Key Insights
The global Blood Transfusion Diagnostics market is poised for substantial growth, projected to reach approximately $5.97 billion in 2025. This expansion is driven by an anticipated Compound Annual Growth Rate (CAGR) of 7.89% throughout the forecast period of 2025-2033. Key growth drivers fueling this upward trajectory include the increasing demand for safe blood transfusions, advancements in diagnostic technologies, and a rising prevalence of blood-related diseases. The growing emphasis on disease screening, particularly for infectious agents like HIV, Hepatitis B, and Hepatitis C, before blood donation, is a significant factor. Furthermore, the expanding healthcare infrastructure, especially in emerging economies, coupled with increased awareness regarding the importance of blood transfusion safety, is contributing to market expansion. The market segmentation reveals a robust demand across both Instruments and Consumables, with Blood Screening and Blood Group Typing being dominant application areas. Hospitals and Diagnostic Laboratories represent the primary end-users, underscoring the critical role of these diagnostics in routine healthcare and specialized medical procedures.
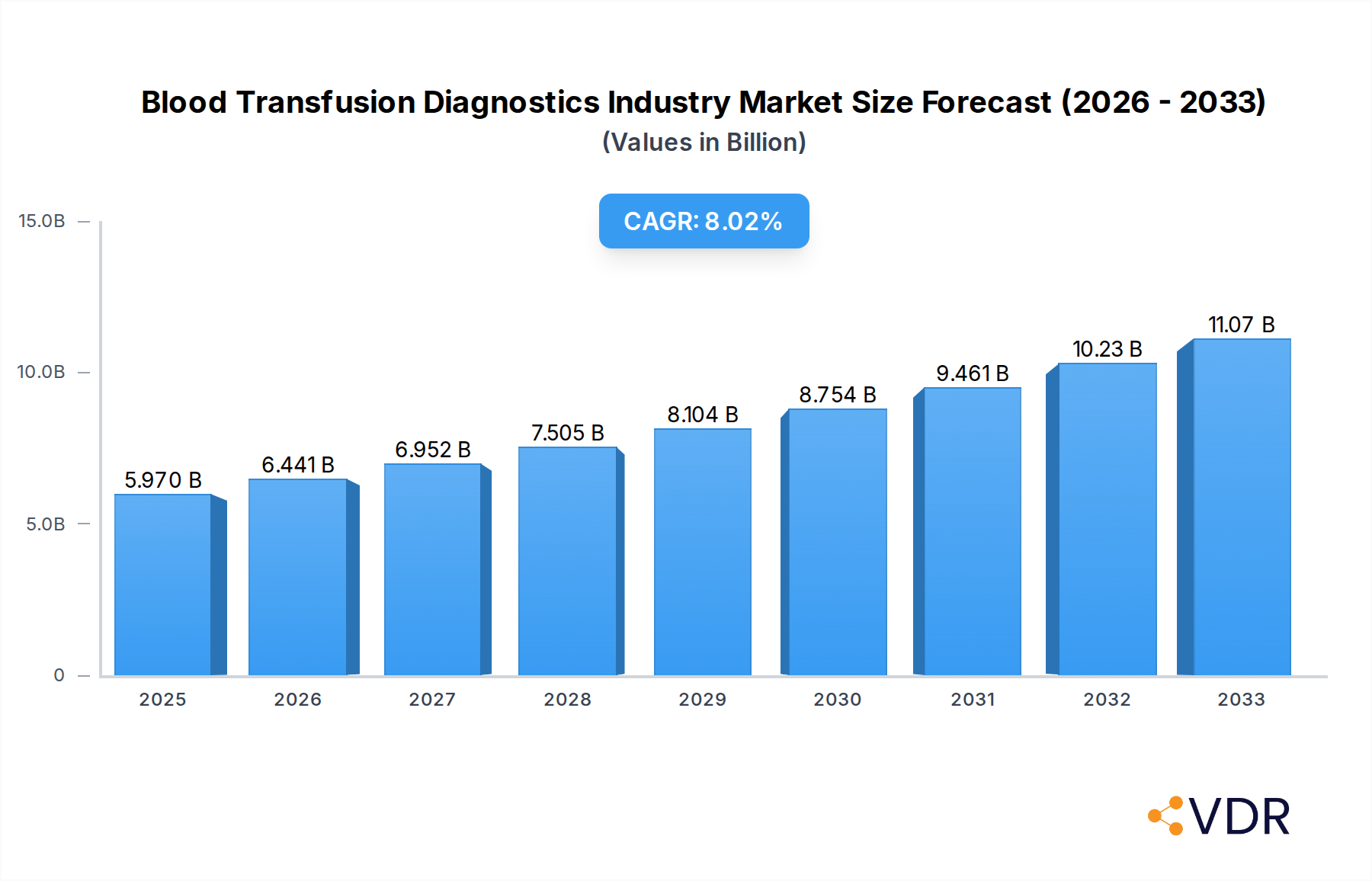
Blood Transfusion Diagnostics Industry Market Size (In Billion)

The Blood Transfusion Diagnostics market is characterized by dynamic trends and certain restraints that shape its evolution. Continuous innovation in immunoassay and molecular diagnostic techniques is enhancing the sensitivity and specificity of blood testing, thereby improving transfusion safety and patient outcomes. The integration of automation and artificial intelligence in diagnostic workflows is also a notable trend, promising increased efficiency and reduced turnaround times. However, the market faces certain restraints, including the high cost of advanced diagnostic equipment and reagents, particularly in resource-limited settings. Stringent regulatory frameworks and the need for consistent validation of new diagnostic assays can also pose challenges to rapid market penetration. Despite these challenges, the commitment to enhancing blood safety, coupled with ongoing research and development efforts by leading companies such as Abbott, Roche Diagnostics, and Bio-Rad Laboratories, ensures a promising outlook for the Blood Transfusion Diagnostics market over the coming decade.
Blood Transfusion Diagnostics Industry Company Market Share

Comprehensive Blood Transfusion Diagnostics Market Analysis: Opportunities, Trends, and Key Players (2019–2033)
This in-depth report provides a critical analysis of the global Blood Transfusion Diagnostics industry, offering a 15-year outlook from 2019 to 2033, with a base and estimated year of 2025. It meticulously examines market dynamics, growth trends, regional dominance, product landscape, key drivers, emerging opportunities, and competitive strategies. Delving into both parent and child markets, this research equips industry professionals, investors, and stakeholders with actionable insights to navigate this crucial sector. All monetary values are presented in billions of units.
Blood Transfusion Diagnostics Industry Market Dynamics & Structure
The Blood Transfusion Diagnostics industry is characterized by a moderate market concentration, driven by significant investments in technological innovation and increasingly stringent regulatory frameworks worldwide. The increasing prevalence of infectious diseases and the growing demand for safe blood transfusions have propelled the adoption of advanced diagnostic solutions. The competitive landscape features established players and emerging innovators, with a constant push towards improved assay sensitivity, specificity, and turnaround times. Barriers to entry include high R&D costs, the need for extensive clinical validation, and the complex regulatory approval processes for new diagnostic kits and instruments.
- Technological Innovation Drivers: Development of molecular diagnostic techniques (NAT), automation of testing processes, and advancements in immunoassay platforms.
- Regulatory Frameworks: Stringent guidelines from regulatory bodies like the FDA, EMA, and WHO mandate rigorous testing protocols to ensure blood safety.
- Competitive Product Substitutes: While direct substitutes are limited, advancements in alternative therapeutic approaches or preventative measures could indirectly impact diagnostic demand.
- End-User Demographics: A growing aging population and increasing incidence of chronic diseases requiring blood transfusions contribute to sustained demand.
- M&A Trends: Strategic acquisitions and collaborations are observed as companies seek to expand their product portfolios, gain market access, and leverage synergistic technologies. For instance, the historical period (2019-2024) witnessed approximately 5-7 significant M&A deals annually within the broader diagnostics sector, with a notable portion focusing on transfusion diagnostics.
Blood Transfusion Diagnostics Industry Growth Trends & Insights
The global Blood Transfusion Diagnostics market is poised for robust expansion, driven by a confluence of factors that are reshaping healthcare delivery and patient safety protocols. The increasing global demand for safe blood products, coupled with the rising incidence of blood-borne diseases and transfusion-transmitted infections (TTIs), has placed a significant emphasis on accurate and rapid diagnostic solutions. Technological advancements are at the forefront of this evolution, with the widespread adoption of Nucleic Acid Testing (NAT) systems significantly enhancing the sensitivity and specificity of pathogen detection in donated blood. This transition from traditional serological methods to molecular diagnostics represents a paradigm shift, offering earlier detection and reduced window periods for infections like HIV, Hepatitis B, and Hepatitis C, thereby bolstering transfusion safety to unprecedented levels.
Furthermore, the growing emphasis on personalized medicine and patient-specific transfusion strategies is also contributing to market growth. Advanced blood grouping techniques, including extended antigen phenotyping and compatibility testing, are becoming increasingly crucial, especially for patients requiring multiple transfusions or those with complex transfusion histories. The integration of automation and artificial intelligence (AI) in diagnostic laboratories is streamlining workflows, reducing manual errors, and improving turnaround times for critical tests. This technological disruption not only enhances operational efficiency but also allows healthcare professionals to make informed decisions more rapidly, leading to better patient outcomes.
Consumer behavior shifts, particularly the growing awareness among the general public and healthcare providers regarding the importance of blood safety, are also acting as significant growth accelerators. This heightened awareness translates into increased demand for sophisticated diagnostic technologies and a greater willingness to invest in advanced testing infrastructure. The expanding healthcare infrastructure in emerging economies, coupled with rising disposable incomes, is further broadening the market reach for blood transfusion diagnostics. Governments and private organizations are also investing in programs aimed at improving blood supply chain management and transfusion safety, creating a favorable ecosystem for market growth. The increasing volume of blood donations globally, driven by public health campaigns and a greater understanding of the critical need for blood, directly fuels the demand for the diagnostic tools that ensure its safety and efficacy. This sustained growth trajectory is projected to continue throughout the forecast period, underpinned by ongoing technological innovation and a steadfast commitment to enhancing patient safety in blood transfusions. The projected CAGR for the forecast period (2025-2033) is estimated to be between 6.5% and 8.0%.
Dominant Regions, Countries, or Segments in Blood Transfusion Diagnostics Industry
The global Blood Transfusion Diagnostics market's dominance is largely dictated by the technological sophistication, healthcare infrastructure, and regulatory stringency present within its key regions. North America, particularly the United States, has consistently held a leading position due to its advanced healthcare system, substantial investments in R&D, and a well-established regulatory framework that mandates high standards for blood safety. The presence of major diagnostic companies and leading research institutions in this region fosters rapid innovation and adoption of cutting-edge technologies.
Europe also represents a significant market, driven by a similar commitment to patient safety and the presence of a robust diagnostics industry across countries like Germany, France, and the United Kingdom. The European Union's directives on blood safety and quality standards ensure a consistent demand for advanced transfusion diagnostics. The region benefits from strong government support for healthcare, advanced laboratory infrastructure, and a growing elderly population that necessitates more frequent blood transfusions.
Asia Pacific is emerging as a high-growth region, propelled by rapid economic development, expanding healthcare access, and a growing awareness of transfusion-transmitted infections. Countries like China and India are witnessing a surge in demand for blood transfusion diagnostics owing to their large populations and increasing healthcare expenditure. Government initiatives to improve blood transfusion services and the rising incidence of infectious diseases are key drivers in this region.
In terms of market segmentation, the Consumables segment, encompassing reagents, assay kits, and disposables, holds a substantial market share. This is attributed to the repetitive nature of blood testing, where each donation and transfusion requires a fresh set of consumables. The Instruments segment, though requiring higher initial investment, is crucial for enabling the accurate and efficient processing of these consumables, with automated platforms gaining significant traction.
By Application, Blood Screening for infectious diseases is a dominant segment, given the paramount importance of ensuring donated blood is free from pathogens. Blood Group Typing also remains a critical application, essential for ensuring compatibility between donor and recipient.
The Hospitals segment, followed by Diagnostics Laboratories and Blood Banks, represents the primary end-users. Hospitals, being the direct recipients of blood products, have a vested interest in robust diagnostic capabilities. Diagnostic laboratories often provide outsourced testing services to a wider network of healthcare facilities, while blood banks are the central hubs for collection, processing, and distribution, making them integral to the transfusion diagnostics ecosystem. The market share within these segments is closely linked to the overall healthcare expenditure and regulatory compliance efforts in each region. For instance, the consumables market is estimated to be valued at approximately \$7.5 billion in 2025, with instruments accounting for an additional \$4.2 billion.
Blood Transfusion Diagnostics Industry Product Landscape
The product landscape in blood transfusion diagnostics is characterized by continuous innovation focused on enhancing accuracy, speed, and efficiency. Key advancements include the development of multiplex assays capable of detecting multiple pathogens simultaneously, significantly reducing testing time and cost. Automated immunoassay analyzers and molecular diagnostic platforms are at the forefront, offering higher throughput and improved sensitivity for detecting even low levels of viral or bacterial loads.
Emerging technologies like microfluidics are enabling the development of point-of-care (POC) diagnostic devices, facilitating rapid testing at the bedside or in remote settings. These products offer unique selling propositions in terms of portability, ease of use, and reduced sample volume requirements. Performance metrics such as sensitivity, specificity, assay linearity, and turnaround time are critical differentiators, with manufacturers constantly striving to achieve superior results to meet stringent regulatory standards and clinical demands for transfusion safety.
Key Drivers, Barriers & Challenges in Blood Transfusion Diagnostics Industry
Key Drivers:
- Increasing Prevalence of Infectious Diseases: Rising global incidence of HIV, Hepatitis B, Hepatitis C, and emerging infectious agents necessitates rigorous blood screening.
- Growing Demand for Safe Blood Transfusions: Heightened awareness of transfusion-transmitted infections (TTIs) and evolving regulatory mandates drive the adoption of advanced diagnostics.
- Technological Advancements: Development of automated platforms, molecular diagnostics (NAT), and multiplex assays enhance accuracy, speed, and efficiency.
- Expanding Healthcare Infrastructure: Growth in emerging economies and increased healthcare spending fuels demand for diagnostic solutions.
- Aging Population: The demographic shift towards an older population increases the need for blood transfusions, thereby driving diagnostic demand.
Barriers & Challenges:
- High Cost of Advanced Technologies: Significant capital investment required for advanced instruments and reagents can be a barrier, especially for smaller facilities.
- Stringent Regulatory Approval Processes: Obtaining regulatory clearance for new diagnostic products is time-consuming and resource-intensive, impacting market entry.
- Skilled Workforce Shortage: A lack of trained personnel to operate and maintain sophisticated diagnostic equipment can hinder adoption.
- Supply Chain Disruptions: Global events can impact the availability of raw materials and finished diagnostic products, leading to delays and increased costs. The estimated impact of supply chain disruptions on market growth in the past year was between 1-2%.
- Reimbursement Policies: Inconsistent or inadequate reimbursement for advanced diagnostic tests can limit their widespread use.
Emerging Opportunities in Blood Transfusion Diagnostics Industry
Emerging opportunities lie in the development and implementation of next-generation sequencing (NGS) for comprehensive pathogen detection and variant analysis in blood. The expansion of point-of-care (POC) diagnostic solutions for rapid testing in remote or resource-limited settings presents a significant untapped market. Furthermore, advancements in artificial intelligence and machine learning for automated data analysis and predictive diagnostics in transfusion medicine offer promising avenues for improved patient outcomes and operational efficiency. There is also a growing focus on diagnostics for rare blood types and for screening donor suitability for complex transfusion scenarios, creating niche market opportunities.
Growth Accelerators in the Blood Transfusion Diagnostics Industry Industry
Technological breakthroughs, particularly in molecular diagnostics and automation, are significant growth accelerators. The increasing adoption of Nucleic Acid Testing (NAT) for pooled or individual samples is a prime example, offering superior sensitivity in detecting viral RNA and DNA. Strategic partnerships between diagnostic manufacturers and blood collection agencies are crucial for driving the implementation of these advanced technologies. Furthermore, market expansion strategies focusing on developing countries, where healthcare infrastructure is rapidly evolving, represent a substantial growth opportunity. The proactive stance of regulatory bodies in promoting blood safety standards also acts as a powerful catalyst for market expansion and technological adoption.
Key Players Shaping the Blood Transfusion Diagnostics Industry Market
- Abbott
- Bio-Rad Laboratories Inc
- Danaher
- Diasorin S p A
- F Hoffmann-La Roche Ltd
- Grifols S A
- Immucor Inc
- Merck KGaA
- QUOTIENT
- Werfen
Notable Milestones in Blood Transfusion Diagnostics Industry Sector
- 2020: Widespread adoption of advanced Nucleic Acid Testing (NAT) for routine blood screening for HIV, HBV, and HCV, significantly reducing the window period for infection detection.
- 2021: Launch of multiplex PCR assays capable of detecting a broader range of pathogens in donated blood, enhancing transfusion safety.
- 2022: Increased investment in automation solutions for blood group typing and antibody screening, improving laboratory efficiency.
- 2023: Regulatory approvals for new diagnostic platforms with enhanced sensitivity and specificity for emerging infectious diseases.
- 2024: Growing interest and research in the application of AI and machine learning for predictive diagnostics and personalized transfusion strategies.
In-Depth Blood Transfusion Diagnostics Industry Market Outlook
The future of the blood transfusion diagnostics market is exceptionally promising, driven by an unwavering commitment to enhancing blood safety and patient well-being. Growth accelerators such as the continued evolution of molecular diagnostic technologies, including next-generation sequencing, will enable more comprehensive and precise pathogen detection. The strategic expansion into emerging markets, coupled with proactive government initiatives to strengthen blood transfusion infrastructure, will unlock significant growth potential. Furthermore, the integration of digital health solutions and artificial intelligence for streamlined laboratory operations and data interpretation will further propel the industry forward. The focus will increasingly shift towards personalized diagnostics and the early detection of a wider spectrum of infectious agents, ensuring the global blood supply remains safer than ever before.
Blood Transfusion Diagnostics Industry Segmentation
-
1. Type
- 1.1. Instruments
- 1.2. Consumables
-
2. Application
- 2.1. Blood Screening
- 2.2. Blood Group Typing
-
3. End-User
- 3.1. Hospitals
- 3.2. Diagnostics Laboratories
- 3.3. Blood Banks
Blood Transfusion Diagnostics Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
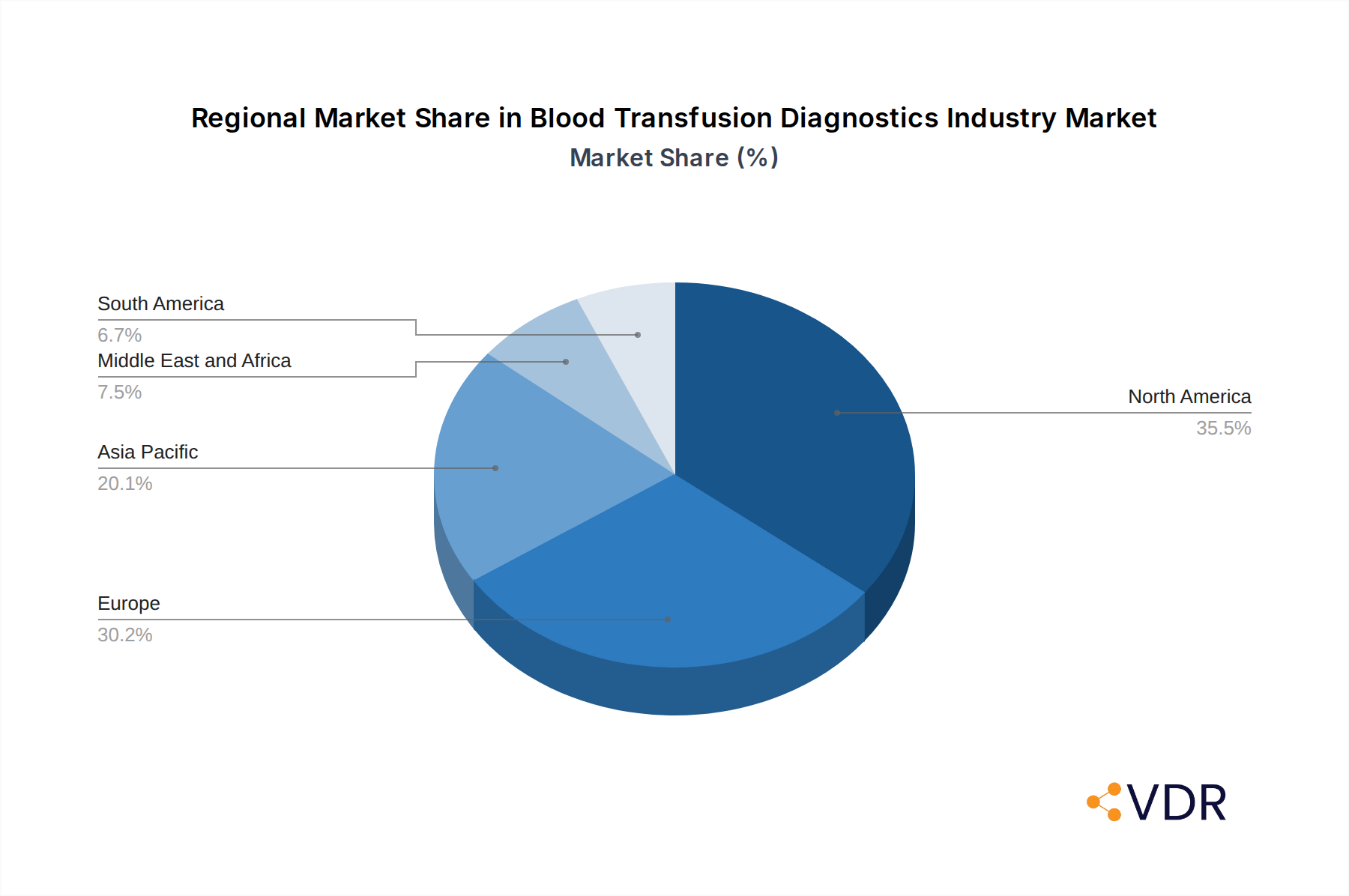
Blood Transfusion Diagnostics Industry Regional Market Share

Geographic Coverage of Blood Transfusion Diagnostics Industry
Blood Transfusion Diagnostics Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.89% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type
- 5.1.1. Instruments
- 5.1.2. Consumables
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Blood Screening
- 5.2.2. Blood Group Typing
- 5.3. Market Analysis, Insights and Forecast - by End-User
- 5.3.1. Hospitals
- 5.3.2. Diagnostics Laboratories
- 5.3.3. Blood Banks
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Type
- 6. Global Blood Transfusion Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type
- 6.1.1. Instruments
- 6.1.2. Consumables
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Blood Screening
- 6.2.2. Blood Group Typing
- 6.3. Market Analysis, Insights and Forecast - by End-User
- 6.3.1. Hospitals
- 6.3.2. Diagnostics Laboratories
- 6.3.3. Blood Banks
- 6.1. Market Analysis, Insights and Forecast - by Type
- 7. North America Blood Transfusion Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Type
- 7.1.1. Instruments
- 7.1.2. Consumables
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Blood Screening
- 7.2.2. Blood Group Typing
- 7.3. Market Analysis, Insights and Forecast - by End-User
- 7.3.1. Hospitals
- 7.3.2. Diagnostics Laboratories
- 7.3.3. Blood Banks
- 7.1. Market Analysis, Insights and Forecast - by Type
- 8. Europe Blood Transfusion Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Type
- 8.1.1. Instruments
- 8.1.2. Consumables
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Blood Screening
- 8.2.2. Blood Group Typing
- 8.3. Market Analysis, Insights and Forecast - by End-User
- 8.3.1. Hospitals
- 8.3.2. Diagnostics Laboratories
- 8.3.3. Blood Banks
- 8.1. Market Analysis, Insights and Forecast - by Type
- 9. Asia Pacific Blood Transfusion Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Type
- 9.1.1. Instruments
- 9.1.2. Consumables
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Blood Screening
- 9.2.2. Blood Group Typing
- 9.3. Market Analysis, Insights and Forecast - by End-User
- 9.3.1. Hospitals
- 9.3.2. Diagnostics Laboratories
- 9.3.3. Blood Banks
- 9.1. Market Analysis, Insights and Forecast - by Type
- 10. Middle East and Africa Blood Transfusion Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Type
- 10.1.1. Instruments
- 10.1.2. Consumables
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Blood Screening
- 10.2.2. Blood Group Typing
- 10.3. Market Analysis, Insights and Forecast - by End-User
- 10.3.1. Hospitals
- 10.3.2. Diagnostics Laboratories
- 10.3.3. Blood Banks
- 10.1. Market Analysis, Insights and Forecast - by Type
- 11. South America Blood Transfusion Diagnostics Industry Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Type
- 11.1.1. Instruments
- 11.1.2. Consumables
- 11.2. Market Analysis, Insights and Forecast - by Application
- 11.2.1. Blood Screening
- 11.2.2. Blood Group Typing
- 11.3. Market Analysis, Insights and Forecast - by End-User
- 11.3.1. Hospitals
- 11.3.2. Diagnostics Laboratories
- 11.3.3. Blood Banks
- 11.1. Market Analysis, Insights and Forecast - by Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Werfen*List Not Exhaustive
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Bio-Rad Laboratories Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 QUOTIENT
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Merck KGaA
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 F Hoffmann-La Roche Ltd
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Diasorin S p A
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Abbott
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Grifols S A
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Immucor Inc
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Danaher
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.1 Werfen*List Not Exhaustive
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Blood Transfusion Diagnostics Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Blood Transfusion Diagnostics Industry Revenue (billion), by Type 2025 & 2033
- Figure 3: North America Blood Transfusion Diagnostics Industry Revenue Share (%), by Type 2025 & 2033
- Figure 4: North America Blood Transfusion Diagnostics Industry Revenue (billion), by Application 2025 & 2033
- Figure 5: North America Blood Transfusion Diagnostics Industry Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Blood Transfusion Diagnostics Industry Revenue (billion), by End-User 2025 & 2033
- Figure 7: North America Blood Transfusion Diagnostics Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 8: North America Blood Transfusion Diagnostics Industry Revenue (billion), by Country 2025 & 2033
- Figure 9: North America Blood Transfusion Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe Blood Transfusion Diagnostics Industry Revenue (billion), by Type 2025 & 2033
- Figure 11: Europe Blood Transfusion Diagnostics Industry Revenue Share (%), by Type 2025 & 2033
- Figure 12: Europe Blood Transfusion Diagnostics Industry Revenue (billion), by Application 2025 & 2033
- Figure 13: Europe Blood Transfusion Diagnostics Industry Revenue Share (%), by Application 2025 & 2033
- Figure 14: Europe Blood Transfusion Diagnostics Industry Revenue (billion), by End-User 2025 & 2033
- Figure 15: Europe Blood Transfusion Diagnostics Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 16: Europe Blood Transfusion Diagnostics Industry Revenue (billion), by Country 2025 & 2033
- Figure 17: Europe Blood Transfusion Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific Blood Transfusion Diagnostics Industry Revenue (billion), by Type 2025 & 2033
- Figure 19: Asia Pacific Blood Transfusion Diagnostics Industry Revenue Share (%), by Type 2025 & 2033
- Figure 20: Asia Pacific Blood Transfusion Diagnostics Industry Revenue (billion), by Application 2025 & 2033
- Figure 21: Asia Pacific Blood Transfusion Diagnostics Industry Revenue Share (%), by Application 2025 & 2033
- Figure 22: Asia Pacific Blood Transfusion Diagnostics Industry Revenue (billion), by End-User 2025 & 2033
- Figure 23: Asia Pacific Blood Transfusion Diagnostics Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 24: Asia Pacific Blood Transfusion Diagnostics Industry Revenue (billion), by Country 2025 & 2033
- Figure 25: Asia Pacific Blood Transfusion Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue (billion), by Type 2025 & 2033
- Figure 27: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue Share (%), by Type 2025 & 2033
- Figure 28: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue (billion), by Application 2025 & 2033
- Figure 29: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue Share (%), by Application 2025 & 2033
- Figure 30: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue (billion), by End-User 2025 & 2033
- Figure 31: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 32: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue (billion), by Country 2025 & 2033
- Figure 33: Middle East and Africa Blood Transfusion Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: South America Blood Transfusion Diagnostics Industry Revenue (billion), by Type 2025 & 2033
- Figure 35: South America Blood Transfusion Diagnostics Industry Revenue Share (%), by Type 2025 & 2033
- Figure 36: South America Blood Transfusion Diagnostics Industry Revenue (billion), by Application 2025 & 2033
- Figure 37: South America Blood Transfusion Diagnostics Industry Revenue Share (%), by Application 2025 & 2033
- Figure 38: South America Blood Transfusion Diagnostics Industry Revenue (billion), by End-User 2025 & 2033
- Figure 39: South America Blood Transfusion Diagnostics Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 40: South America Blood Transfusion Diagnostics Industry Revenue (billion), by Country 2025 & 2033
- Figure 41: South America Blood Transfusion Diagnostics Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 2: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 3: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 4: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 5: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 6: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 7: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 8: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 9: United States Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Canada Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 11: Mexico Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 12: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 13: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 14: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 15: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 16: Germany Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: France Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 19: Italy Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Spain Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 23: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 24: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 25: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 26: China Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Japan Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: India Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 29: Australia Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: South Korea Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 33: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 34: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 35: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 36: GCC Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: South Africa Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: Rest of Middle East and Africa Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 39: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Type 2020 & 2033
- Table 40: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Application 2020 & 2033
- Table 41: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 42: Global Blood Transfusion Diagnostics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 43: Brazil Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: Argentina Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Rest of South America Blood Transfusion Diagnostics Industry Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Blood Transfusion Diagnostics Industry?
The projected CAGR is approximately 7.89%.
2. Which companies are prominent players in the Blood Transfusion Diagnostics Industry?
Key companies in the market include Werfen*List Not Exhaustive, Bio-Rad Laboratories Inc, QUOTIENT, Merck KGaA, F Hoffmann-La Roche Ltd, Diasorin S p A, Abbott, Grifols S A, Immucor Inc, Danaher.
3. What are the main segments of the Blood Transfusion Diagnostics Industry?
The market segments include Type, Application, End-User.
4. Can you provide details about the market size?
The market size is estimated to be USD 5.97 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Number of Blood Related Disorders; Technological Advancement in the Blood Testing Kits and Assays.
6. What are the notable trends driving market growth?
Reagents & Kits Segment is Anticipated to Hold a Significant Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Certain Adverse Reaction Associated with the Procedure.
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Blood Transfusion Diagnostics Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Blood Transfusion Diagnostics Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Blood Transfusion Diagnostics Industry?
To stay informed about further developments, trends, and reports in the Blood Transfusion Diagnostics Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
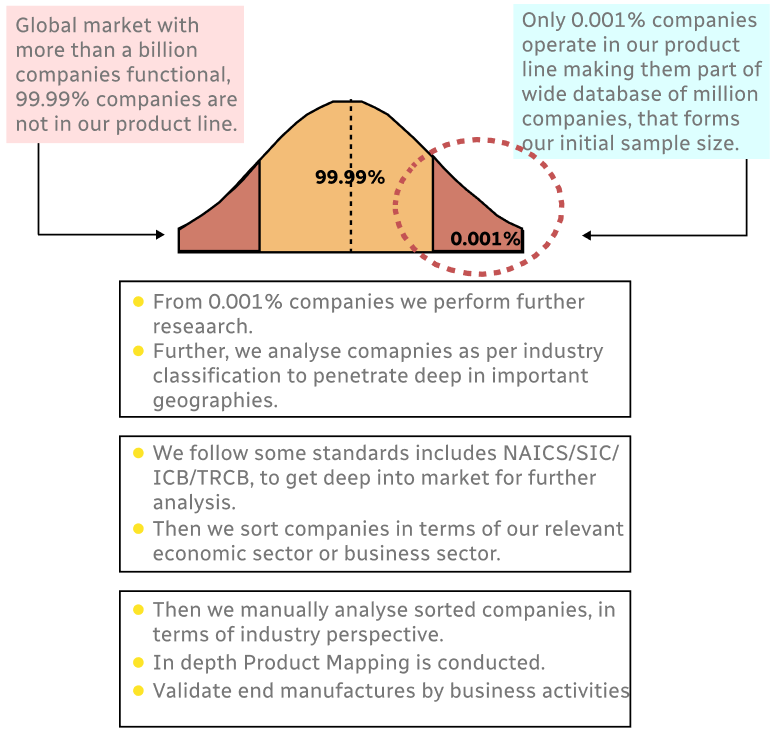
Step 1 - Identification of Relevant Samples Size from Population Database
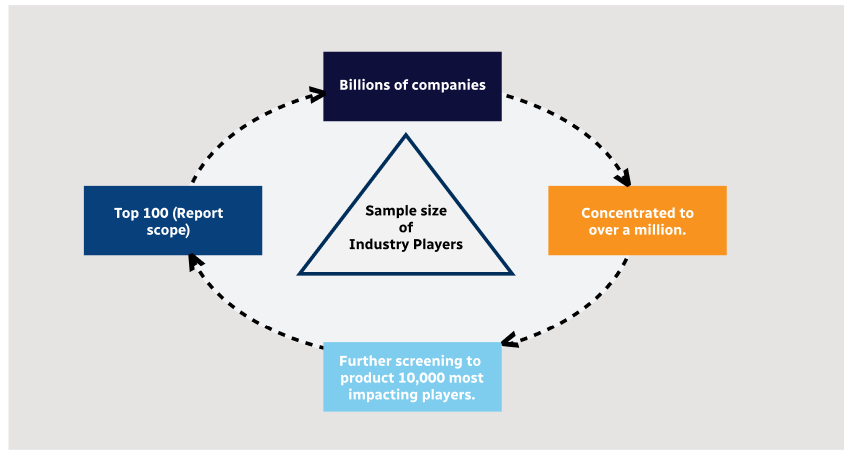
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
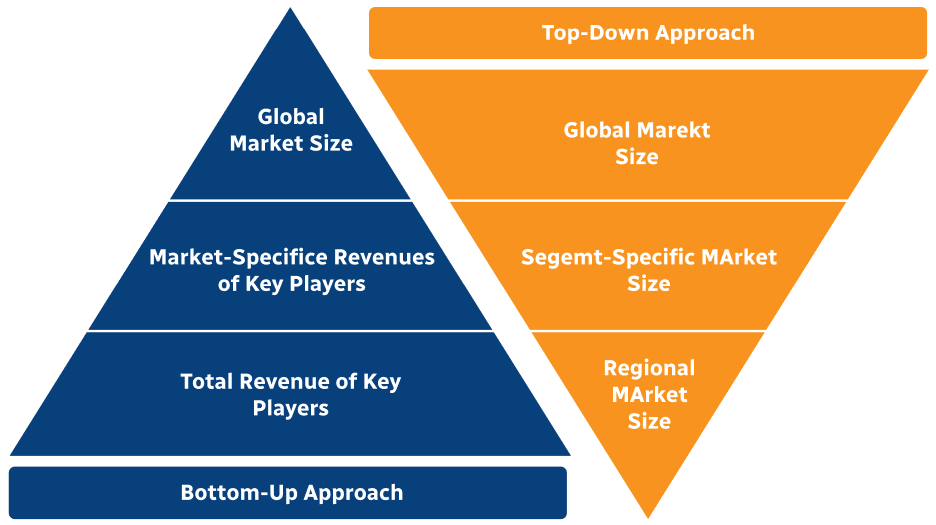
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
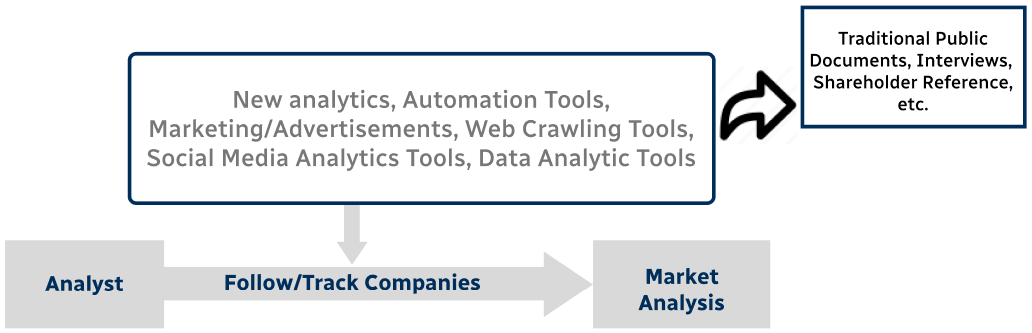
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


