
- 9.1.1. Cell Therapy
- 9.1.1.1. Allogeneic
- 9.1.1.2. Autologous
- 9.1.1.3. Viral Vector
- 9.1.2. Gene Therapy
- 9.1.2.1. Non-viral Vectors
- 9.1.2.2. Viral Vectors
- 9.1.1. Cell Therapy
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Clinical Manufacturing
- 9.2.2. Commercial Manufacturing
- 9.3. Market Analysis, Insights and Forecast - by Indication
- 9.3.1. Oncology
- 9.3.2. Cardiovascular Diseases
- 9.3.3. Orthopedic Diseases
- 9.3.4. Infectious Diseases
- 9.3.5. Other Indications
- 9.4. Market Analysis, Insights and Forecast - by End User
- 9.4.1. Pharmaceutical and Biotechnology Companies
- 9.4.2. Academic and Research Institutes
- 9.4.3. Other End Users
- 9.1. Market Analysis, Insights and Forecast - by Service Type
- 10. Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Service Type
- 10.1.1. Cell Therapy
- 10.1.1.1. Allogeneic
- 10.1.1.2. Autologous
- 10.1.1.3. Viral Vector
- 10.1.2. Gene Therapy
- 10.1.2.1. Non-viral Vectors
- 10.1.2.2. Viral Vectors
- 10.1.1. Cell Therapy
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Clinical Manufacturing
- 10.2.2. Commercial Manufacturing
- 10.3. Market Analysis, Insights and Forecast - by Indication
- 10.3.1. Oncology
- 10.3.2. Cardiovascular Diseases
- 10.3.3. Orthopedic Diseases
- 10.3.4. Infectious Diseases
- 10.3.5. Other Indications
- 10.4. Market Analysis, Insights and Forecast - by End User
- 10.4.1. Pharmaceutical and Biotechnology Companies
- 10.4.2. Academic and Research Institutes
- 10.4.3. Other End Users
- 10.1. Market Analysis, Insights and Forecast - by Service Type
- 11. South America Cell and Gene Therapy Manufacturing Services Market Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Service Type
- 11.1.1. Cell Therapy
- 11.1.1.1. Allogeneic
- 11.1.1.2. Autologous
- 11.1.1.3. Viral Vector
- 11.1.2. Gene Therapy
- 11.1.2.1. Non-viral Vectors
- 11.1.2.2. Viral Vectors
- 11.1.1. Cell Therapy
- 11.2. Market Analysis, Insights and Forecast - by Application
- 11.2.1. Clinical Manufacturing
- 11.2.2. Commercial Manufacturing
- 11.3. Market Analysis, Insights and Forecast - by Indication
- 11.3.1. Oncology
- 11.3.2. Cardiovascular Diseases
- 11.3.3. Orthopedic Diseases
- 11.3.4. Infectious Diseases
- 11.3.5. Other Indications
- 11.4. Market Analysis, Insights and Forecast - by End User
- 11.4.1. Pharmaceutical and Biotechnology Companies
- 11.4.2. Academic and Research Institutes
- 11.4.3. Other End Users
- 11.1. Market Analysis, Insights and Forecast - by Service Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Charles River Laboratories
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Merck KGaA
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Cell and Gene Therapy Catapult
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 F Hoffmann-La Roche Ltd
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Thermo Fisher Scientific Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Takara Bio Inc
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 WuXi AppTec
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Lonza
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Oxford Biomedica PLC
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Catalent Inc
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Nikon CeLL innovation Co Ltd
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Fujifilm Holdings Corporation
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.1 Charles River Laboratories
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Cell and Gene Therapy Manufacturing Services Market Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Service Type 2025 & 2033
- Figure 3: North America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Service Type 2025 & 2033
- Figure 4: North America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Application 2025 & 2033
- Figure 5: North America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Indication 2025 & 2033
- Figure 7: North America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Indication 2025 & 2033
- Figure 8: North America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by End User 2025 & 2033
- Figure 9: North America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by End User 2025 & 2033
- Figure 10: North America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Country 2025 & 2033
- Figure 11: North America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Country 2025 & 2033
- Figure 12: Europe Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Service Type 2025 & 2033
- Figure 13: Europe Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Service Type 2025 & 2033
- Figure 14: Europe Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Indication 2025 & 2033
- Figure 17: Europe Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Indication 2025 & 2033
- Figure 18: Europe Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by End User 2025 & 2033
- Figure 19: Europe Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by End User 2025 & 2033
- Figure 20: Europe Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Country 2025 & 2033
- Figure 21: Europe Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Country 2025 & 2033
- Figure 22: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Service Type 2025 & 2033
- Figure 23: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Service Type 2025 & 2033
- Figure 24: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Application 2025 & 2033
- Figure 25: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Application 2025 & 2033
- Figure 26: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Indication 2025 & 2033
- Figure 27: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Indication 2025 & 2033
- Figure 28: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by End User 2025 & 2033
- Figure 29: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by End User 2025 & 2033
- Figure 30: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Country 2025 & 2033
- Figure 32: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Service Type 2025 & 2033
- Figure 33: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Service Type 2025 & 2033
- Figure 34: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Application 2025 & 2033
- Figure 35: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Application 2025 & 2033
- Figure 36: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Indication 2025 & 2033
- Figure 37: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Indication 2025 & 2033
- Figure 38: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by End User 2025 & 2033
- Figure 39: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by End User 2025 & 2033
- Figure 40: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Country 2025 & 2033
- Figure 41: Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Country 2025 & 2033
- Figure 42: South America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Service Type 2025 & 2033
- Figure 43: South America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Service Type 2025 & 2033
- Figure 44: South America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Application 2025 & 2033
- Figure 45: South America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Application 2025 & 2033
- Figure 46: South America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Indication 2025 & 2033
- Figure 47: South America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Indication 2025 & 2033
- Figure 48: South America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by End User 2025 & 2033
- Figure 49: South America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by End User 2025 & 2033
- Figure 50: South America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined), by Country 2025 & 2033
- Figure 51: South America Cell and Gene Therapy Manufacturing Services Market Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 2: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 3: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Indication 2020 & 2033
- Table 4: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by End User 2020 & 2033
- Table 5: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 7: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Indication 2020 & 2033
- Table 9: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by End User 2020 & 2033
- Table 10: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 11: United States Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 12: Canada Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 13: Mexico Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 15: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 16: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Indication 2020 & 2033
- Table 17: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by End User 2020 & 2033
- Table 18: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: Germany Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: United Kingdom Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Rest of Europe Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 26: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 27: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Indication 2020 & 2033
- Table 28: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by End User 2020 & 2033
- Table 29: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 30: China Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 31: Japan Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: India Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: Australia Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: South Korea Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: Rest of Asia Pacific Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 37: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Indication 2020 & 2033
- Table 39: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by End User 2020 & 2033
- Table 40: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 41: GCC Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: South Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: Rest of Middle East and Africa Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Service Type 2020 & 2033
- Table 45: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 46: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Indication 2020 & 2033
- Table 47: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by End User 2020 & 2033
- Table 48: Global Cell and Gene Therapy Manufacturing Services Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 49: Brazil Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Argentina Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 51: Rest of South America Cell and Gene Therapy Manufacturing Services Market Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Cell and Gene Therapy Manufacturing Services Market?
The projected CAGR is approximately 28.1%.
2. Which companies are prominent players in the Cell and Gene Therapy Manufacturing Services Market?
Key companies in the market include Charles River Laboratories, Merck KGaA, Cell and Gene Therapy Catapult, F Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc, Takara Bio Inc, WuXi AppTec, Lonza, Oxford Biomedica PLC, Catalent Inc, Nikon CeLL innovation Co Ltd, Fujifilm Holdings Corporation.
3. What are the main segments of the Cell and Gene Therapy Manufacturing Services Market?
The market segments include Service Type, Application, Indication, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
High Prevalence of Cancer and Other Target Diseases; Increasing Grants and R&D Investments in the Pharmaceutical Sector; Increasing Organic and Inorganic Developments Between Pharmaceutical Companies and CDMOs.
6. What are the notable trends driving market growth?
Allogeneic Segment is Expected to Have a Significant Market Share During the Forecast Period.
7. Are there any restraints impacting market growth?
High Operational Costs Associated with the Cell and Gene Therapy Manufacturing.
8. Can you provide examples of recent developments in the market?
March 2023: Thermo Fisher Scientific opened a cell therapy facility at the University of California, San Francisco (UCSF) to accelerate the development of breakthrough therapies for cancer, rare diseases, and other illnesses.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Cell and Gene Therapy Manufacturing Services Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Cell and Gene Therapy Manufacturing Services Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Cell and Gene Therapy Manufacturing Services Market?
To stay informed about further developments, trends, and reports in the Cell and Gene Therapy Manufacturing Services Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
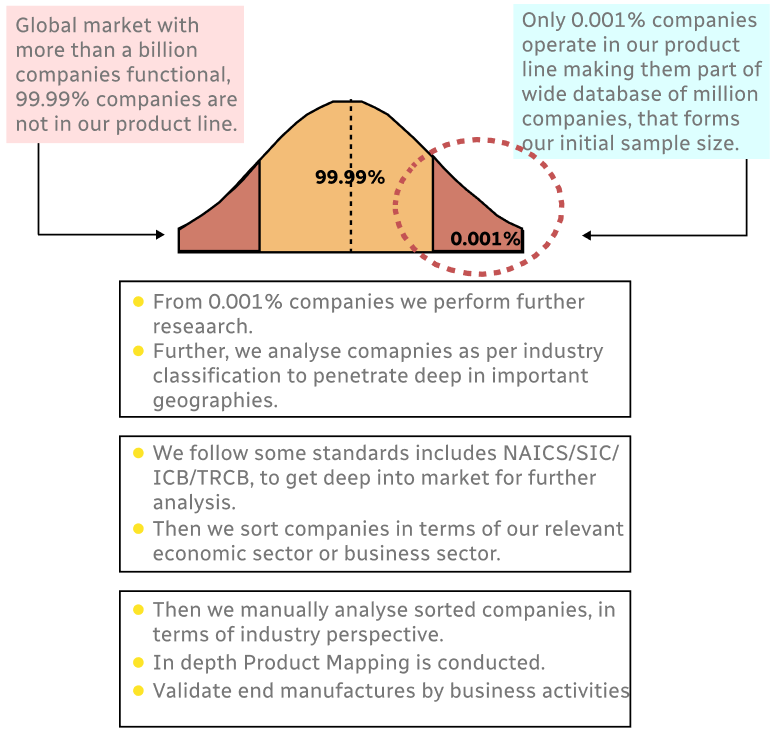
Step 1 - Identification of Relevant Samples Size from Population Database
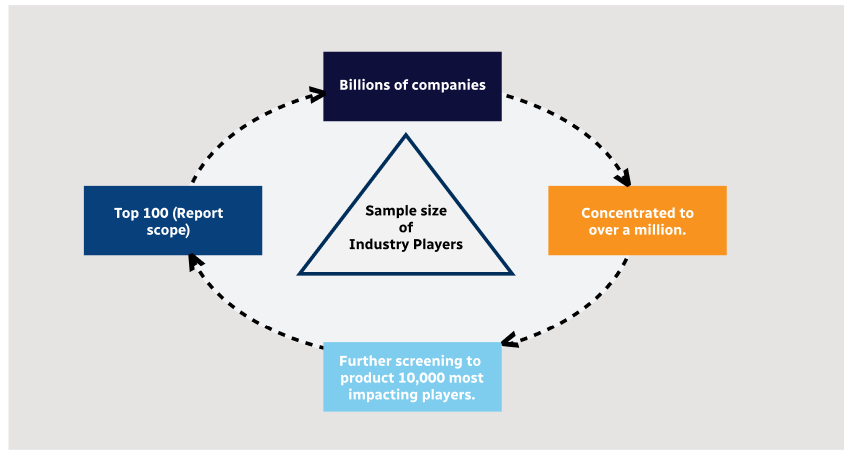
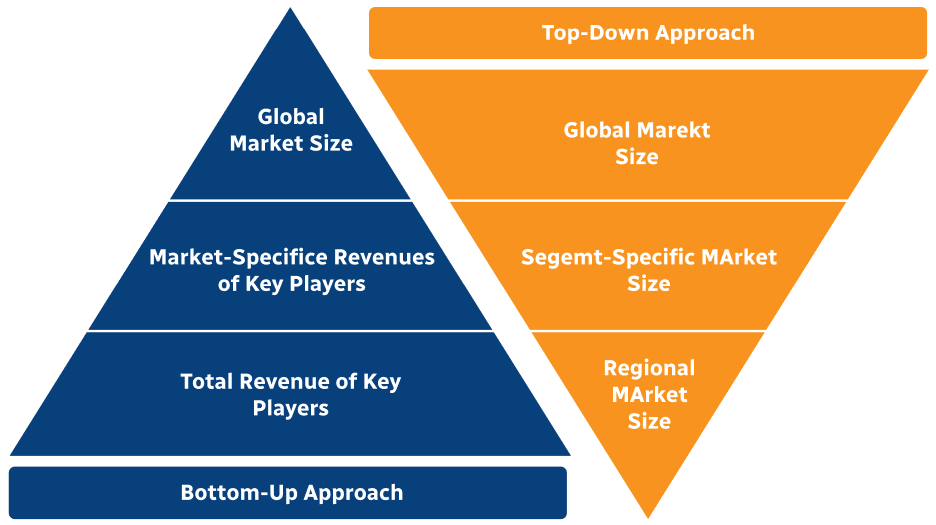
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
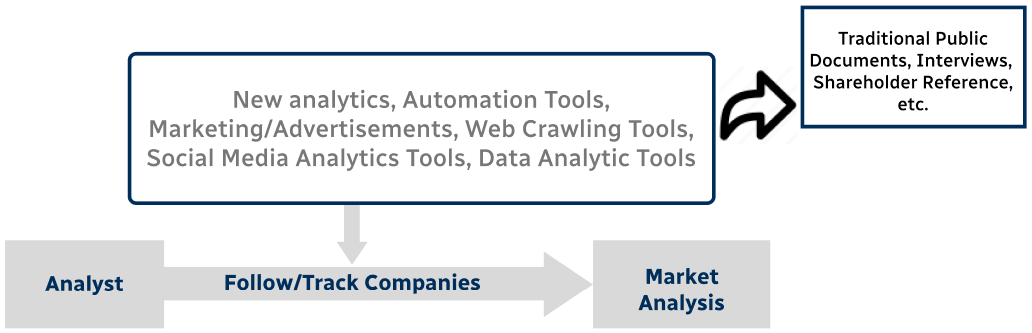
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


