Key Insights
The global Balloon Introducer System market is poised for substantial growth, projected to reach an estimated $4.77 billion in 2025. This robust expansion is underpinned by a significant Compound Annual Growth Rate (CAGR) of 9.9%, indicating a dynamic and evolving landscape for these critical medical devices. The increasing prevalence of cardiovascular diseases, including coronary artery and peripheral artery diseases, serves as a primary driver for market expansion. As minimally invasive procedures gain traction in treating these conditions, the demand for sophisticated balloon introducer systems to facilitate accurate device delivery escalates. Furthermore, advancements in drug-coated balloon technology are revolutionizing treatment paradigms, offering improved patient outcomes and driving innovation in introducer system design, thereby contributing to market growth. The expanding applications in neurovascular interventions and the continuous need for enhanced procedural efficiency also fuel this upward trajectory.
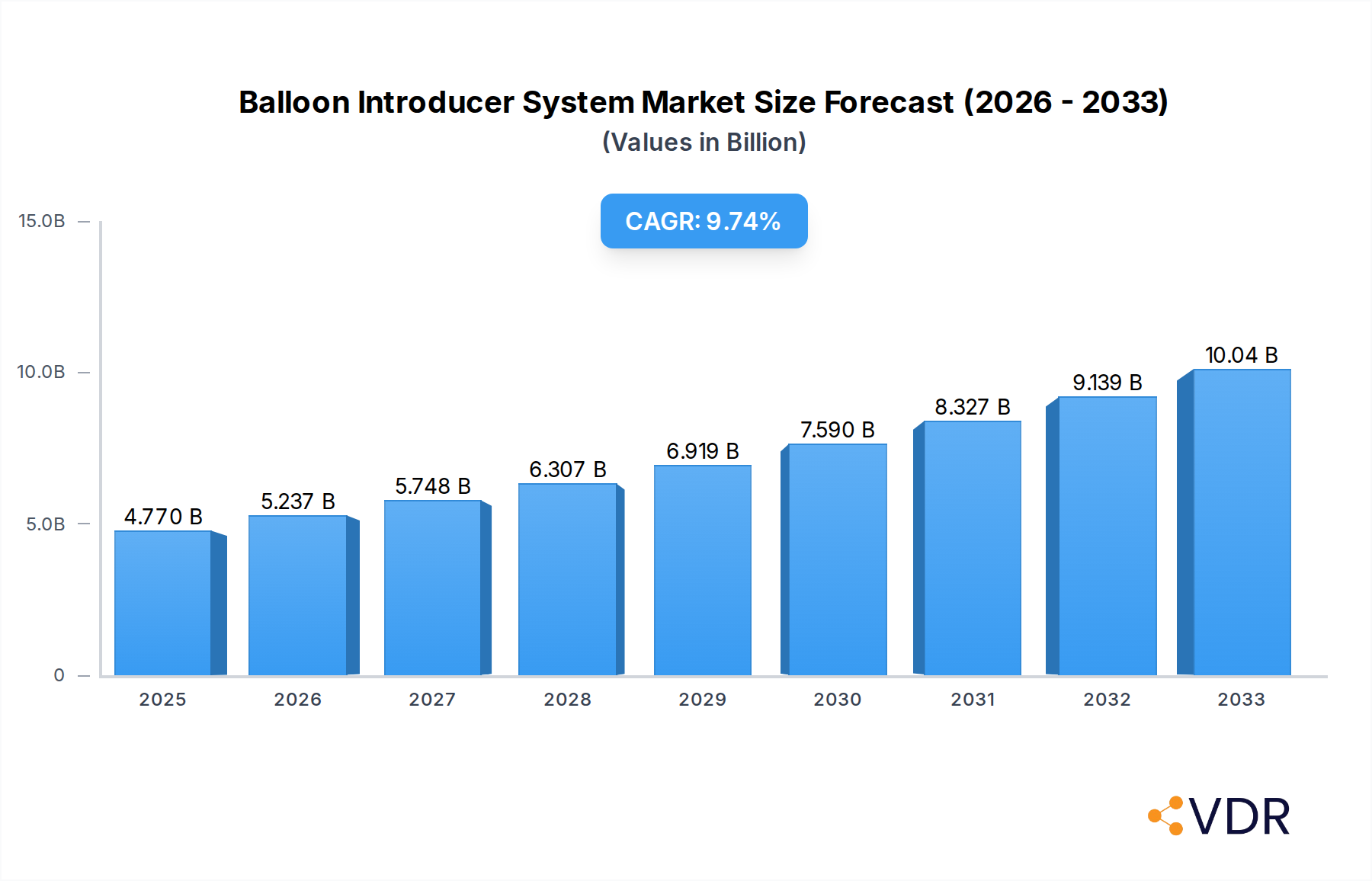
Balloon Introducer System Market Size (In Billion)

The market is segmented by application and type, reflecting diverse end-user needs and technological advancements. Coronary Artery Diseases and Peripheral Artery Diseases represent significant application segments, while advancements in Cutting and Scoring Balloon Introducer Systems and Drug-Coated Balloon Introducer Systems highlight key technological trends. While the market benefits from strong drivers, certain restraints such as the high cost of advanced systems and stringent regulatory approvals in some regions can pose challenges. However, the ongoing research and development efforts by leading companies like Abbott Laboratories, Medtronic plc, and Boston Scientific Corporation, coupled with the expanding healthcare infrastructure in regions like Asia Pacific, are expected to mitigate these restraints. Emerging economies, in particular, present considerable untapped potential, driven by increasing healthcare expenditure and a growing awareness of advanced treatment options.
Balloon Introducer System Company Market Share

This in-depth report provides a definitive analysis of the global Balloon Introducer System market, exploring its intricate dynamics, growth trajectory, and competitive landscape. With a focus on coronary artery diseases, peripheral artery diseases, and neurovascular diseases, this study offers critical insights for stakeholders in the cardiovascular and endovascular device industries. We delve into key market segments including Cutting and Scoring Balloon Introducer System, Drug-Coated Balloon Introducer System, and Stent Graft Balloon Introducer System, presenting a holistic view of market evolution. The report covers the Study Period: 2019–2033, with Base Year: 2025, and a detailed Forecast Period: 2025–2033, building upon the Historical Period: 2019–2024.
Balloon Introducer System Market Dynamics & Structure
The global Balloon Introducer System market exhibits a moderately concentrated structure, with leading players like Abbott Laboratories, Medtronic plc, and Boston Scientific Corporation holding significant market shares. Technological innovation is a primary driver, fueled by advancements in catheter design, material science, and imaging compatibility, leading to improved procedural outcomes and patient safety. Regulatory frameworks, while stringent, are evolving to accommodate novel technologies that demonstrate clear clinical benefits, particularly for complex interventions in coronary, peripheral, and neurovascular applications. Competitive product substitutes include alternative delivery systems and emerging minimally invasive techniques, though balloon introducer systems remain a cornerstone in interventional cardiology and radiology. End-user demographics are shifting towards an aging global population with a higher prevalence of cardiovascular and cerebrovascular conditions, augmenting demand. Mergers and acquisitions (M&A) activity is notable, with companies strategically acquiring innovative technologies and expanding their product portfolios to capture market share and diversify offerings. For instance, the past five years have seen an average of 10 M&A deals annually, with valuations often exceeding $100 million. Key innovation barriers include the high cost of R&D, lengthy clinical trial durations, and reimbursement challenges for novel devices.
Balloon Introducer System Growth Trends & Insights
The global Balloon Introducer System market is poised for robust growth, projected to reach an estimated $X.XX billion in 2025 and expanding to approximately $Y.YY billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of Z.ZZ% during the forecast period. This expansion is underpinned by a confluence of factors, including the escalating global burden of cardiovascular diseases (CVDs), such as coronary artery disease (CAD) and peripheral artery disease (PAD), alongside a rising incidence of neurovascular disorders. These chronic conditions necessitate frequent interventional procedures, directly driving the demand for advanced balloon introducer systems. The increasing adoption of minimally invasive techniques over traditional open surgeries is a significant trend, favoring the use of sophisticated introducer systems that facilitate precise navigation and deployment within challenging vascular anatomies. Technological advancements are playing a pivotal role in shaping market dynamics. The development of low-profile catheters, improved steerability, enhanced imaging compatibility, and the integration of drug-eluting technologies on balloons are revolutionizing treatment paradigms. These innovations not only improve procedural efficacy and patient outcomes but also broaden the scope of applications for balloon introducer systems, including complex lesions and previously inoperable cases.
Consumer behavior is also influencing market growth. Patients are increasingly seeking less invasive treatment options with shorter recovery times and improved quality of life. This patient preference, coupled with physician advocacy for advanced interventional tools, is accelerating the uptake of balloon introducer systems. Furthermore, the growing healthcare expenditure in emerging economies, coupled with improving healthcare infrastructure and increased access to advanced medical technologies, presents significant market penetration opportunities. The prevalence of conditions like diabetes and hypertension, which are major risk factors for cardiovascular and cerebrovascular diseases, further contributes to the sustained demand for these critical medical devices. The market penetration for advanced balloon introducer systems, particularly in coronary and peripheral interventions, is expected to rise from approximately 65% in 2025 to over 80% by 2033 in developed regions.
Dominant Regions, Countries, or Segments in Balloon Introducer System
The Coronary Artery Diseases (CAD) segment, within the Application category, stands as the dominant force driving growth in the global Balloon Introducer System market. This dominance is attributed to the persistently high and growing prevalence of CAD worldwide, characterized by atherosclerosis leading to the narrowing or blockage of coronary arteries. Interventional cardiology procedures, such as percutaneous coronary intervention (PCI), heavily rely on balloon introducer systems for balloon angioplasty and stent deployment. The market for CAD-focused balloon introducer systems is projected to account for an estimated 60% of the total application market share by 2025, valued at approximately $A.AA billion.
Key drivers for the dominance of the CAD segment include:
- Epidemiological Trends: Cardiovascular diseases remain the leading cause of death globally, with CAD being the most common manifestation. An aging population, sedentary lifestyles, and the increasing incidence of risk factors like obesity, hypertension, and diabetes contribute to a continuous and escalating need for CAD interventions.
- Technological Advancements: Innovations in balloon catheter technology, including low-profile designs, high-pressure balloons, drug-coated balloons, and specialized balloons for calcified lesions, are crucial for treating complex CAD anatomies effectively. Companies like Abbott Laboratories and Boston Scientific Corporation are at the forefront of these advancements.
- Healthcare Infrastructure and Reimbursement: Developed regions, particularly North America and Europe, possess well-established healthcare infrastructures, advanced medical facilities, and robust reimbursement policies that support elective and complex interventional procedures. This enables widespread adoption of advanced balloon introducer systems.
- Physician Expertise and Training: A highly skilled and trained workforce of interventional cardiologists ensures the effective utilization of sophisticated balloon introducer systems, leading to better patient outcomes and driving market demand.
- Market Share and Growth Potential: The substantial installed base of interventional cardiology labs and the continuous pipeline of new product development specifically tailored for CAD interventions solidify its leading position and promising growth potential. The projected market growth for CAD applications is expected to remain robust, with a CAGR of around 5.5% during the forecast period.
While Peripheral Artery Diseases (PAD) and Neurovascular Diseases also represent significant and growing segments, their current market share and growth trajectory are somewhat less pronounced compared to CAD. PAD interventions are gaining traction with advancements in device technology, and neurovascular interventions are witnessing rapid innovation, but the sheer volume and established nature of CAD interventions ensure its continued market leadership.
Balloon Introducer System Product Landscape
The balloon introducer system product landscape is characterized by continuous innovation focused on enhancing deliverability, precision, and patient safety. Cutting and scoring balloon introducer systems offer enhanced lesion crossing capabilities by disrupting fibrous tissue, facilitating better balloon angioplasty and stent deployment. Drug-Coated Balloon (DCB) introducer systems are gaining significant traction, delivering therapeutic agents directly to the vessel wall to prevent restenosis, thereby reducing the need for repeat interventions. Stent Graft Balloon Introducer Systems are designed for precise deployment of stent grafts in complex aortic aneurysms and dissections. These products feature advanced materials, low-profile designs for navigating tortuous vasculature, and improved lubricity to minimize friction and trauma during insertion. Unique selling propositions include enhanced trackability, rapid balloon inflation/deflation times, and superior force transmission.
Key Drivers, Barriers & Challenges in Balloon Introducer System
Key Drivers:
- Rising prevalence of cardiovascular and cerebrovascular diseases: An aging global population and the increasing incidence of lifestyle-related conditions like diabetes and hypertension are major drivers.
- Technological advancements: Development of low-profile, highly steerable, and drug-eluting balloon introducer systems enhances treatment efficacy and expands applications.
- Growth of minimally invasive procedures: Shifting preference towards less invasive interventions fuels demand for advanced delivery systems.
- Increasing healthcare expenditure: Growing investments in healthcare infrastructure, especially in emerging economies, expand market access.
Key Barriers & Challenges:
- High R&D costs and regulatory hurdles: Developing and obtaining approval for new devices is time-consuming and expensive.
- Reimbursement policies: Inconsistent or unfavorable reimbursement rates in certain regions can hinder market adoption.
- Competitive pressures: A crowded market with established players and emerging technologies necessitates continuous innovation and cost-effectiveness.
- Supply chain disruptions: Global events can impact the availability of raw materials and manufacturing capacity.
- Stringent quality control requirements: Ensuring the safety and efficacy of medical devices involves rigorous testing and compliance.
Emerging Opportunities in Balloon Introducer System
Emerging opportunities lie in the development of highly specialized introducer systems for complex neurovascular interventions, including those targeting acute ischemic stroke. The expanding use of drug-coated balloons in peripheral interventions, particularly for femoropopliteal arteries, presents a significant growth avenue. Furthermore, advancements in bioresorbable materials for balloon catheters and the integration of real-time imaging feedback systems during procedures could revolutionize treatment delivery and patient outcomes. Untapped markets in lower- and middle-income countries, with improving healthcare access, also represent substantial long-term potential.
Growth Accelerators in the Balloon Introducer System Industry
Technological breakthroughs in biomaterials and nanotechnology are poised to accelerate growth by enabling the development of even smaller, more flexible, and more effective balloon introducer systems. Strategic partnerships between device manufacturers and research institutions will foster innovation and expedite the translation of novel technologies into clinical practice. Market expansion strategies, focusing on the increasing demand for outpatient procedures and same-day discharge protocols, will drive the adoption of user-friendly and highly reliable introducer systems. The growing emphasis on preventative cardiology and early intervention will further stimulate the market for advanced diagnostic and therapeutic delivery tools.
Key Players Shaping the Balloon Introducer System Market
- Abbott Laboratories
- B. Braun Melsungen AG
- MicroPort Scientific Corporation
- OrbusNeich Medical
- Terumo Corporation
- Biotronik AG
- Koninklijke Philips N.V.
- Cardinal Health Inc.
- Boston Scientific Corporation
- Cardionovum GmbH
- Cook Group
- Hexacath
- Medtronic plc
- Concept Medical Inc.
- Becton Dickinson and Company
Notable Milestones in Balloon Introducer System Sector
- 2019: Launch of next-generation drug-coated balloon catheters with improved drug elution profiles and expanded size offerings.
- 2020: Introduction of novel microcatheters with enhanced pushability and trackability for complex neurovascular procedures.
- 2021: Significant increase in M&A activity focused on acquiring innovative stent graft delivery systems.
- 2022: Regulatory approvals for advanced scoring balloon catheters designed for severely calcified lesions.
- 2023: Growing market penetration of bioresorbable balloon catheters in certain niche applications.
- 2024: Continued research and development into AI-assisted navigation for introducer systems.
In-Depth Balloon Introducer System Market Outlook
The future outlook for the Balloon Introducer System market remains exceptionally strong, driven by a synergistic interplay of escalating disease burdens, continuous technological innovation, and evolving healthcare practices. Growth accelerators such as advancements in drug-eluting technologies, the development of ultra-low profile catheters for challenging anatomies, and the expanding scope of percutaneous interventions will continue to propel market expansion. Strategic collaborations between manufacturers and healthcare providers will further refine product development and adoption. The increasing focus on patient-centric care and the demand for less invasive, faster recovery procedures will solidify the indispensable role of advanced balloon introducer systems in modern interventional medicine. The market is well-positioned for sustained growth, with significant opportunities in both established and emerging geographical regions.
Balloon Introducer System Segmentation
-
1. Application
- 1.1. Coronary Artery Diseases
- 1.2. Peripheral Artery Diseases
- 1.3. Neurovascular Diseases
- 1.4. Others
-
2. Types
- 2.1. Cutting and Scoring Balloon Introducer System
- 2.2. Drug-Coated Balloon Introducer System
- 2.3. Stent Graft Balloon Introducer System
- 2.4. Others
Balloon Introducer System Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
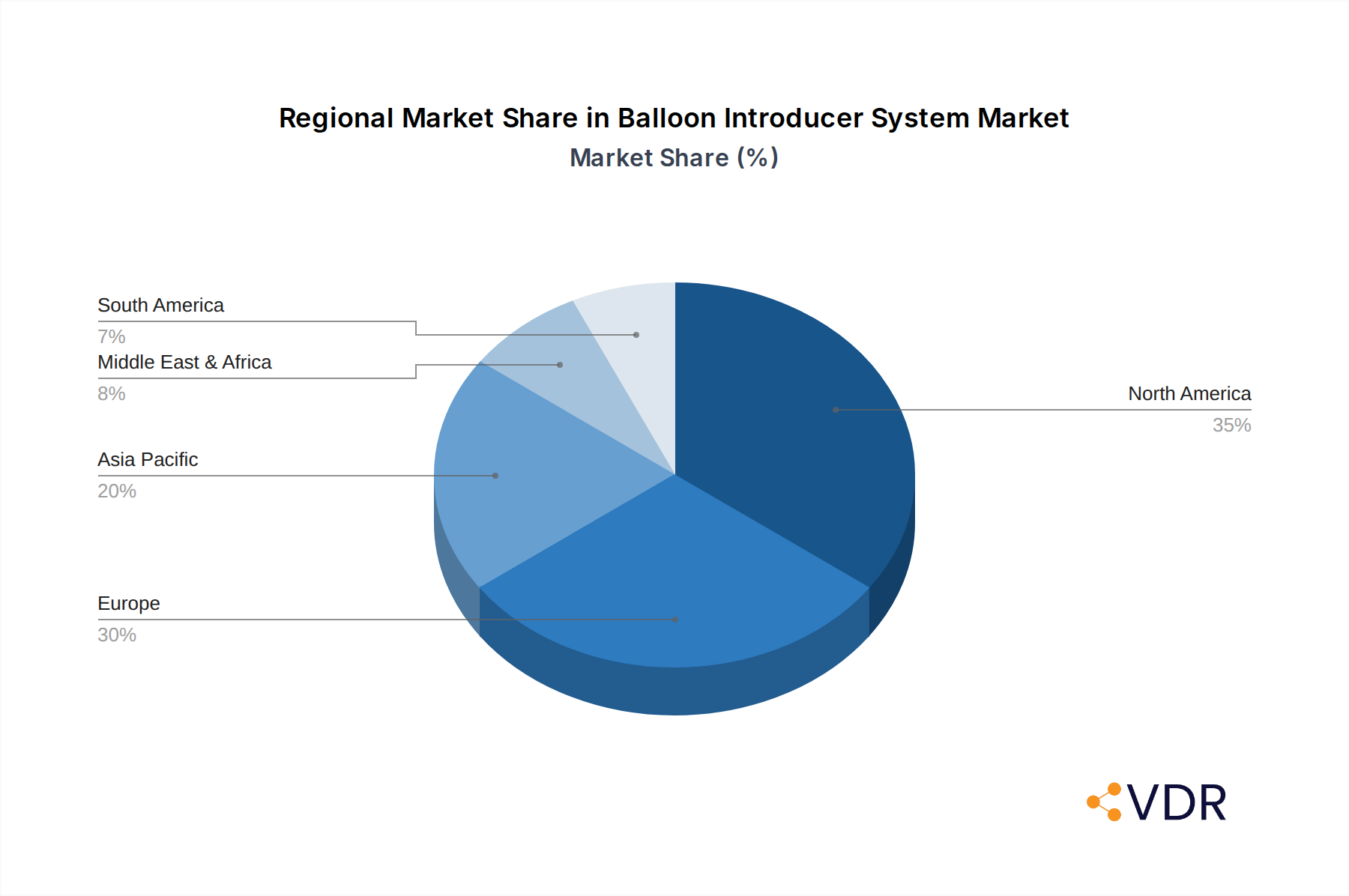
Balloon Introducer System Regional Market Share

Geographic Coverage of Balloon Introducer System
Balloon Introducer System REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.9% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. VDR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Coronary Artery Diseases
- 5.1.2. Peripheral Artery Diseases
- 5.1.3. Neurovascular Diseases
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Cutting and Scoring Balloon Introducer System
- 5.2.2. Drug-Coated Balloon Introducer System
- 5.2.3. Stent Graft Balloon Introducer System
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Balloon Introducer System Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Coronary Artery Diseases
- 6.1.2. Peripheral Artery Diseases
- 6.1.3. Neurovascular Diseases
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Cutting and Scoring Balloon Introducer System
- 6.2.2. Drug-Coated Balloon Introducer System
- 6.2.3. Stent Graft Balloon Introducer System
- 6.2.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Balloon Introducer System Analysis, Insights and Forecast, 2021-2033
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Coronary Artery Diseases
- 7.1.2. Peripheral Artery Diseases
- 7.1.3. Neurovascular Diseases
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Cutting and Scoring Balloon Introducer System
- 7.2.2. Drug-Coated Balloon Introducer System
- 7.2.3. Stent Graft Balloon Introducer System
- 7.2.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Balloon Introducer System Analysis, Insights and Forecast, 2021-2033
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Coronary Artery Diseases
- 8.1.2. Peripheral Artery Diseases
- 8.1.3. Neurovascular Diseases
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Cutting and Scoring Balloon Introducer System
- 8.2.2. Drug-Coated Balloon Introducer System
- 8.2.3. Stent Graft Balloon Introducer System
- 8.2.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Balloon Introducer System Analysis, Insights and Forecast, 2021-2033
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Coronary Artery Diseases
- 9.1.2. Peripheral Artery Diseases
- 9.1.3. Neurovascular Diseases
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Cutting and Scoring Balloon Introducer System
- 9.2.2. Drug-Coated Balloon Introducer System
- 9.2.3. Stent Graft Balloon Introducer System
- 9.2.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Balloon Introducer System Analysis, Insights and Forecast, 2021-2033
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Coronary Artery Diseases
- 10.1.2. Peripheral Artery Diseases
- 10.1.3. Neurovascular Diseases
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Cutting and Scoring Balloon Introducer System
- 10.2.2. Drug-Coated Balloon Introducer System
- 10.2.3. Stent Graft Balloon Introducer System
- 10.2.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Balloon Introducer System Analysis, Insights and Forecast, 2021-2033
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Coronary Artery Diseases
- 11.1.2. Peripheral Artery Diseases
- 11.1.3. Neurovascular Diseases
- 11.1.4. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Cutting and Scoring Balloon Introducer System
- 11.2.2. Drug-Coated Balloon Introducer System
- 11.2.3. Stent Graft Balloon Introducer System
- 11.2.4. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Abbott Laboratories
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 B. Braun Melsungen AG
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 MicroPort Scientific Corporation
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 OrbusNeich Medical
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Terumo Corporation.
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Biotronik AG
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Koninklijke Philips N.V.
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Cardinal Health Inc.
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Boston Scientific Corporation
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Cardionovum GmbH
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 Cook Group
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Hexacath
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 Medtronic plc
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 Concept Medical Inc.
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 Becton Dickinson and Company
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.1 Abbott Laboratories
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Balloon Introducer System Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: Global Balloon Introducer System Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Balloon Introducer System Revenue (billion), by Application 2025 & 2033
- Figure 4: North America Balloon Introducer System Volume (K), by Application 2025 & 2033
- Figure 5: North America Balloon Introducer System Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Balloon Introducer System Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Balloon Introducer System Revenue (billion), by Types 2025 & 2033
- Figure 8: North America Balloon Introducer System Volume (K), by Types 2025 & 2033
- Figure 9: North America Balloon Introducer System Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Balloon Introducer System Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Balloon Introducer System Revenue (billion), by Country 2025 & 2033
- Figure 12: North America Balloon Introducer System Volume (K), by Country 2025 & 2033
- Figure 13: North America Balloon Introducer System Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Balloon Introducer System Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Balloon Introducer System Revenue (billion), by Application 2025 & 2033
- Figure 16: South America Balloon Introducer System Volume (K), by Application 2025 & 2033
- Figure 17: South America Balloon Introducer System Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Balloon Introducer System Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Balloon Introducer System Revenue (billion), by Types 2025 & 2033
- Figure 20: South America Balloon Introducer System Volume (K), by Types 2025 & 2033
- Figure 21: South America Balloon Introducer System Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Balloon Introducer System Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Balloon Introducer System Revenue (billion), by Country 2025 & 2033
- Figure 24: South America Balloon Introducer System Volume (K), by Country 2025 & 2033
- Figure 25: South America Balloon Introducer System Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Balloon Introducer System Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Balloon Introducer System Revenue (billion), by Application 2025 & 2033
- Figure 28: Europe Balloon Introducer System Volume (K), by Application 2025 & 2033
- Figure 29: Europe Balloon Introducer System Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Balloon Introducer System Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Balloon Introducer System Revenue (billion), by Types 2025 & 2033
- Figure 32: Europe Balloon Introducer System Volume (K), by Types 2025 & 2033
- Figure 33: Europe Balloon Introducer System Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Balloon Introducer System Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Balloon Introducer System Revenue (billion), by Country 2025 & 2033
- Figure 36: Europe Balloon Introducer System Volume (K), by Country 2025 & 2033
- Figure 37: Europe Balloon Introducer System Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Balloon Introducer System Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Balloon Introducer System Revenue (billion), by Application 2025 & 2033
- Figure 40: Middle East & Africa Balloon Introducer System Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Balloon Introducer System Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Balloon Introducer System Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Balloon Introducer System Revenue (billion), by Types 2025 & 2033
- Figure 44: Middle East & Africa Balloon Introducer System Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Balloon Introducer System Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Balloon Introducer System Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Balloon Introducer System Revenue (billion), by Country 2025 & 2033
- Figure 48: Middle East & Africa Balloon Introducer System Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Balloon Introducer System Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Balloon Introducer System Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Balloon Introducer System Revenue (billion), by Application 2025 & 2033
- Figure 52: Asia Pacific Balloon Introducer System Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Balloon Introducer System Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Balloon Introducer System Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Balloon Introducer System Revenue (billion), by Types 2025 & 2033
- Figure 56: Asia Pacific Balloon Introducer System Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Balloon Introducer System Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Balloon Introducer System Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Balloon Introducer System Revenue (billion), by Country 2025 & 2033
- Figure 60: Asia Pacific Balloon Introducer System Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Balloon Introducer System Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Balloon Introducer System Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Balloon Introducer System Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Balloon Introducer System Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Balloon Introducer System Revenue billion Forecast, by Types 2020 & 2033
- Table 4: Global Balloon Introducer System Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Balloon Introducer System Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Global Balloon Introducer System Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Balloon Introducer System Revenue billion Forecast, by Application 2020 & 2033
- Table 8: Global Balloon Introducer System Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Balloon Introducer System Revenue billion Forecast, by Types 2020 & 2033
- Table 10: Global Balloon Introducer System Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Balloon Introducer System Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Global Balloon Introducer System Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United States Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Canada Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Mexico Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Balloon Introducer System Revenue billion Forecast, by Application 2020 & 2033
- Table 20: Global Balloon Introducer System Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Balloon Introducer System Revenue billion Forecast, by Types 2020 & 2033
- Table 22: Global Balloon Introducer System Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Balloon Introducer System Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Global Balloon Introducer System Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Brazil Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Argentina Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Balloon Introducer System Revenue billion Forecast, by Application 2020 & 2033
- Table 32: Global Balloon Introducer System Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Balloon Introducer System Revenue billion Forecast, by Types 2020 & 2033
- Table 34: Global Balloon Introducer System Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Balloon Introducer System Revenue billion Forecast, by Country 2020 & 2033
- Table 36: Global Balloon Introducer System Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 40: Germany Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: France Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: Italy Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Spain Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 48: Russia Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 50: Benelux Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 52: Nordics Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Balloon Introducer System Revenue billion Forecast, by Application 2020 & 2033
- Table 56: Global Balloon Introducer System Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Balloon Introducer System Revenue billion Forecast, by Types 2020 & 2033
- Table 58: Global Balloon Introducer System Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Balloon Introducer System Revenue billion Forecast, by Country 2020 & 2033
- Table 60: Global Balloon Introducer System Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 62: Turkey Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 64: Israel Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 66: GCC Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 68: North Africa Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 70: South Africa Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Balloon Introducer System Revenue billion Forecast, by Application 2020 & 2033
- Table 74: Global Balloon Introducer System Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Balloon Introducer System Revenue billion Forecast, by Types 2020 & 2033
- Table 76: Global Balloon Introducer System Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Balloon Introducer System Revenue billion Forecast, by Country 2020 & 2033
- Table 78: Global Balloon Introducer System Volume K Forecast, by Country 2020 & 2033
- Table 79: China Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 80: China Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 82: India Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 84: Japan Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 86: South Korea Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 90: Oceania Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Balloon Introducer System Revenue (billion) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Balloon Introducer System Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Balloon Introducer System?
The projected CAGR is approximately 9.9%.
2. Which companies are prominent players in the Balloon Introducer System?
Key companies in the market include Abbott Laboratories, B. Braun Melsungen AG, MicroPort Scientific Corporation, OrbusNeich Medical, Terumo Corporation., Biotronik AG, Koninklijke Philips N.V., Cardinal Health Inc., Boston Scientific Corporation, Cardionovum GmbH, Cook Group, Hexacath, Medtronic plc, Concept Medical Inc., Becton Dickinson and Company.
3. What are the main segments of the Balloon Introducer System?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 4.77 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3350.00, USD 5025.00, and USD 6700.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Balloon Introducer System," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Balloon Introducer System report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Balloon Introducer System?
To stay informed about further developments, trends, and reports in the Balloon Introducer System, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
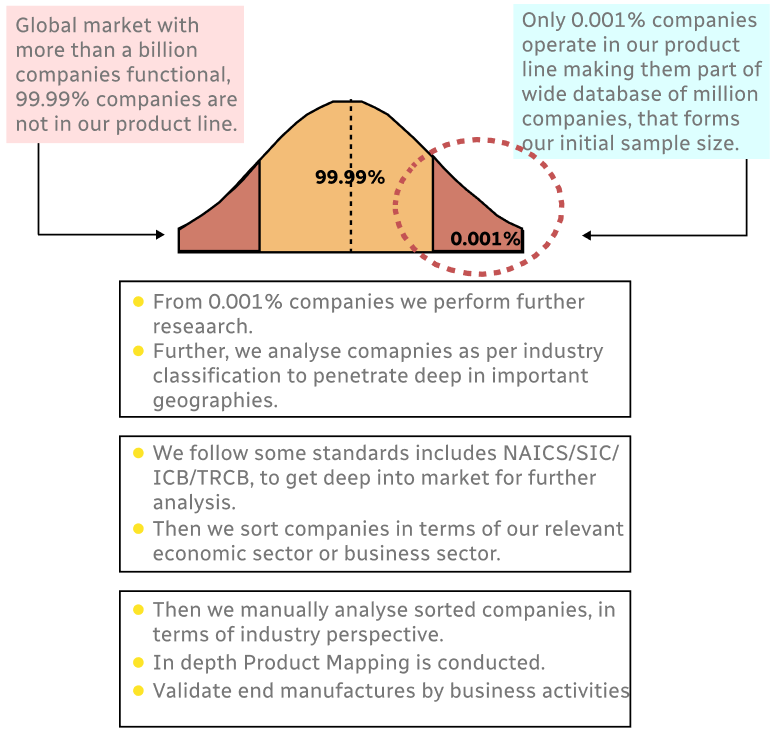
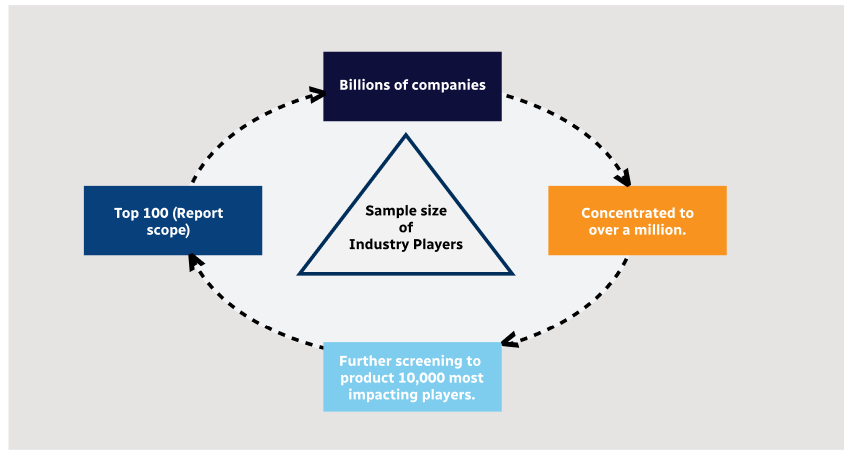
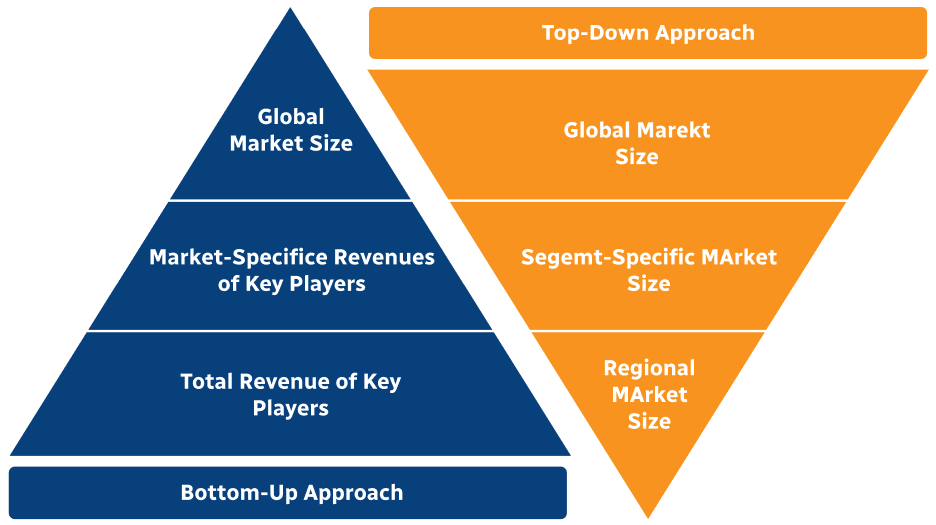
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


